Pollution
1/140
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
141 Terms
What is pollution
The discharge of solids, liquids, gases, or energy into the environment that cause harm to the environment and living organisms
Primary pollutant
A pollutant emitted by human activity
Secondary pollutant
A pollutant produced via chemical reactions between one or more primary pollutants and environmental features eg light
All the properties of pollutants
Energy form, density, persistence, toxicity, specificity, adsorption, mobility, solubility, bioaccumulation, biomagnification, synergism, mutagens, teratogenic
Properties of pollutants (teratogenic)
Terotogens act upon unborn babies in the womb by preventing normal gene expression which can lead to birth defects caused by dysfunctional proteins/enzymes being produced, they don’t impact DNA structure they just prevent some genes from being expressed
Properties of pollutants (Mutagens)
Mutagens or mutagenic agents increase the chance of a mutation to DNA occurring such as UV and ionising radiation.
Carcinogens are specific types of mutagens that cause cancer, the mutations caused in leads lead to uncontrollable cell division so it replicates at a very high rate forming tumours
Properties of pollutants (synergism)
When the effects of two or more pollutants combine to produce a heightened effect
Eg. Tropospheric ozone damages lead tissue creating holes which enables acid rain to damage the increased number of exposed cells

Properties of pollutants (biomagnification)
Increase in concentration of the pollutant as you move up the food chain, producers having the lowest concentration while secondary/tertiary consumers have the highest, this is because each high tropic level consumes multiple organisms from the level below concentrating the pollutant

Properties of Pollutants (Bioaccumulation)
The gradual buildup of a pollutant in an organism over time which resukts from long term exposure often through the food chain leading to toxic levels causing illness or death in the organism
Properties of pollutants (Solubility)
Pollutants may be water or lipid soluble, being water soluble means they dissolve easy in water exposing organisms living there to that pollutant so can increase its toxicity for example fish as they pass water over their gills to gain oxygen
Being lipid soluble means they can dissolve in lipids so in terms of living organs is this means a pollutant can dissolve through a cell membrane and enter cells which contain DNA so can increase the risk of mutations or DNA damage, these are always able to biomagnify snd bioaccumulate
Properties of pollutants (mobility)
The ability of a pollutant to move around in the environment and therefore disperse longer distances, the more mobile the further it can travel expanding the effected area, wether a pollutant is mobile depends on the state of matter, adsorption, persistence and density
Properties of pollutants (Adsorption)
Occurs when a pollutant can attach to the surface of environmental materials such as soil, clay particles in the soil can adsorb charged particles to their surface, if a pollutant is attached to the surface of something it’s less likely to be able to move and disperse throughout the environment reducing its impact, adsorption can concentrate pollutants in smaller areas leading to toxic levels
Absorption vs adsorption
Absorption- the adhesion of particles ON TO the surface of a substance
Adsorption- the particles of a substance enter IN TO the volume/bulk of another
Properties of pollutants (specificity)
The number of organisms it can negatively impact, pollutants with a high specificity will only impact a few/one organisms whereas those with a low specificity will be toxic to a wider range of species increasing their negative impact on the environment
Eg. Pesticides on crops- farmers prefer non specific pesticides to kill a wider range of pests yet it’s better for the environment to use specific ones as they target fewer non-pest organisms reducing harm to non target species
Properties of pollutants (energy form)
Energy form eg. Noise, heat and light, the energy form a pollutant has will alter how the pollutant behaves in the environment and the areas it will impact
Some energy forms dissipate quickly like heat whereas others remain in the environment for longer eg radiation increasing damage to living organisms
Properties of pollutants (toxicity)
How poisonous a pollutant is to living organisms, usually toxins target proteins inside living organisms such as enzymes, disrupting metabolic processes causing illness or death
For example carbon monoxide prevents our blood proteins from binding to oxygen to transport it to our respiring cells, pollutants with a higher toxicity will have more negative impacts on the environment generally
Properties of pollutants (density)
The quantity of mass per unit volume of a substance, a pollutant with higher density will have more mass per unit volume and vice versa
This impacts:
Movement- low density pollutants require more energy to move and travel shorter distances
Water impact- whether a pollutant floats or sinks affecting the ability of water transport the pollutant
Environment- floating pollutants block light penetration in water leading to death of plants
Cleanup- floating pollutants can be skimmed, sinking ones need dredging
Properties of pollutants (persistence)
A measure of the amount of time a pollutant remains in the environment
Pollutants with a high persistence remain in the environment a long time before being broken down
Persistence is measured by using half lives which is the amount of time it takes half of the pollutant to break down, rate of breakdown can be linked to its reactivity with abiotic factors in the environemnt eg does it react with light, oxygen or water so if so it’s likely to break down quickly in the environment and have low persistence
High persistence pollutants remain in the environment longer increasing exposure for organisms, which can disperse over larger areas causing widespread environmental damage
4 types of degradation that reduce persistence
Biodegradation- caused by living organisms such as bacteria and fungi
Phytodegradation- caused by plants
Photodegradation- caused by reactions with light eg UV light
Thermal degradation- caused by reactions with heat
Whether a pollutant is solid, liquid or gas it can impact two things:
The severity of the impact the pollutant has when released into environment
The control methods we may use to clean up the pollutant
State of matter
Determined if a pollutant is solid liquid and gas, it affects how easily it spreads and the area impacted, helps predict and manage affected areas, air one gases require specific control methods for collection and storage
Features of the environment that may affect how quickly a pollutant reacts and degrades
Abiotic factors
Temp- higher temps lead to particles having more kinetic energy so more likely they’ll collide and react (increased reaction rate)
Light- for example UV can cause photolysis -the splitting of compounds leading to production of non pollutants or secondary pollutants
Oxygen- needed for aerobic respiration in aerobic species of bacteria/fungi which may break down pollutants (biodegradation), also increases solubility of some metals in water
pH- acidic conditions make some pollutants such as heavy metal more double increasing chance of breakdown
Other pollutants- synergism where effects combine or production of secondary pollutants
Features of the environment that affect how far the pollutant is dispersed (Wind/water/topography)
Abiotic Factors
Wind/water currents- increase dispersal of a pollutant, the velocity and direction of travel of these currents can move the pollutant to new areas and recuse concentration of it, the ease of this depends on certain properties: state of matter, mobility, density, persistence, water solubility, adsorption
Examples of these environments- rivers, streams ocean
Topography can also affect the rate of dispersal, if the area has steep long slopes then the velocity will be greater, less likely to travel further and disperse the pollutants more than on flat land
Permeable/impermeable ground- permeable ground allows infiltration and percolation of water into the soil/groundwater source which can then feed into rivers/lakes, impermeable ground leads to increased surface runoff so more chance of water flowing straight into nearby water bodies

Features of the environment that affect how far the pollutant is dispersed (Temp inversion and adsorption)
When a warm pollutant gas is released into the troposphere in normal conditions it’s heat will mean it’s less dense than the cooler air so will rise and disperse as a result,
but with a temperature inversion where there is a small section of the troposphere that gets warmer as altitude increases (not normal conditions) any pollutants released here will become cold and dense and not be able to rise above less dense warm layer preventing dispersal
Adsorption- if pollutants are immobilised they can’t be dispersed
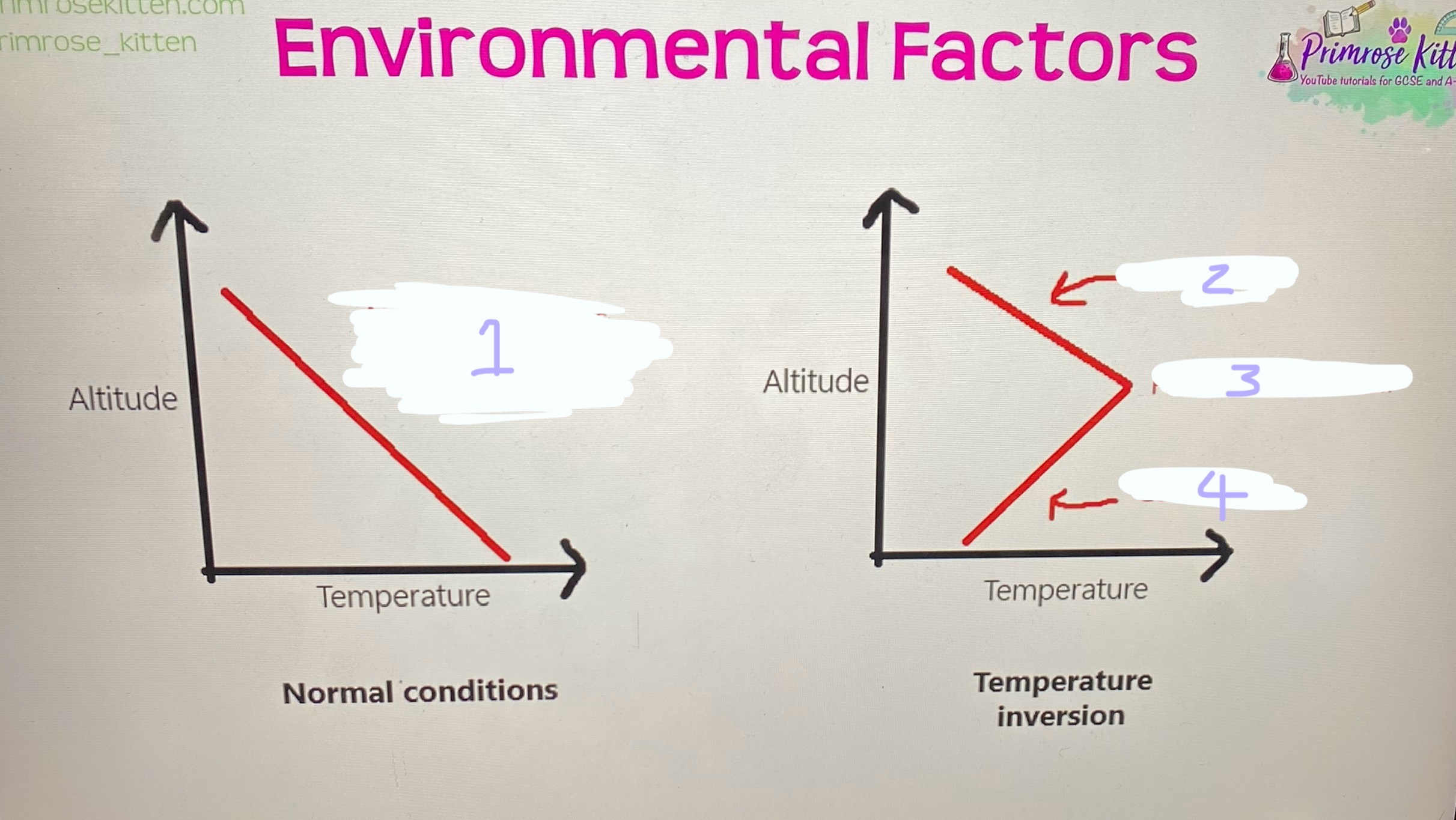
Temperature inversion shown on a graph
1- air gets cooler as you increase the altitude
2- colder air
3- pocket of warm air
4- colder air
Environmental features that make a temperature inversion more likely
Valleys- provides a confined area where it’s easier for cold air to accumulate without mixing with upper layers to warm it
Low wind velocity- reduces the mixing of air between layers causing an increased likelihood of cold lower layer forming
Cloud cover - cloudless skies mean any IR released from the ground can rise easily cooling the lower layers of the atmosphere down faster, if clouds were present they would insulate the IR and prevent it from escaping as quickly
High mist/fog- have high albedo reflecting UV away from the ground so UV can’t be absorbed and converted to IR increasing the likelihood of a cold lower layer forming
General Pollutant control
CPA (critical pathway analysis- predicts the rout that a pollutant will take once it entered the environment, to accurately predict this you need to know the pollutants properties and the environmental features it’s released into, this allows most vulnerable areas in the pathway to be protected and prioritized
CGM (critical group monitoring- studies gather a group of people most vulnerable to the pollutant if it was released due to lifestyle factors such as smoking in, this is the critical group, if the risk is deemed low enough for this group then for others the assumed risk is lower so the pollutant may be deemed safe for release
Releasing pollutant downwind so it is blown away from urban areas instead of up
Releasing them into an area of choppy waters so the currents can disperse and dilute the pollutant
Releasing when the tide is on its way out instead of in so it’s carried out to sea instead of towards land
General principles of pollution control
Polluter pays principle- those responsible for causing pollution shoukd be responsible for paying for its prevention/clean up done by giving fines to companies for example,
D- this is hard to monitor as pollutants can be released across multiple locations and once released it’s difficult to know who released it definitively
Precautionary principle- every new chemical/product is assumed to be polluting unless it’s proven otherwise by detailed research,
A- this means that companies can’t use a lack of knowledge as an excuse for releasing large scale pollutants with CFCs for example
Smoke pollution overview
Causes- Smoke is released during incomplete combustion of organic molecules containing carbon such as incomplete combustion of fossil fuels in vehicle engines, smoke can be referred to as particulate matter because there are a range of sizes (pm10, pm5, pm1)
Smaller particles likely to be more mobile because lower density so remain in atmosphere for longer periods of time -more persistent and likely to be inhaled
Smoke contains toxins including heavy metals leading to damage lunge tissue when inhaled, can act synergistically with other pollutants like sulphur dioxide to create heightened negative impacts
Properties of smoke
Low density
High mobility
Toxic
Smaller particles more persistent
State of matter- solid
Synergistic
Primary pollutant
Reactive
Effects of smoke pollution
Living-
in humans/mammals smoke pollution affects the respiratory system as it’s inhaled increasing disease like bronchitis/asthma, people with existing lung conditions are especially vulnerable (CGM)
Smoke reduces the rate of photosynthesis in plants as it can block UV light from reaching their leaves, toxins carried in smoke can damage leaf tissue making it more vulnerable
Climate-
Smoke has high albedo as it reflects UV back into the atmosphere so less is absorbed by the ground and converted to IR, leads to reduced temps in areas
Smoke is an ozone depleting substance contributing to ozone holes increasing damaging UV radiation that can reach earths surface and cause mutations
Control methods of smoke pollution
Legislation- The clean air act- created smokeless zones in large urban areas where the use of fuels that produced smoke were heavily restricted, it also banned bonfires for a period of time and stated that dorsal cars shoukd have smoke emissions regularly tested
Transport-
because of the clean air act diesel cars now have Diesel particulate filters (DPFs) fitted into their exhausts trapping smoke particles in the mesh to reduce the amount released into the atmosphere
Turbos in engines increases the efficiency of the combustion of the fuel by supplying more oxygen
Driverless cars should prevent over-acceleration that can happen when humans drive more high can often lead to too much fuel entering the engine without enough oxygen to combust it completely
Industrial sources-
electrostatic precipitators- effluent gas passed through a chamber with electrically charged plates
cyclone separators- based on the principles of vacuums
scrubber- uses a fine water spray applied to effluent gases in a chamber to wash out suspended particles and dissolve gaseous pollutants
Coal treatment- removing tar in coal producing smokeless coal
Bag filters- remove smoke particles from effluence gases by trapping them on a fabric filter
Photochemical smog
Secondary pollutant formed by the combination of nitrous oxides, tropospheric ozone and it burned hydrocarbons in the troposphere using UV light,
Sources of photochemical smog
Nitrous oxides- Released in exhaust gases from combustion in vehicle engines, formed when nitrogen and oxygen in the air combine under high pressure and temperatures in the engine
Tropospheric ozone- secondary pollutant that is formed when nitrous oxides are split by UV light (photolysis) to release a monoatomic oxygen atom which can then combine with a dip tonic molecule to form 03
Unburied hydrocarbons- released in exhaust gases and are formed form unburnt fuel in the engine
These three are required as well as UV light from the sun, the formation process happens when nitrous oxides are split to form tropospheric ozone which then combined with nitrous oxides and unburnt hydrocarbons to produce the photochemical smog which is known as peroxyacetyl nitrates (PANs)
Properties photochemical smog
Secondary pollutant
State of matter- gas
Toxic
Mobile
Persistent- depending on where it forms and the conditions in that area it can last for weeks
Cyclone separators
Used in industry as effluent gases are fed into a cylinder shaped chamber where they are forced to go around the edges, the motion of the gases causes smoke particles to be thrown to the outer surfaces of the cylinder as they are denser than the gases, the smoke particles can then fall and be collided as ash and put into landfill releasing smoke free gases

Electrostatic precipitators
Used in coal fired power plants consisting of electrically charged plates, waste (effluent) gas passes between the plates, the electrical charge attracts the smoke particles and they adsorb to the plate surface, once the gas has passed through the plates can be turned off, causing the smoke particles to fall this is then collected as ash which can be disposed of in landfill
Smog
if smoke is released on a foggy day then the smoke and fog will combine to form smoke smog, smoke smog is more easily inhaled than smoke, which increases the risk of respiratory illnesses and diseases, it also has a very high albedo reflecting more UV light away into the atmosphere
Temp inversions causing fog
Temperature inversions make it more likely for fog to form as colder air trapped near the ground in a temperature inversion will cool the water vapour to dew point forming fog, once the fog has formed it will reflect UV light meaning it will take longer for the ground to warm up again and break the inversion
Smoke smog case study for negative impacts
London in the mid 90s had a lot of smoke pollution due to large numbers of factories and vehicles in an area, it’s located in a valley and this event happened when there was low wind velocities and clear skies, the combination lead to a temperature inversion trapping all that smoke produced by cars and factories close to the ground forming smog. It was so concentrated that people had to wear masks to go out safely
What is fog
Moist air that has cooled until the water reaches its dew point and becomes liquid again
Effects of photochemical smog
Living- respiratory system and eyes, they are potent at low concentrations meaning they can have a negative affect even in small doses,
Reduces the growth rate of plant soecies reducing productivity as well as causing damage to the leaf surface, they can also corrode metals and any surfaces that have been painted
What makes photochemical smog more likely?
Valleys: the topography makes it more likely for pollutants to concentrate without dissipating
Sunny conditions: as sunlight (UV) needed for photolysis reactions high numbers of cars and industry: they release the nitrous oxides and unburnt hydrocarbons
Temp inversions: they would prevent the exhaust gases from rising and therefore allow the concentration to build up in lower levels of the troposphere making it more likely photochemical smog will form
Methods to prevent/reduce photochemical smog formation
its a secondary pollutant so to control it we need to reduce the release the release of the primary pollutants into the atmosphere, without them then the chemical reactions can’t occur to produce PANS
Nitrous oxide- catalytic converters into vehicle exhausts which contain metal catalysts eg platinum, this reduce nitrous oxides back to nitrogen and oxygen before they are released into the atmosphere this then prevents the production of tropospheric ozone. These also worl to oxidise urnburned hydrocarbons into c02 and h20 preventing release into the atmosphere
-can also collect effluent gases which are passed over an activated carbon filter
Carbon monoxide- released from vehicle engines due to incomplete combustion, odourless colourless gas which binds to the protein in our blood called haemoglobin so oxygen can’t be carried to our respiring cells, catalytic converters can also be used to prevent these release of carbon monoxide as they oxidise it to form carbon dioxide instead
Atmospheric pollutants
Acid rain, photochemical smog, smoke pollution
Acid rain overview
Any precipitation that has a pH reading lower than 5.6 as most rain is slightly acidic naturally
Acid rain causes
the release of atmospheric pollutants which mix with water ro produce acids
Oxides of sulphur- released during the combustion of fossil fuels, when they mix with water vapour in the atmosphere, sulphur dioxide (s02) forms sulphurous acid and sulphur trioxide (s02) forms sulphuric acid which is stronger
If tropospheric ozone is released at the same time as sulphur dioxide it can cause its oxidation into sulphur trioxide by adding an oxygen atom and increasing the concentration of sulphuric acid produced which decreases the pH of the rain further
Nitrous oxides- released from combustion of vehicle engines as the high temps in the engine cause nitrogen and oxygen to bind, when the N0x dissolve in water they can form nitrous acid and nitric acids
Hydrogen chloride- released from the combustion of fossil fuels or the in incineration of some plastic wastes containing chlorine, when they mix hydrogen chloride reacts with water vapour it forms hydrochloride acid which has a very low pH
Environmental impacts of acid rain
Direct impacts
-Toxic to plants- damage enzymes in root hair cells or leaves, causes stunted growth and can damage bark making them vulnerable to insects
-Inhibits seed germination
-Lichens are particularly sensitive to acidic pHS
-asthma and other respiratory illness
-prevents formation of calcium carbonate exoskeletons in marine species such as coral
Corrosion and damage to buildings
Indirect impacts
Acids break up ped structure, reducing soil permeability to water and aeration, may cause death of soil biota and plants
Acidic pHs increase solubility of heavy metals making them more mobile so they can poison organisms
Decomposers killed by acid rain can lead to reduced soil fertility, killing plants
Loss of food soecies due to acid rain will cause population to starve
Loss of predator may cause a prey population to increase
Increased solubility of inorganic ions so more risk f eutrophication which can kill aerobic species
Direct impacts
Contact has been made and the organism will die or become injured/ill as a result
Indirect impacts
Effects that occur to organisms that have not come into direct contact with the pollutant, eg loss of inter species relationships like pollinators or a food source
Properties of acid rain
State of matter: liquid
Mobility- high
Water soluble
Toxic
Synergistic
Specificity- low
Density- low
Secondary pollutant
Environemtal factors increasing the impact of acid rain
Soil lime content- having a high concentration of calcium carbonate (lime) in the soil will neutralise the acidic pH reducing the severity of the impact
Fog- acidic fog remains in the environment for longed and remain in contact with the plant leaf for a longer period of time increasing severity
Snow- if the pH of the snow is acidic then acidic meltwater will runoff into water bodies in a short period of time increasing the severity
Methods to reduce acid rain
it’s a secondary pollutant so need to reduce the primary pollutant
Tropospheric ozone and nitrous oxides are both released from internal combustion in vehicle engines so by reducing our use of petrol and diesel cars and switching to electric or hydrogen we can reduce the production of acid rain and
Sulphur dioxide and hydrogen chloride are both released from the combustion of fossil fuels so by reducing our reliance on using them for energy and switching to renewables we can also reduce their release
Specific techniques for controlling primary pollutants that form acid rain
Desulphurisation- oxides of sulphur can be removed before a fossil fuel is burned, or removed from the waste gases produced during combustion in flue-gas desulphurisation
These techniques vary for each fossil fuel so the import thing to remember is that it’s the removal of sulphur before the fuel is burned eg crude oil can undergo desulphurisation as part of the fractional distillationp
Flue-gas desulphurisation
The removal of sulphur after a fuel has been burned
Dry flue gas desulphurisation- where the waste gases are passed through bed of crushed limestone (calcium carbonate) which reacts with sulphur dioxide to produce calcium sulphate and carbon dioxide
This is a neutralisation reaction because the calcium sulphate can be used to manufacture building plaster that can be used in construction
Wet glue gas desulphurisation- a type of scrubbing which is where the water containing sodium sulphite is sprinkled over the rising effluent gases
The oxides of sulphur react with sodium sulphite in the water to produce sodium hydrogen sulphite removing oxides of sulphur from the flue-gas and dissolving it into the water which could be used to scrub more waste gases
What does the primary pollutant sulphur dioxide form when mixed with water
Sulphurous acid
What does the primary pollutant sulphur trioxide form when mixed with water
Sulphuric acid
What does the primary pollutant nitrous oxide form when mixed with water
Nitric acid
What does the primary pollutant hydrogen chloride form when mixed with water
Hydrochloric acid
What does the primary pollutant tropospheric ozone form when mixed with water
Oxidises sulphur dioxide into trioxide

What control method is this?
An electrostatic precipitator
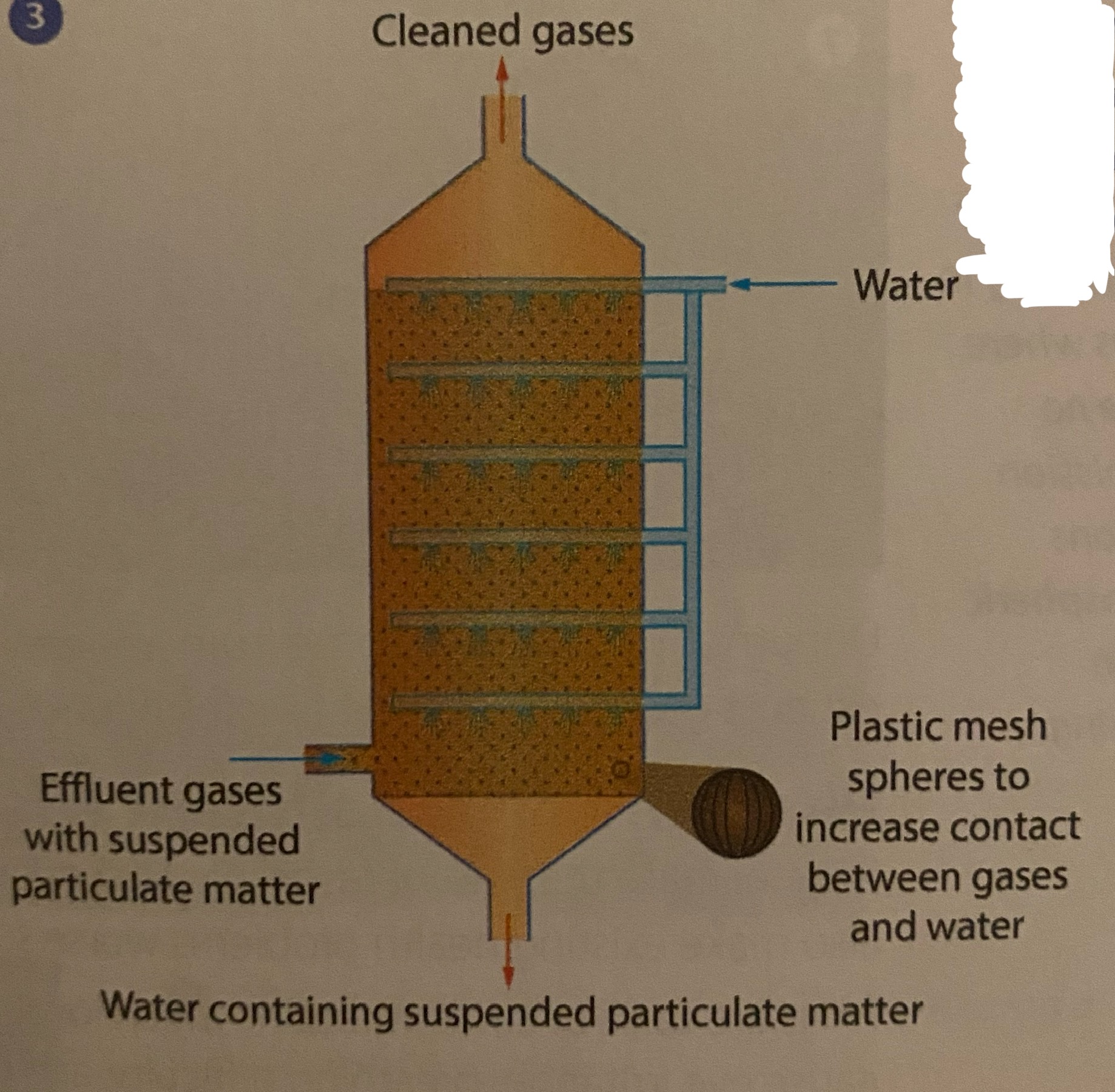
What control method is this?
A scrubber

What control method is this?
A cyclone separator

What control method is this?
Catylictic converter
2 types of aquatic pollution
Thermal pollution, oil pollution
Features of water bodies that will affect pollution severity
Depth of water- deep water more able to dilute pollutant reducing concentration
residence time of water- the longer the water kept in one reservoir the more time the pollutant has to build up and concentrate
Distance from coast- pollution deposited closer to the coat line is more likely to be transported to the coast impacting habitats there
kinetic energy of the water- high kinetic energy like fast flowing rivers are able to disperse more pollutant reducing concentration
protected areas/land use- if released into a protected area like a MPA or MCZ then more likely a vulnerable species will be effected, if released into fishing grounds it will impact populations of food species and availability
How can pollutants in water break down
Heat- thermal degradation
Light- photo degradation
Oxygen- oxidation
Bacteria- species are able to decompose or break down the pollutants -biodegradation
Properties of oil pollution in water
Persistent
Toxic
Unreactive
Insoluble in water so it floats on the surface
Low density
State of matter: liquid
High mobility in choppy/current driven waters
Carcinogenic
Sources of oil pollution in water
Oil exploration methods eg drilling- large drills need lubrication to reduce the amount of friction which would cause energy losses, this is oil based, and when drilling under the seabed it can be released. During extraction accidental release can happen like pipeline leaks oil tanker spills or oil rig accidents
Vehicle engines/ industrial machinery- the waste lubricating oil can be accidentally released or deliberately discarded into water bodies as a waste product
Environmental Impacts/effects of oil pollution in water
The severity of the impact will depend on the location eg if it was close to the coast then more likely the oil will be washed onto land
Chemicals in crude oils are toxic to some organisms -direct
Oil can smother crustaceans and molluscs on the coastlines asphyxiating them so they can’t obtain oxygen or feed -direct
Filter feeders may become poisoned or unable to eat if the oil is filtered through their digestive systems causing blockages -indirect
Reduces insulating stability of bird feathers by clumping them together causing hypothermia and death -direct
Birds spend time trying to clean off the oil so they can’t obtain fly meaning they don’t have time to eat -direct
Potent smells of chemicals in the crude oil can reduce some organisms ability to scent food and mates -direct
When released into water it’s low density means it floats on the surface blocking sunlight preventing photosynthesis as well as oxygen in the atmosphere from dissolving into the water lowering the dissolved oxygen levels -direct
4 categories of control methods for oil pollution in water
Safe disposal of waste oil
Improving oil tanker operation
Improved tanker design
Oil spills clean up
Methods of controlling oil pollution (safe disposal)
Lubricating oil from engines and machinery can be recycled by performing a range of treatment processes to remove contaminants for reuse, this reuse would be found in the circular economy model ensuring less waste oil is disposed of incorrectly snd reducing need for new oil,
this waste oil can also be combusted as a source of heat energy reducing reliance on other sources like coal yet GHG, sulphur dioxide and carbon monoxide are released from the combustion
Methods of controlling oil pollution (Improving oil tanker operations)
Operations designed to reduce spillages:
open shipping routes in the open ocean avoiding vulnerable coastlines
Navigation systems that can track possible collision risks
Inert (unreactive) gases pumped into the oil tank to prevent oxygen entering and causing an explosion
Extracting and transporting viscous sludge that’s also removed from the reservoir rock so when the destination is reached it will have sunk to the bottom and not come out with the oil
Instead of using water to clean out the sludge tankers can now recirculate the oil through the tank and use it to mobilise the sludge so it can be removed
Methods of controlling oil pollution (Improving oil tanker designs)
Tanker designs improved to minimise risk of spillages if disasters happens:
Installing twin engines and fuel tanks- so there’s a backup available for each if one has a problem, without this the ship wouldn’t be able to reach its destination and is more likely to cause collisions
Bund wall- fitted around the oil tank providing an extra layer of protection to prevent the oil from being released even if the internal tank is broken, materials used include concrete or steel
Double layered hull- the hull is the main body of the boat, when there is a collision the outer layer will be greatly impacted but the inner layer should remain more intact
Ballast water- needed for a boats buoyancy, when transporting oil acts as this but on the way back ocean water is used so when the ship arrives at its destination oily ballast water would be released into the ocean causing pollution, to combat this new tankers have separate oil and ballast tanks

Treating oily wastewater
Treating oily wastewater before being released- using an oil interceptor which relies on the hydrophobic nature of oil (doesnt dissolve in water) to separate from the water and settle on the surface where it can then be collected and clean water then leaves the machine
Methods of controlling oil pollution (cleaning up oil pollution)
Booms- long inflatable tube that floats on the surface with a net skirt underneath, this helps reduce dispersion ensuring the oil remains in one place, doesnt activity remove the oil
D- can’t be used in areas with choppy water or strong currents as it would move around too much allowing oil to escape,
Skimmers- used once a boom has trapped the oil spill consisting of large discs that rotate across the ocean surface skimming it to remove the oil sticking to the discs, these are then rinsed to collect oil and dispose of it
d- can only be used in at calm waters as they need a flat surface to be able to pick up the oil
Polymers- long chain molecules that will absorb the oil from the ocean, these designed to have as large surface area as possible so they can absorb most oil, once material is saturated then it can be collected and separated for disposal, also works best in calmer waters as waves could end up washing oil from the surface of the material
Detergents/dispersants- added to the ocean to emulsify (break up) the oil into droplets instead of one large body, chemicals are then more mobile so it will disperse more readily throughout the ocean diluting the concentration A- can be used in choppy waters where successful clean up isn’t possible
Polymerising agents- opposite to dispersants as they cause the oil to clump together and form a solid polymer, this could make it easier to collect yet skimmers snd absorbing materials then won’t work
Steam washing- hot water/steam at high pressure used to wash oil off of beaches and rocks on the coastline D- can sometimes cause harm to the area as the hot water can force the oil deeper into the sand/sediment and erode the beach
Bioremediation - using bacterial species to hydrolyse (break up) the hydrocarbons using enzymes -slow process
Case study of oil pollution
Deep water horizon oil spill
This was the largest oil spill in the history of the petroleum industry, a massive response was issued including skimmer ships, floating booms, and controlled burns.
Oil flowed for 87 days that affected 180,000km of ocean and washed up on the coastline and had mixed with deep ocean sediments risking damage to ecosystems and commercial fisheries.
Health risks with humans from the cleanup- eye nose and throat irritation, respiratory issues, seizures, violent vomiting, short term memory loss
Health risks to marine life- oil contained methane that can suffocate marine life and create dead zones, infant dolphins died at 6x the normal amount, mutated fish found and shrimp without eyes and eye sockets
What is thermal water pollution?
The influx of heat energy into a water body usually in the form of hot effluent (waste) water from industrial processes such as manufacturing or energy generation
Properties of thermal water pollution
Low persistence
Energy form: heat
Low mobility as low persistence
Toxic: denatures enzymes
Reactive: causes chemical reactions to happen faster
Effects of thermal water pollution on oxygen
Decreased solubility of oxygen: leads to a decrease in dissolved oxygen levels of the water, this is an inverse relationship meaning the warmer the temperatures, the less dissolved oxygen
With lower concentrations aerobic species are going to be at risk of dying as they need oxygen for respiration
Some species can survive in a range of oxygen concentrations and therefore won’t be as severely impacted yet some require oxygen at specific levels and if it drops then they will be out of their range of tolerance
Effects/impact of thermal water pollution
Higher temps causing enzymes denaturing in aquatic organisms
Faster growth rates due to increased metabolic rate from higher temperatures
Eggs developing faster and hatching earlier leading to increased risk of fatality
Lower disease resistance from heat stress
Faster chemical reactions increasing pollutant toxicity
Introduced species may become invasive if they can survive in warmer conditions, if they’re a competitor/predator of native species that capsule cause population decline
Control of thermal pollution
This involves removing heat energy from wastewater
A cooling tower- hot effluent water is sprayed into tower from sprinklers near top, cool air fed into bottom absorbing heat from effluent and rising, warm air then leaves cooling tower through hole at the top and cooled effluent released into environment not causing thermal pollution, A- the air system saturates the effluent with oxygen ensuring it won’t harm aerobic species when released into the environment
Within this the water and air are fed in opposite directions known as a counter current exchange mechanism , allowing max heat transfer into the air by maintaining a gradient between the two

What control method is this?
Water cooling tower
Properties of pesticides
Different types of pesticides have varying properties
Properties that benefit farmers:
High persistence- don’t need to reapply all the time
Low specificity- target wide range of pets with less use
Lipid soluble- absorbed into pest tissue
Properties benefiting the environment
Low persistence- organisms are not exposed for a long time
High specificity- fewer non target soecies affected eg pollinators
Both
Low water solubility- cant disperse and leach from soil
all pesticides are toxic which is why they’re used to kill pest organisms, the space can impact the toxic it level
If pesticides are persistent they are more likely to bioaccumulate and biomagnify
Different dosages that can be applied for pesticides
MDAF- minimum dose always fatal, the lowest dose of the pesticide that we use that will kill all members of a population
MDNF- maximum dose never fatal- the highest dose of a pesticide that will not kill any members of a population
Most effective pesticides are able to be applied to reach MDAF for the target species to ensure they’re all killed without exceeding MDNF for non target species
Eg. A farmer wants to kill aphids but not bees, aphid MDAF- 20mg, Aphid MDNF- 1mg, the Bee MDAF- 55mg and the bee MDNF- 22mg so the farmer can apply 20mg to kill aphids but not bees
Indirect impacts of pesticides
Pesticides may kill a food species leading to death of the predator due to lack of food
Pesticide may kill a pollinator leading to reduced populations of plant species
how pesticides/insecticides kill pests
They damage the nervous system by blocking enzymes which reduce their ability to fly and move, they become paralysed and die
4 different types of pesticides
Organochlorines
Organophosphates
Pyrethoids
Neonicotinoids
Two types of application methods for pesticides
Systemic pesticides- absorbed into the crop roots so are within crop cells
Contact- sprayed onto exterior of crops
Neonicotinoids (pesticides)
Benefits to farmer- high insect toxicity, persistent, potent at low dosages, lipid soluble
Negatives to environments- low specificity neurotoxin, water soluble so can leach from soil, very toxic to bees, synergistic, biomagnification,
Advantages to environment- low vertebrate toxicity
Organophosphates (pesticides)
Benefits to farmer- high insect toxicity
Negatives to environment- neurotoxic (poisonous/destructive to nerve tissue), high mammalian toxicity so low specificity, carcinogen
Advantages to environments- low persistence, not lipid soluble so do not bioaccumulate or biomagnify
Organochlorines (pesticides) eg DDT
Benefits to farmer- high insect toxicity, persistent
Negatives to environment- lipid soluble so can bioaccumulate and biomagnify, low specificity as killed bees and butterfly species, caused bird egg shell thinning
Advantages to environment- low vertebrate toxicity, low water solubility so low mobility
Pyrethroids (pesticides)
This an example of biomimetics as mimic natural insecticide produced by some plants
Benefits to farmer- high insect toxicity
Negatives to environments- toxic to fish
Advantages to environment- low mammal toxicity, low persistence, not lipid soluble so cannot bioaccumulate or biomagnify, insoluble in water so low mobility, not carcinogenic
Control methods to reduce impacts of pesticides
Ban/restrict use of most environmentally damaging pesticides eg DDT which is an organochlorine was banned after it was found to be causing huge declines in bird populations
Replacement pesticides- can be manufactured that have properties that are less harmful to the environment such as less persistence and more specific
Systemic pesticides- used instead of contact as systemic are less likely to disperse and affect non target organisms as they remain within the plant tissue
Timing of application- eg not applying pesticides just before heavy rainfall as they may end up leaching into nearby water bodies if they are water soluble,
Buffer strips- around fields that are sprayed which act as a buffer to prevent chemicals leaching into surrounding water bodies
Using cultural pest methods-crop rotation, barrier crops instead of using pesticides
Difference between inorganic and organic pollutants
Organic matter- materials that contain carbons eg glucose and sewage
Inorganic matter- doesn’t contain carbon eg nitrates, ammonium and phosphates
Properties of inorganic nutrients as a pollutant
Highly water soluble
Some forms of heavy metal are lipid soluble
If lipid soluble they can bioaccumulate and biomagnify
High mobility
Toxic
Synergistic
Carcinogenic
Impacts of inorganic fertilisers
Examples: nitrates and phosphates
are extremely soluble in water so if it rains they can dissolve and leach from the soil, this causes them to runoff into a waterbody where they trigger the process of eutrophication
Sewage effluent can contain large volumes of phosphates which can also cause eutrophication if released into water bodies
Nitrates in fertilisers can be toxic if injected within drinking water, they get converted into nitrites in our gut that react with haemoglobin in the blood to reduce its ability to carry oxygen
Oligotrophic lakes
Low concentration of nutrients, small plant diversity - young lakes
Eutrophic lakes
High concentration of nutrients, high plant diversity -older lakes

Eutrophication (inorganic pollutants)
Likelihood of eutrophication is even greater in areas designed as nitrate vulnerable zones (NVZ) which are categorised as being extremely vulnerable to the use of nitrogen fertilisers
Eutrophication causes blocking of light so submerged plants cant photosynthesise, the dead plants are decomposed by aerobic bacteria which remove oxygen from the water in the process, deoxygenation in the water causes aerobic species to die like fish this is cultural eutrophication
Organic material doesn’t directly cause eutrophication, it happens because of inorganic material inputs