Bond enthalpies (3.1.4.4)
1/3
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
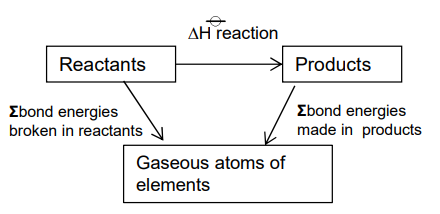
What is mean bond enthalpy?
Mean bond enthalpy - the enthalpy needed to break one mole of a particular covalent bond in a gaseous molecule.
The mean is used as each bond has a slightly different bond energy in a molecule.
Mean bond enthalpies are positive as bond breaking is endothermic.
How can mean bond enthalpies be used to calculate an approximate value of ∆H for reactions in the gaseous phase?
Or more generally:
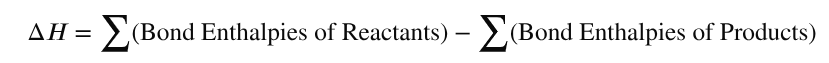
Why do values from mean bond enthalpy calculations differ from those determined using Hess’s law?
Values calculated using mean bond enthalpies often differ from those determined using Hess's law for two main reasons:
1) Mean bond enthalpies are averages taken from a wide variety of molecules. In a specific reaction, the actual energy needed to break a bond may be slightly different because the molecular environment affects the bond strength.
2) ΔH⦵ applies to substances under standard conditions in their standard states, whereas calculations using mean bond enthalpies assume all reactants and products are in a gaseous state
1
1