Chemistry - Solids, Liquids, and Mixtures
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
vaporization
phase transition from a liquid or a solid state into a gas or vapor
viscosity
resistance to being poured
volatile
substance’s tendency to vaporize or turn into a gas or vapor at normal temperatures and pressures
deposition
phase transition → gas into a solid
cohesion
attraction of molecules to the same substance
adhesion
ability of liquids to stick to a surface
capillary action
flow of a liquid through narrow spaces, driven by surface tension and interaction between adhesive and cohesive forces (ex: liquid through paper towels/thin tubes)
surface tension
when liquids pull their particles close together to form a barrier
allotrope
different structural forms of the same element in the same physical state
characteristics of liquids
particles are relatively close, inter molecular forces are present, non-compressable, definite volume; no shape
fluidity
the ability for liquids to take the shape of their containers, flow easily, and form at minimal effortand can move past one another.
emulsion
substances that don’t mix well
emulsifier
agent added to mixtures that will dissolve the suspended particlesand stabilize the emulsion, preventing separation.
sublimation
transition from a solid to a gas
evaporation
liquid turns to gas below boiling point
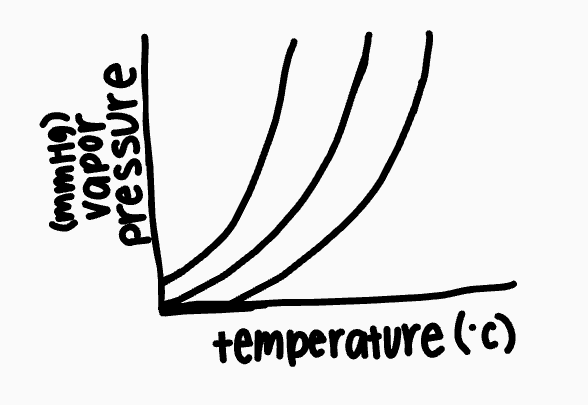
What is this graph called?
vapor pressure curve
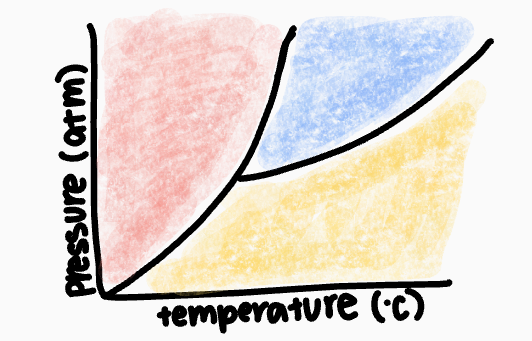
What is this graph called?
phase diagram
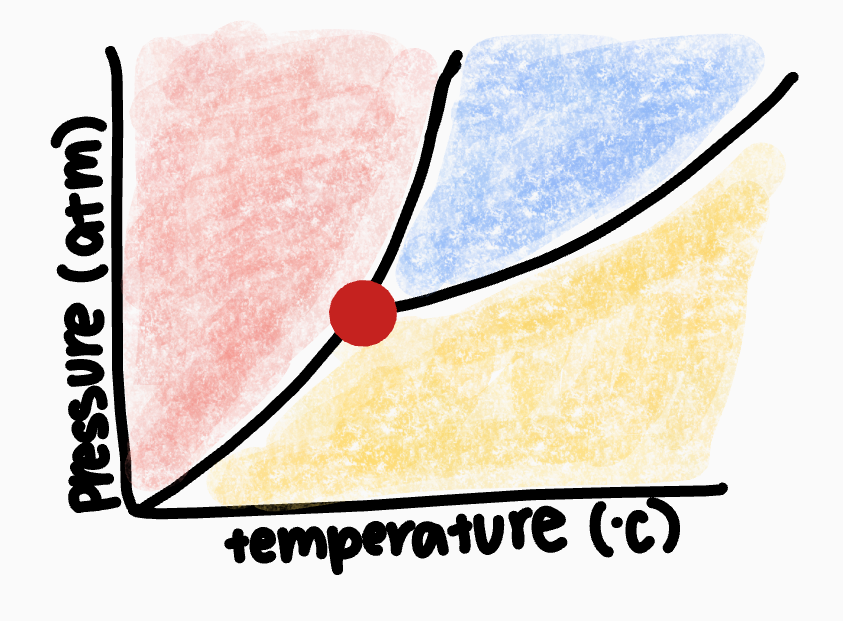
What point is the red dot on?
triple point
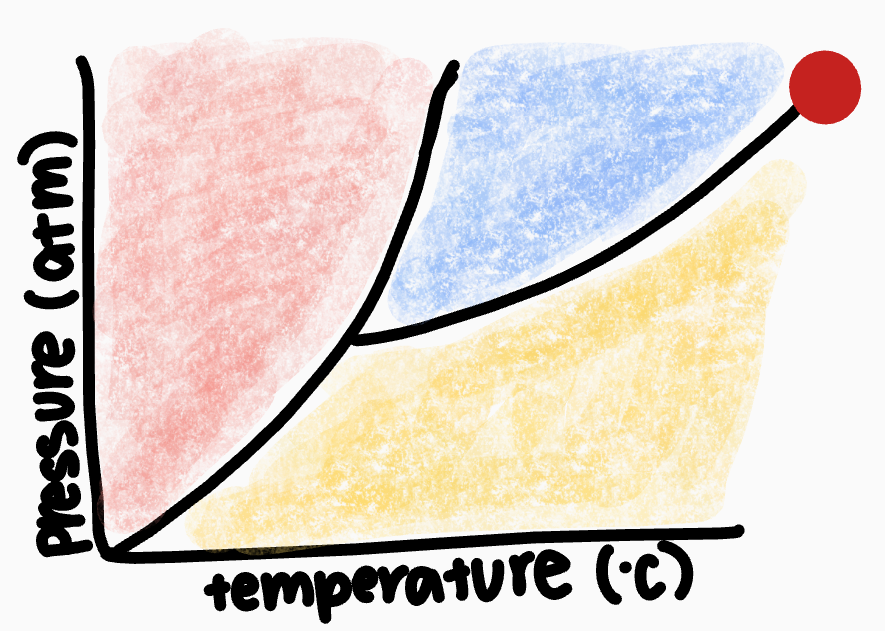
What point is the red dot on?
critical point
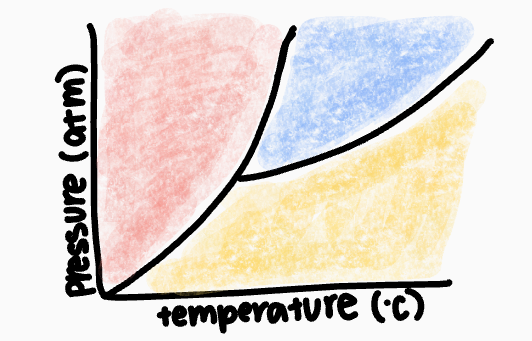
What phase occurs in the red zone?
solid phase
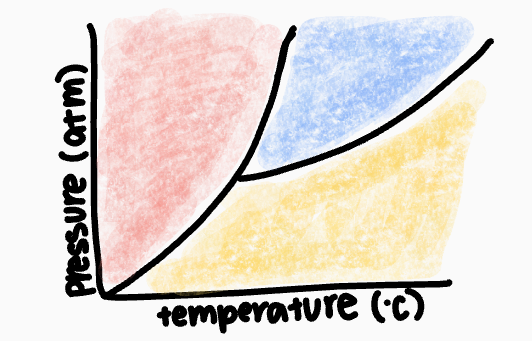
What phase occurs in the blue zone?
liquid phase
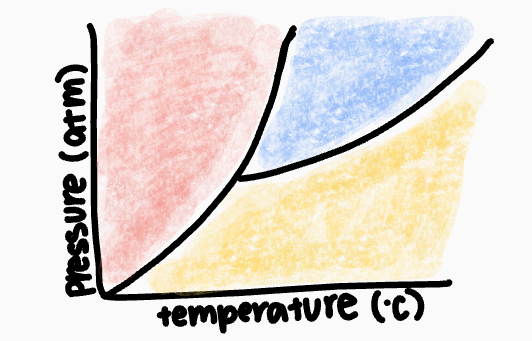
What phase occurs in the yellow zone?
gas phase
characteristics of solids
definite volume, definite shape, particles are packed closely, non compressible, high density
What are crystalline solids? (crystals)
solids where particles are arranged in geometric patterns
What are the five categories of crystalline solids?
atomic, molecular, covalent, ionic, metallic
What are examples of covalent crystals?
diamonds, graphite, Buckminster fullerance
unit cell
basic geometric pattern
crystal lattice
bunch of unit cells
What are examples of ionic crystals?
table salt, lithium fluoride, magnesium oxide
What are examples of metallic crystals?
gold, aluminum, silver
What are amorphous solids?
anything not a crystal
What is the sea of electrons?
electrons surrounding the nucleus, but are not connected to
Examples of amorphous solids
desk, floor, dirt
malleable
put into sheets
ductile
drawn into wires
Characteristics of heterogeneous mixtures
not well blended, not same throughout
Examples of heterogeneous mixtures
vegetable soup, tossed salad, mixed nuts
Characteristics of homogeneous mixtures
well blended, same throughout
What is another name for something being the same throughout?
solution
colloid
group of mixtures
solvent
substance that does the dissolving
solute
substance that is dissolved
What is the universal solvent?
water
What is an aqueous solution?
water solvent mixture
What is a miscible mix?
liquid-liquid mix
immiscible
two liquids that don’t mix
insoluable
solute does not dissolve well
Tyndall Effect
shine light into colloid → light scatters
Examples of colloids
smoke, cream cheese, whipped cream
percent by mass formula
mass solute/ mass solution x 100
percent by volume formula
volume solute/volume solution x 100
molarity formula
mole solute/ liter of solution
What is Brownian motion?
random zig zag movement of particles in a liquid, caused by constant collisions with surrounding molecules