Module 4: Cell Adhesion and Communication w ECM
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
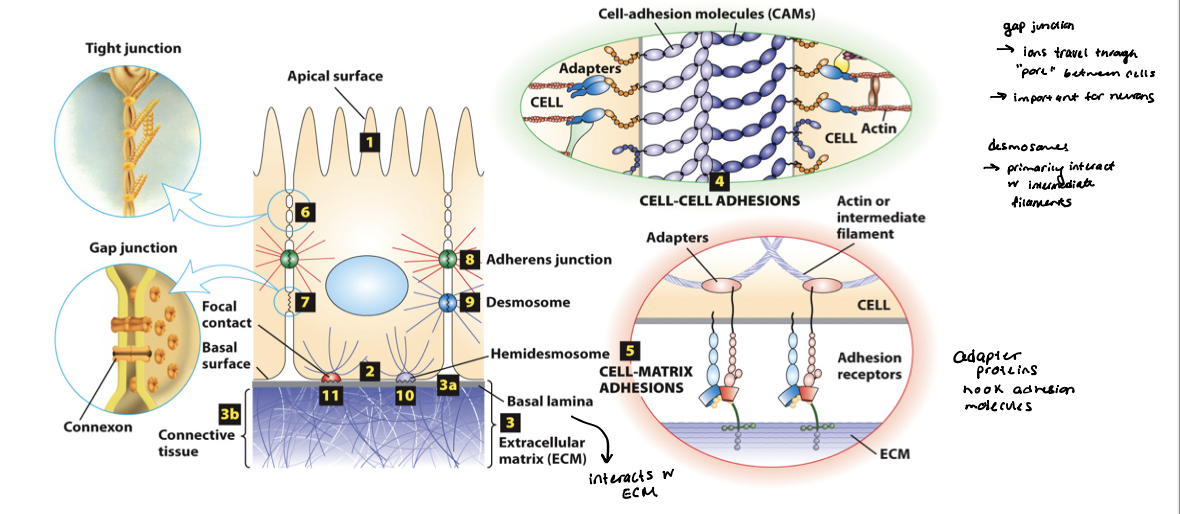
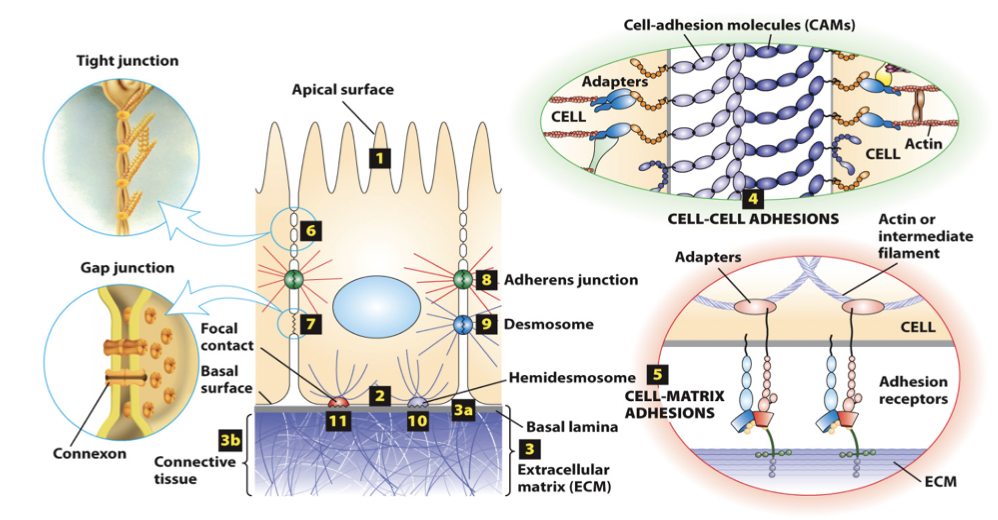
cell-adhesion molecules (CAMs)
mediate direct cell-cell adhesions (homotypic and heterotypic), and adhesion receptors mediate cell-matrix adhesions
primarily integral membrane proteins, which cytosolic domains that bind intracellular adaptor proteins that link the surface with the cytoskeleton (typically actin and intermediate filaments)
extracellular matrix (ECM)
a dynamic, complex meshwork of proteins and polysaccharides that contributes to the structure and function of a tissue
cell-matrix adhesion molecules → adhesion receptors bind to ECM components; link the external environment to the internal cytoskeleton
bidirectional information transfer
outside-in → from CAMs and bound extracellular macromolecules to the cytoplasm
inside-out → from the cytoplasm through adapter proteins to CAMs and bound extracellular macromolecules
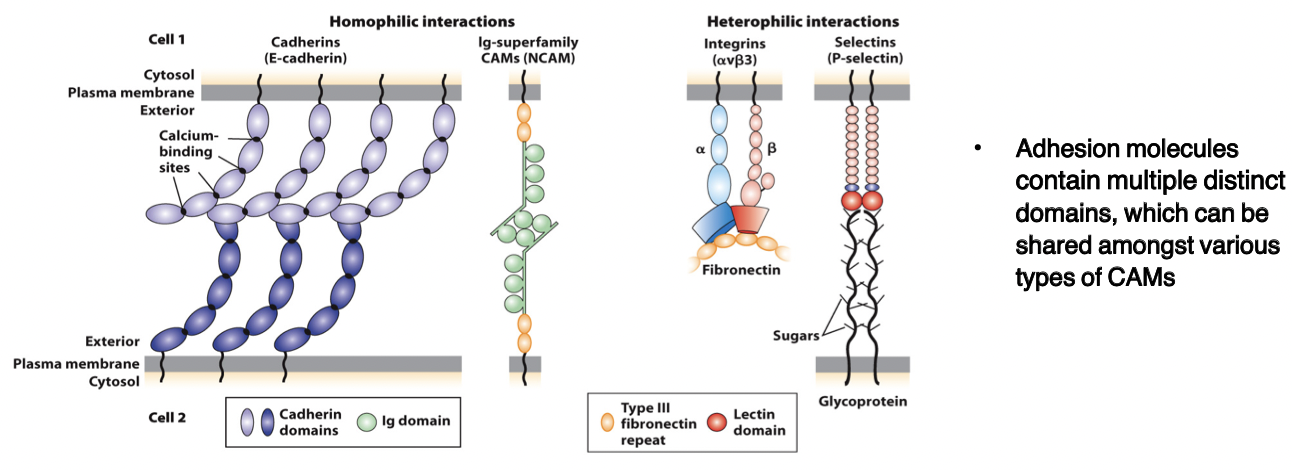
cadherins
bind to each other (homophilic and heterophilic) via domains
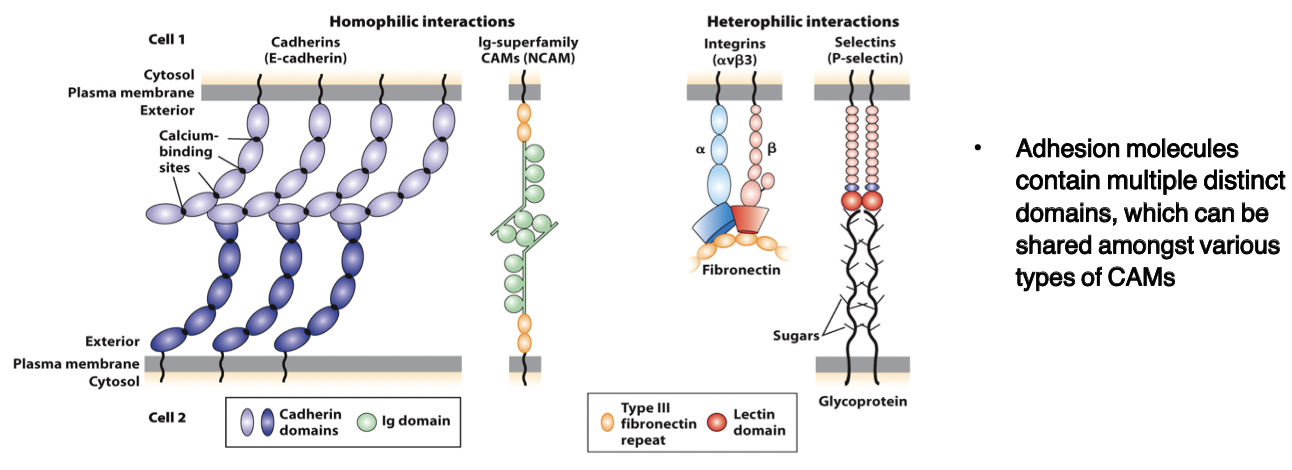
CAMs
members of the immunoglobulin superfamily
form both homophilic and heterophilic interactions
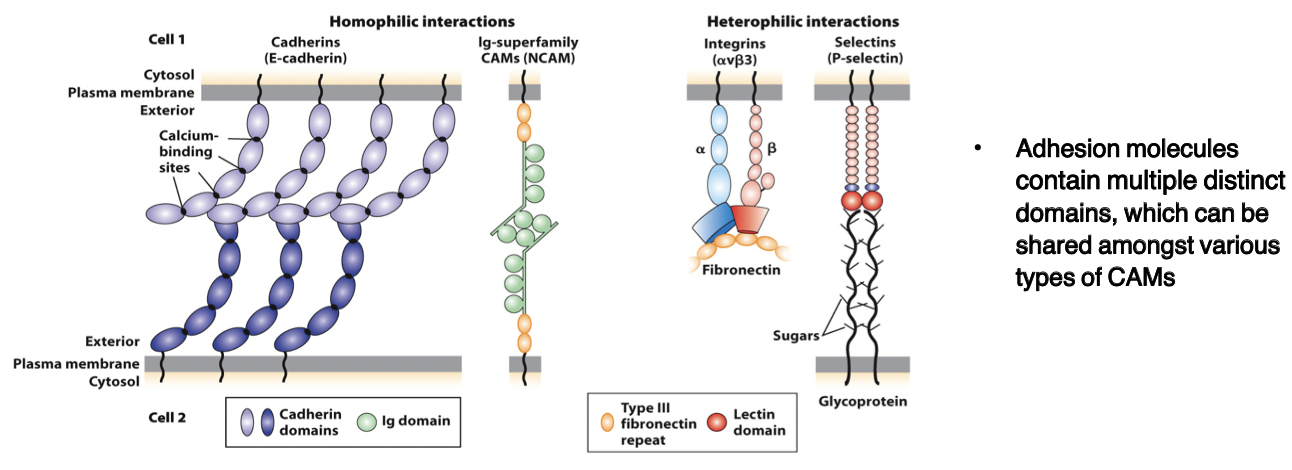
integrins
heterodimeric, consist of α and β chains
adhesion receptors, bind to large adhesive proteins such as fibronectin in the ECM
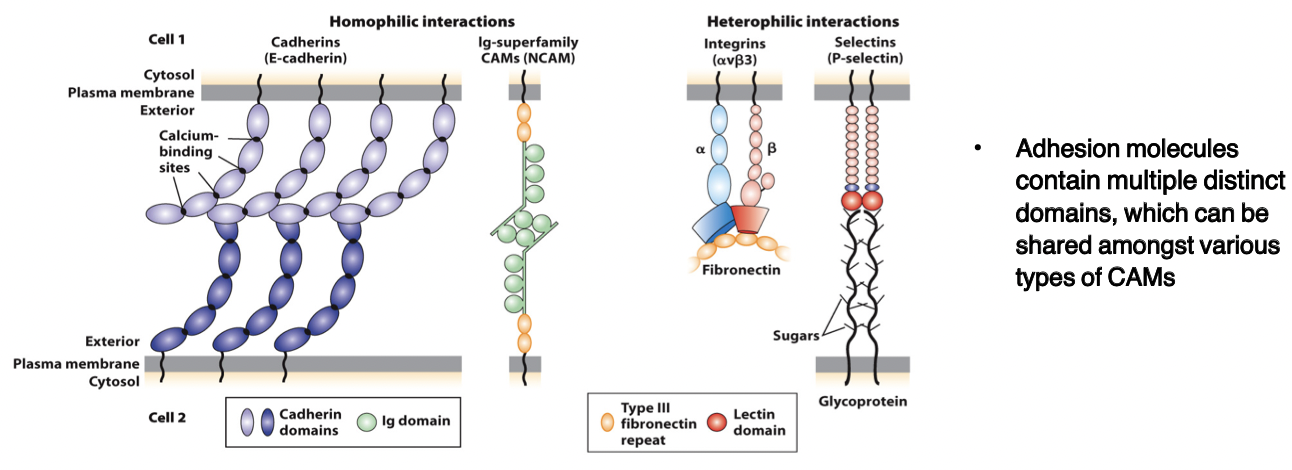
selectins
contain a carbohydrate-binding lectin domain that recognizes specialized sugar structures on adjacent cell glycoproteins/glycolipids
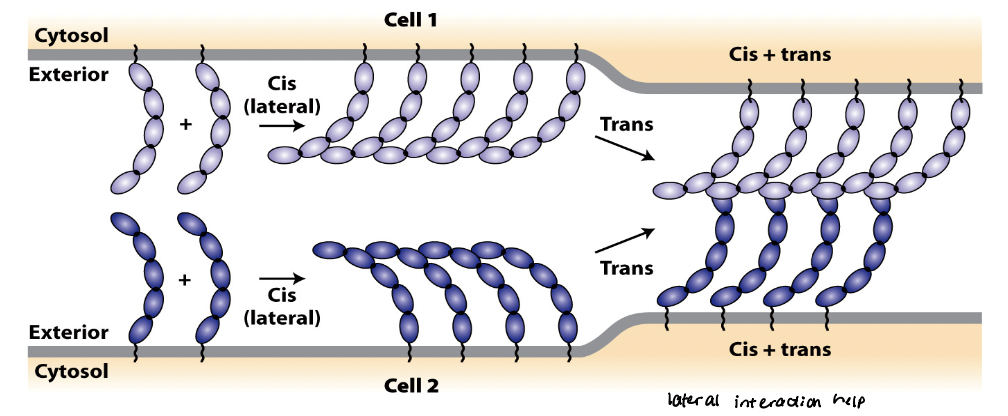
CAM cis interactions
referred to as intracellular or lateral interactions
forms lateral clusters within the plasma membrane of the same cell
regions that form cis interactions vary among different CAMs
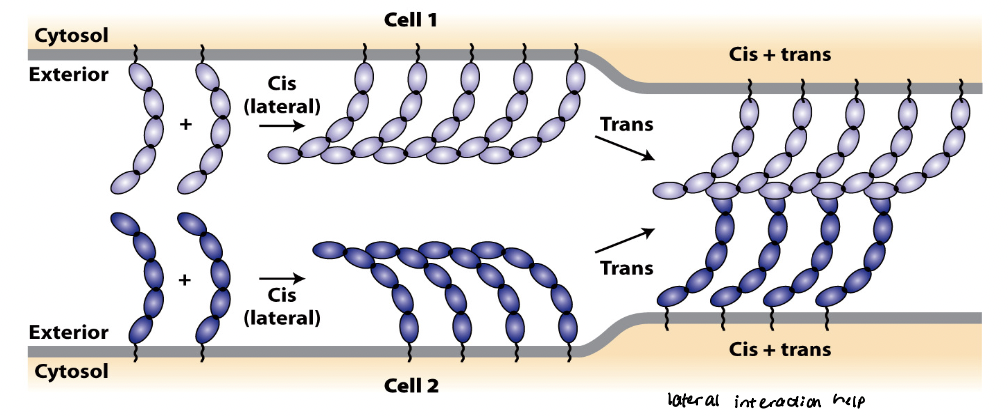
CAM trans interactions
referred to as intracellular or adhesive interactions
generate strong, velcro-like adhesions between neighboring cells
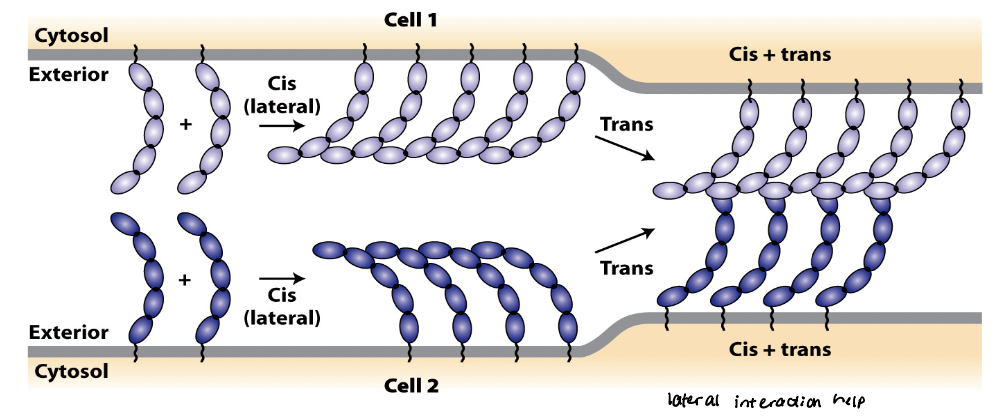
mutually reinforcing
Trans and cis interactions are ______________.
cis interactions can increase probability of forming trans interactions
trans interactions can induce cis interactions, which in turn strengthen trans interactions
factors that regulate adhesive strength
clustering → at cell junctions where CAMs tend to cluster, the CAMs can generate very tight adhesion when many weak interactions are combined
binding affinities
kinetic properties of the CAMs (“on or off” rates) control the association/dissociation properties, and thus the strength of the adhesion
spatial distribution and density of molecules (ensemble properties)
biochemical properties and “active” vs. “inactive” states
external forces such as stretch and pulling
ECM stiffness
varies across tissues
brain → soft ECM
muscle → intermediate ECM
bone → rigid ECM
influences cell behavior
regulates cell shape, migration, proliferation, and differentiation
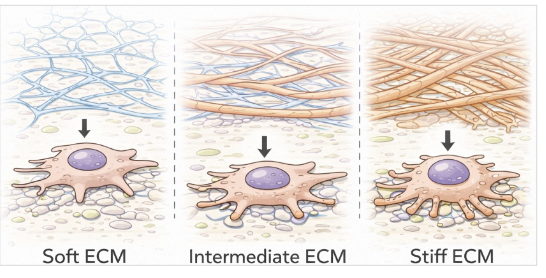
mechanical properties from ECM compositions
collagen provides tensile strength
proteoglycans provide compressive resistance
cross-linking proteins (laminin) provide increased stiffness
true
T/F: Cells can sample and “sense” the ECM through transmembrane adhesion receptors (integrins) which then communicate to the cytoskeleton
instructs cells how to behave within their environment
cell convert mechanical properties of the ECM into biochemical signals
mechanotransduction
alters cell behavior; drives gene expression, migration, and fate decisions
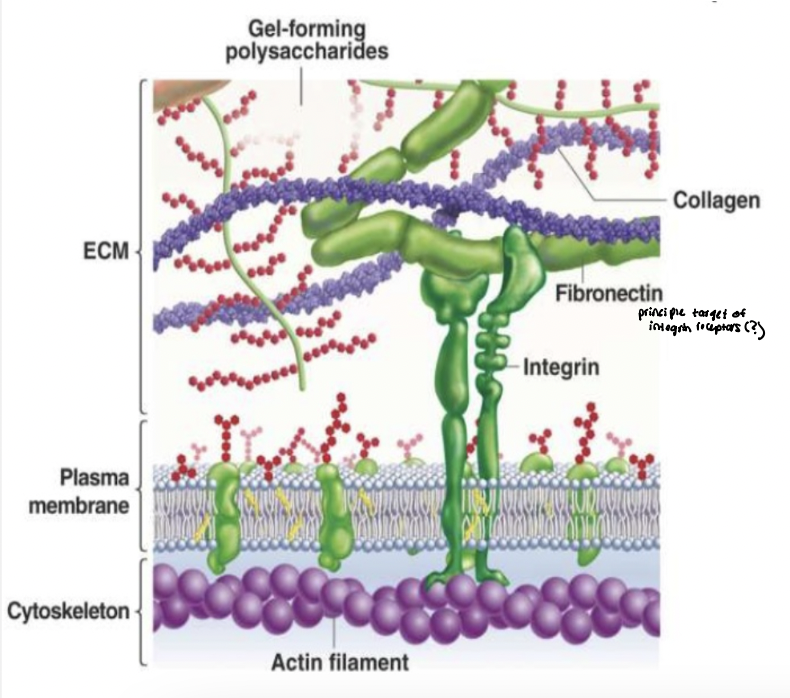
extracellular matrix proteins
proteoglycans → unique type of glycoprotein
collagens → form fibers
multi-adhesive matrix proteins → organizers of the ECM
fibronectin and laminin
long, flexible molecules that contain multiple domains
bind various types of collagen, other matrix proteins, polysaccharides, and extracellular signaling molecules as well as adhesion receptors
interactions w adhesion receptors - regulate cell-matrix adhesion and cell shape and behavior
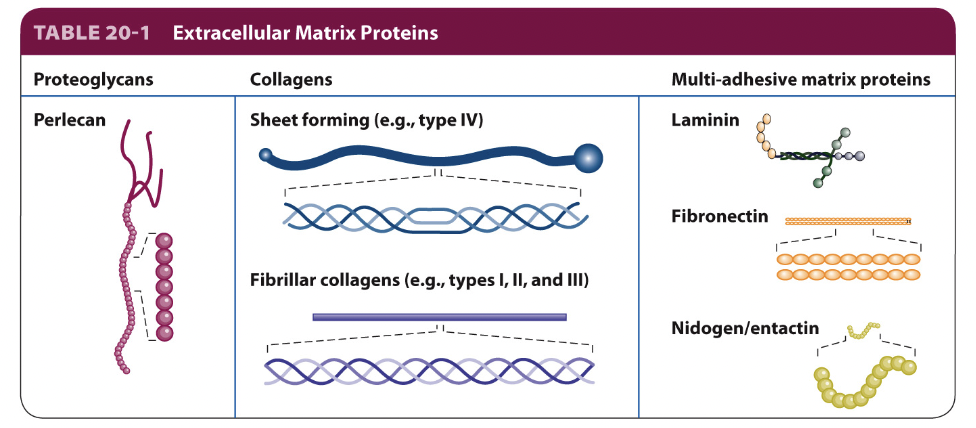
collagen
most abundant ECM proteins; provide the primary structural scaffold in many tissues
form fibrils and fibers to create tensile strength and resistance to stretching
highly organized and cross-linked
enables mechanical stability and durability of tissues
type I → skin, bone, tendon
type II → cartilage
type III → basement membrane (network forming)
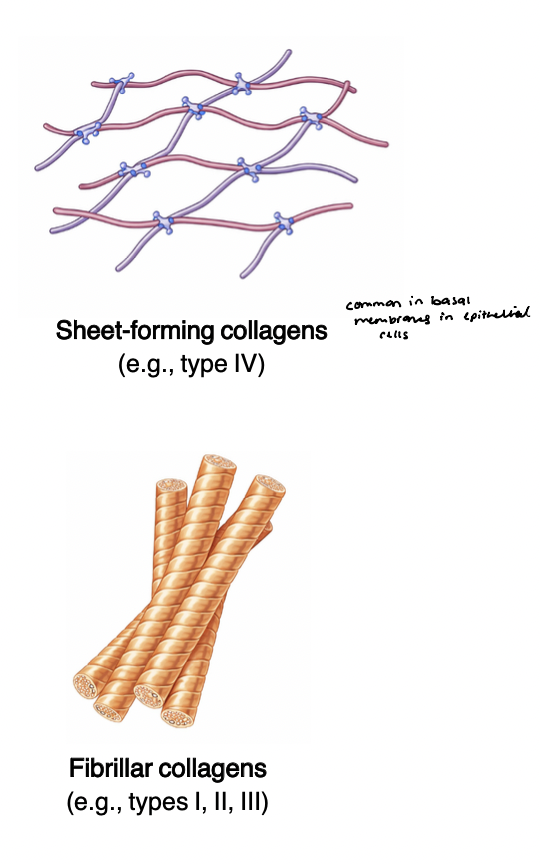
proteoglycans
act as structural “fillers” for the ECM
hydration and cushioning
(-) charged CAG chains, long unbranched sugar molecules that attract water
form hydrated gel-like environments in tissue
provide resistance to compression
act as reservoirs for growth factors and cytokines
regulate diffusion of signaling molecules
protects them from degradation and controlling availability for cell surface receptors
contribute to tissue biomechanics
control viscosity, porosity, and ECM spacing
sensors for mechanical force in tissues (bone, cartilage) in response to physical activity
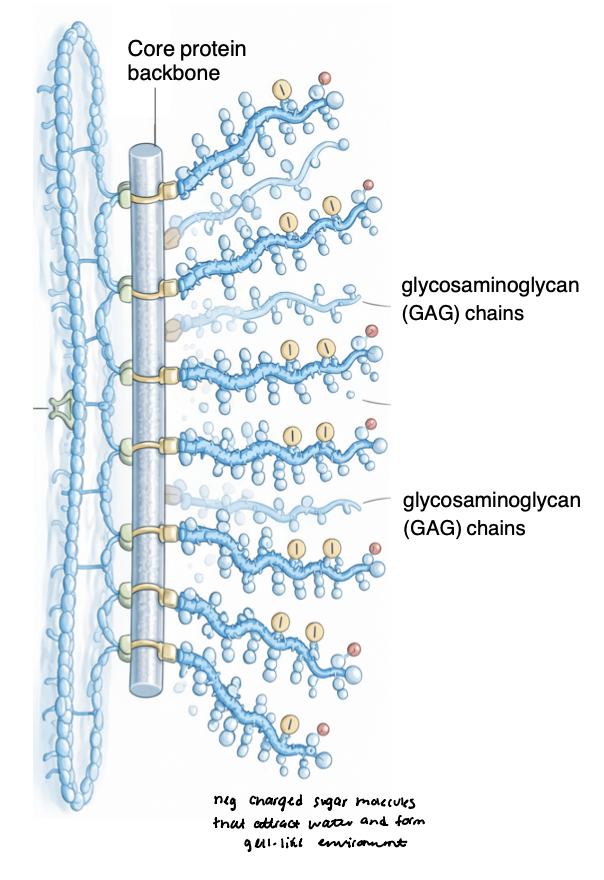
multi-adhesive glycoproteins
links cells to the ECM and organizes matrix structure
bind to collagens, proteoglycans, integrins
modular, multi-domain proteins → enable simultaneous interactions with multiple partners and itself (networks)
examples
fibronectin → connective tissue ECM
laminin → basement membrane
true
T/F: Cells can contribute to the assembly of the ECM by:
secreting its components
directing the assembly of these components into complex, interwoven structures
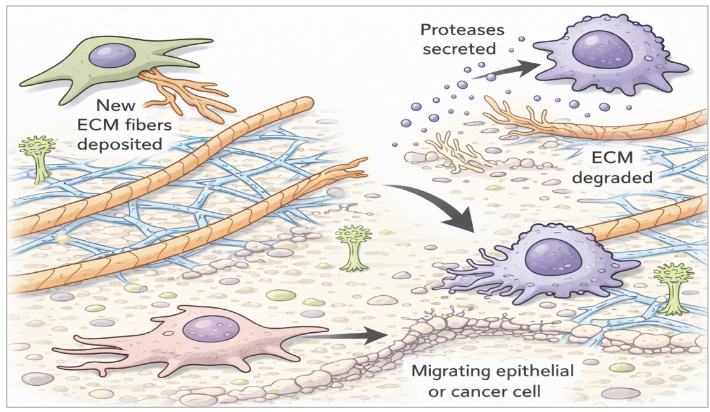
highly dynamic
Once assembled, the ECM is not a static structure, but rather __________
chemical, physical, and biological properties can be altered as cells secrete enzymes and other molecules into ICM → ECM remodeling (cross-linking components, protease cleavage of ECM components)
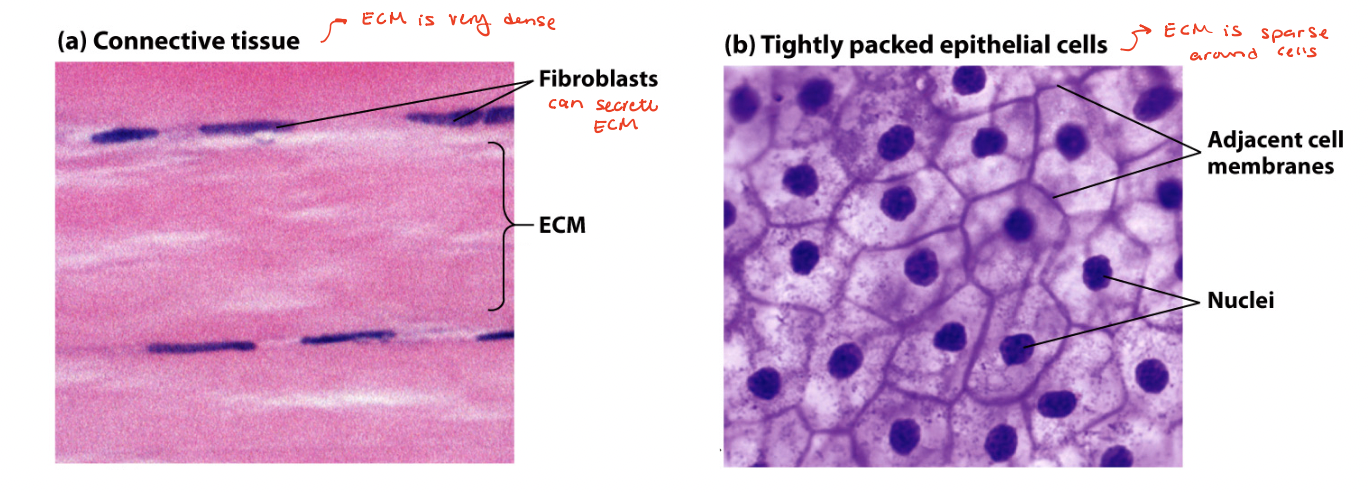
density of cells and ECM
dense connective tissue → contains mostly ECM containing tightly packed ECM fibers interspersed with rows of relatively sparse fibroblasts (cells that synthesized ECM)
sparse connective tissue → squamous epithelial cells tightly packed into a quilt-like pattern with little ECM between the cells
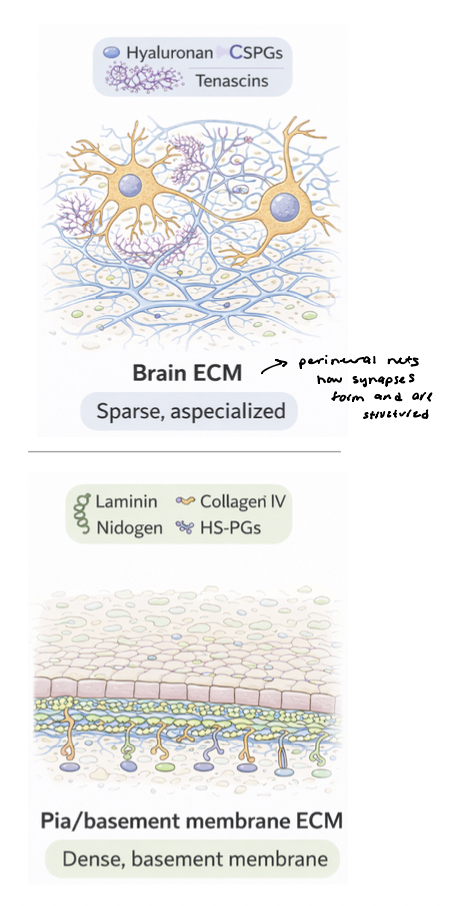
brain ECM
sparse and highly specialized, occupying small extracellular spaces between densely packed neurons and glia
dominated by proteoglycans and glycoproteins (tenascins)
forms specialized structures → perineuronal nets, organize around neurons to stabilize synapses and limit plasticity
pia membrane → contains dense basement membrane-like ECM
rich in laminin, collagen IV, nidogen, and heparan sulfate proteoglycans
provides structural support and barrier function
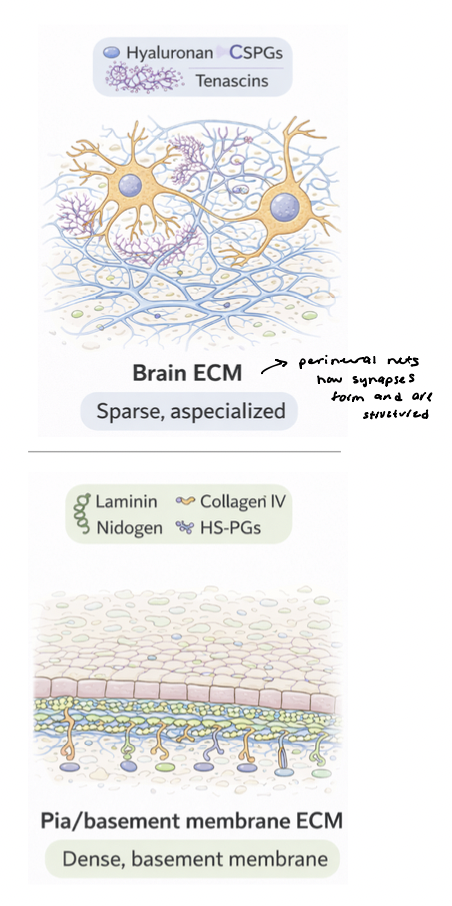
brain ECM vs. pia ECM
brain ECM is soft, permissive, signaling-rich
pia ECM is dense, structured, barrier-forming scaffold
different isoforms
Diversity of cell adhesion molecules arise from _________
different members of a family (integrins) can be encoded by different genes
gene products can be alternatively spliced to produce different protein products
tissue morphogenesis
disruptions in cell-matrix and cell-cell interactions interfere with tissue development
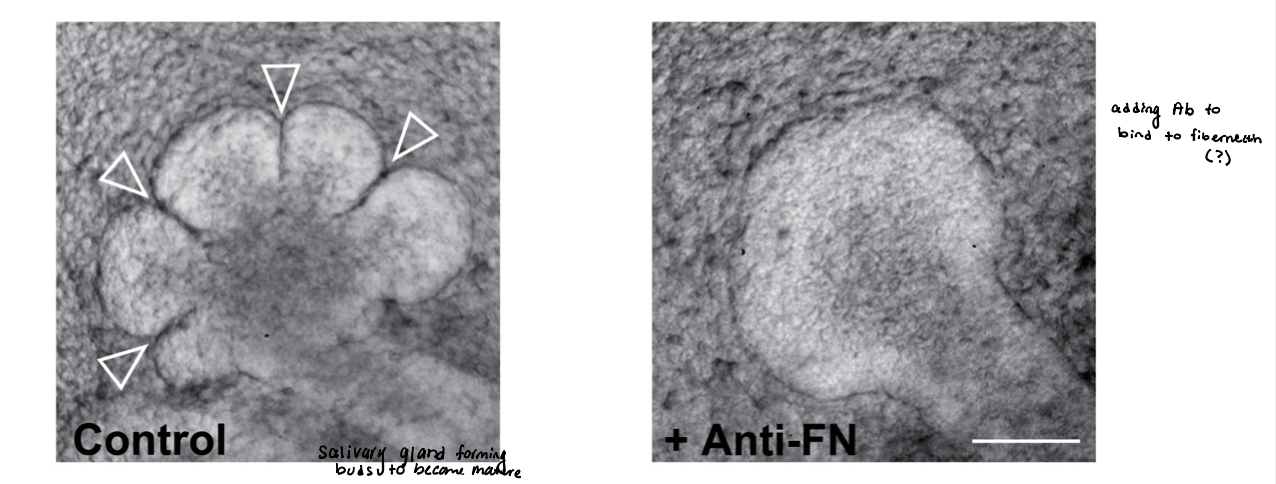
experiment:
immature salivary glands → isolated from murine embryos
undergo branching morphogenesis in vitro for 10 hours
results:
absence of added Ab → normal branching
presence of Ab (anti-FN) → blocks fibronectin activity
conclusion → integrin-fibronectin interaction is required for branch formation
inhibition of integrin fibronectin receptor blocks branch formation
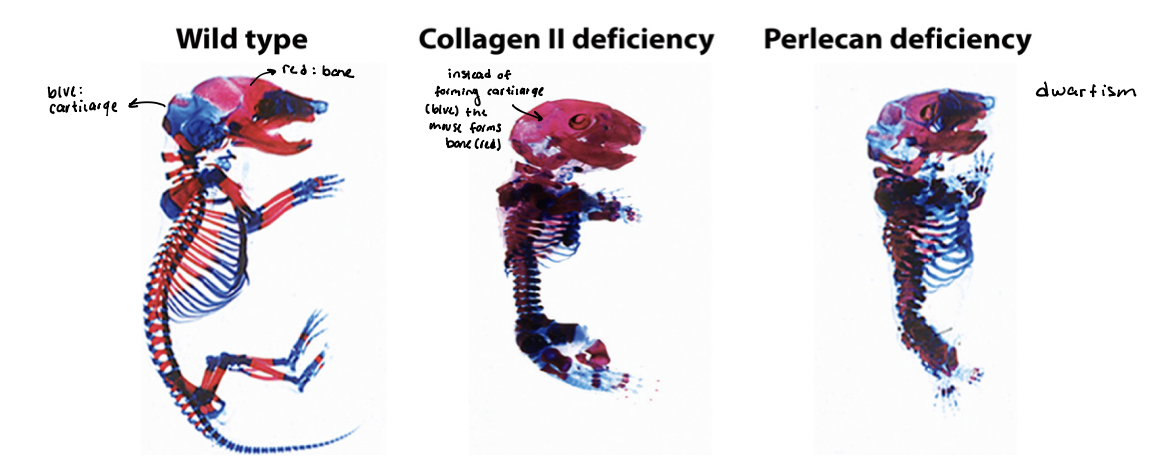
disruptions to adhesion and ECM functions
characteristic of various pathologies
skeletons (mouse) → cartilage (blue) and bone (red)
WT → normal
collagen II → deficient
perlecan → deficient
fibronectin → deficient
absence of key ECM components leads to dwarfism, with many skeletal elements shortened and disfigured
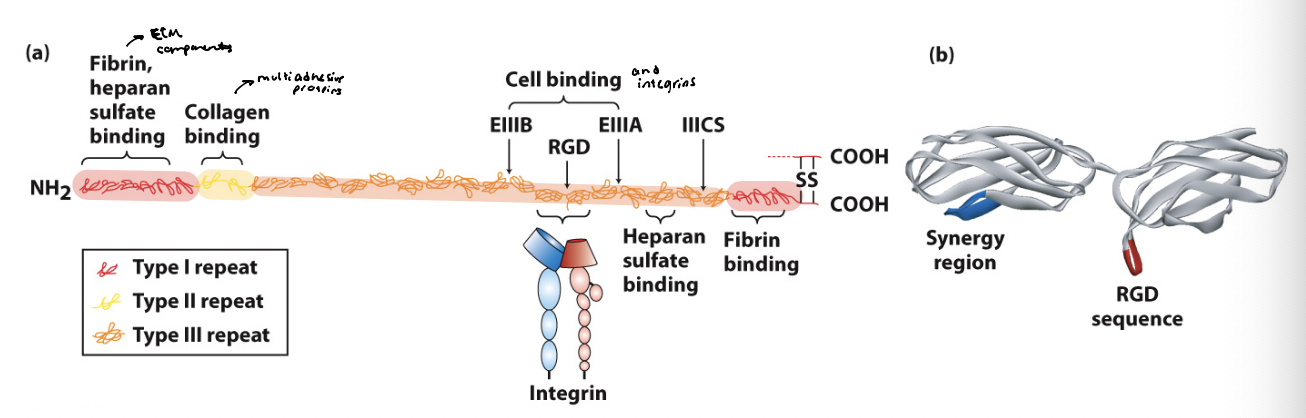
fibronectin
a dimer consisting of two polypeptides linked at the C-terminus by disulfide bonds
contains 3 functional domains → type I/II/III repeats
combination of repeats on different isoforms allows it to bind multiple ligands
consists of 20 different isoforms generated via alternative splicing from a single gene transcript
interacts with other ECM components like fibrillar collagen and heparan sulfate proteoglycans
binds to adhesion receptors (integrins) to influence shape and movement of cells
essential for cell migration and differentiating during embryogenesis
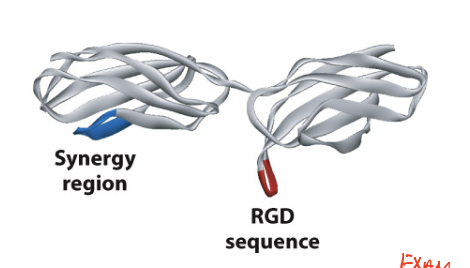
RGD motif
a tripeptide sequence in the cell-binding region of fibronectin is required for cell adhesion (Arg-Gly-Asp)
minimal sequence required for recognition by integrins
found in a loop that protrudes outward from fibronectin
upon synthesis, absorption of fibronectin into ECM helps fold the protein and exposes the sequence
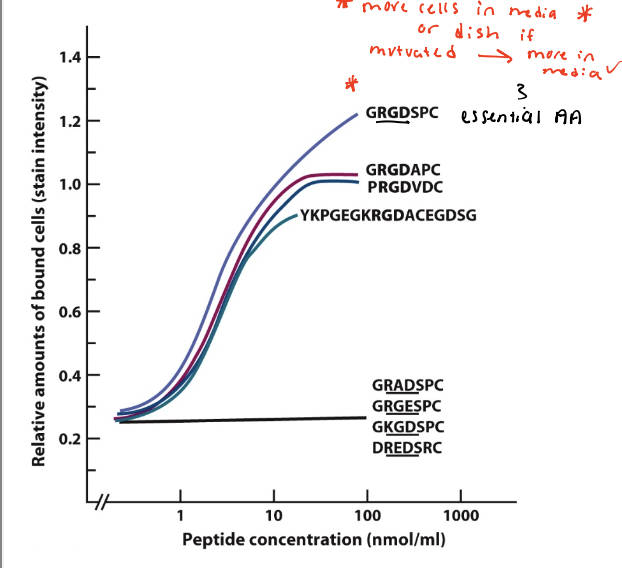
do cells bind RGD-containing peptides?
Various peptide sequences with RGD or scrambled RGD synthesized chemically and plated on dishes
Cultured normal rat kidney cell allowed to adhere to the dishes for 30 minutes
results → cell adhesion increased above the background level with increasing concentration of peptides containing the RGD motif, but not for peptides with scrambled RGD
conclusion → cell surface receptors (integrins) bind to RGD
true
T/F: All integrins evolved from 2 ancient general subgroups:
integrins that bind proteins containing the tripeptide sequence (R) Arg - (G) Gly - (D) Asp motif (fibronectin)
integrins that bind laminin (occurs thru non-RGD recognition motifs)
made up of α and β subunits
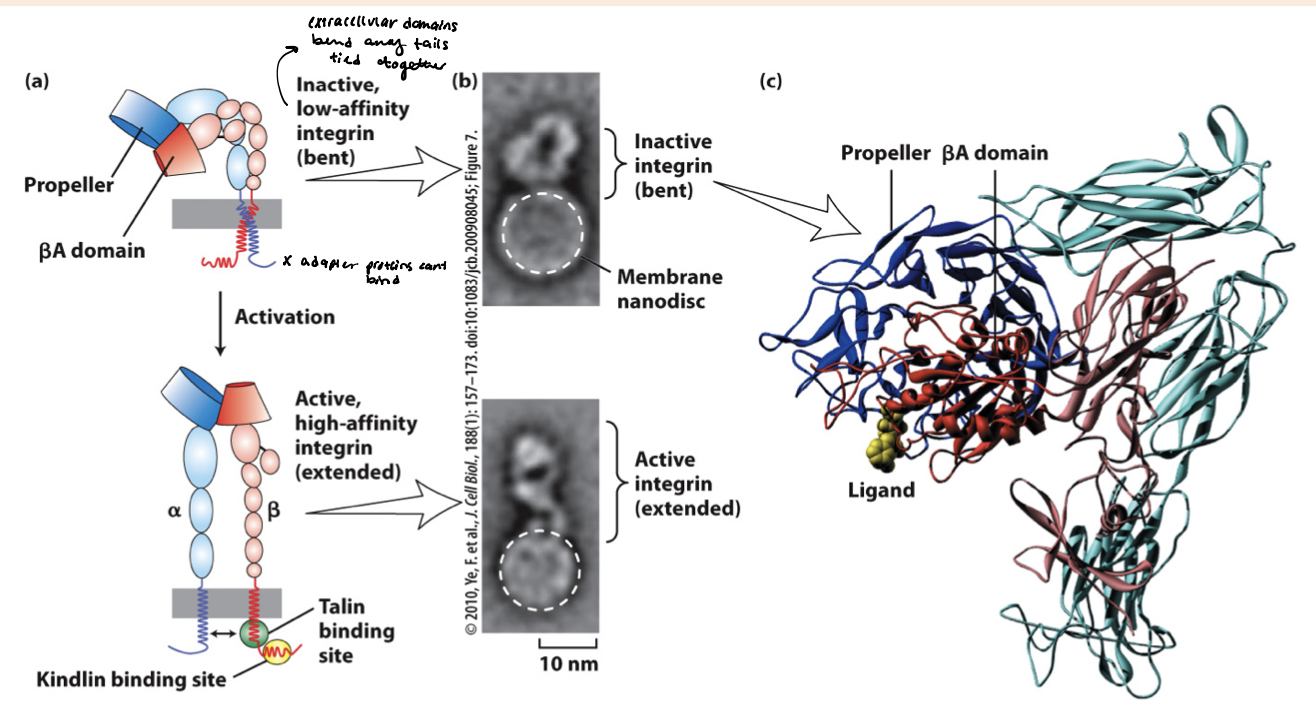
integrin active/inactive state
inactive state → the BA propeller domains are bent (permitting low affinity ligand binding) and cytoplasmic tails are closely intertwined
active state → separation of heterodimers transmembrane and cytoplasmic domains, revealing binding sites for adapter proteins
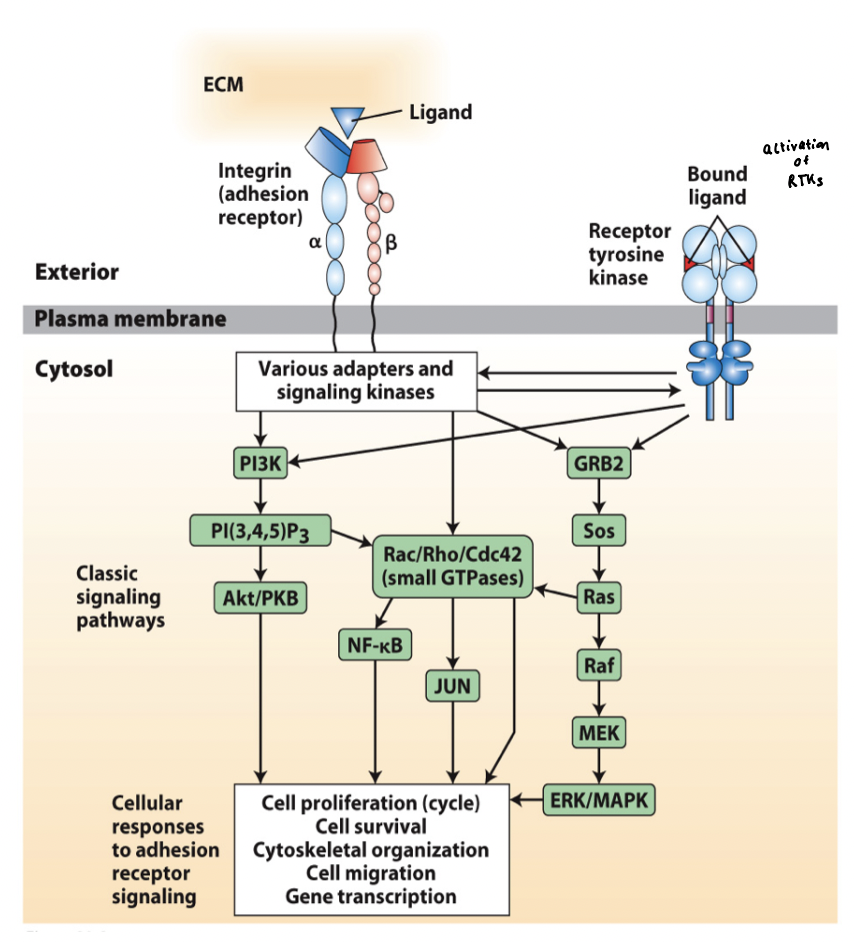
integrin receptor mediated signaling
Integrins interact via adapter proteins and signaling molecules with a broad array of intracellular signaling pathways
Integrin signaling is activated by both ECM binding and cytoskeletal interaction (bidirectional integrin signaling pathway)
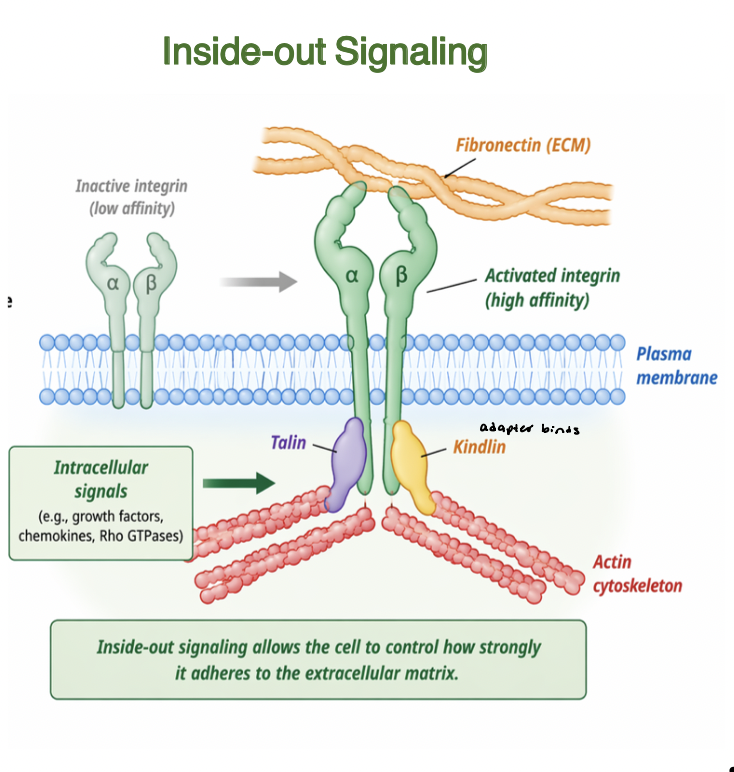
inside-out signaling
Intracellular signals regulate integrin activation
adapter proteins (talin, kindlin) bind integrin cytoplasmic tails
induce conformational change to high affinity state
increases binding to ECM ligands (fibronectin)
enhances adhesion strength and clustering
links intracellular cell state to ECM
outside-in signaling
Binding of ECM ligands activates integrins
induces conformational changes in cytoplasmic domains
recruits adapter proteins (talin, paxillin, vinculin)
activates signaling pathways FAK, Src, and ILK
regulates cytoskeletal organization and cell behavior
links ECM properties to intracellular responses
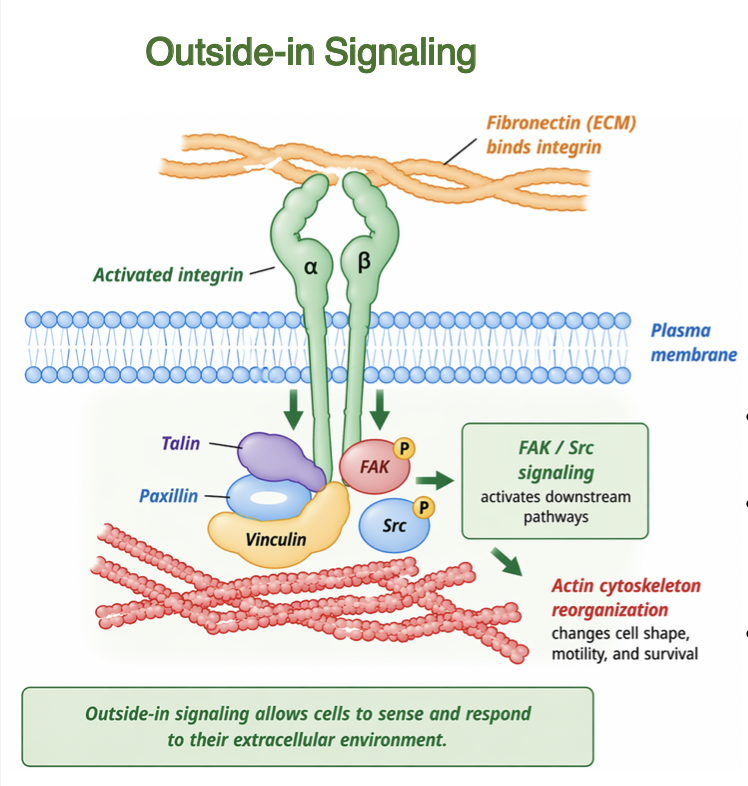
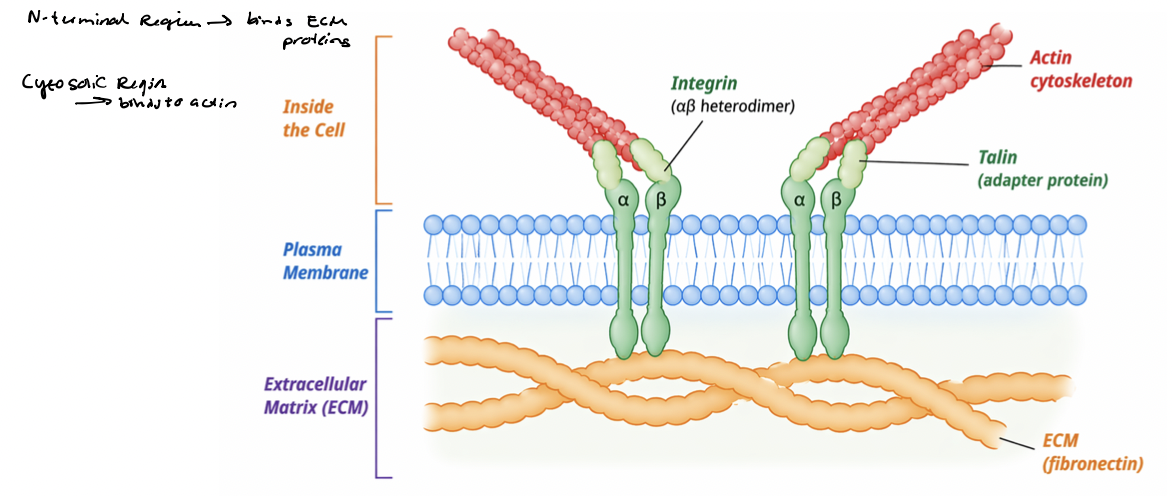
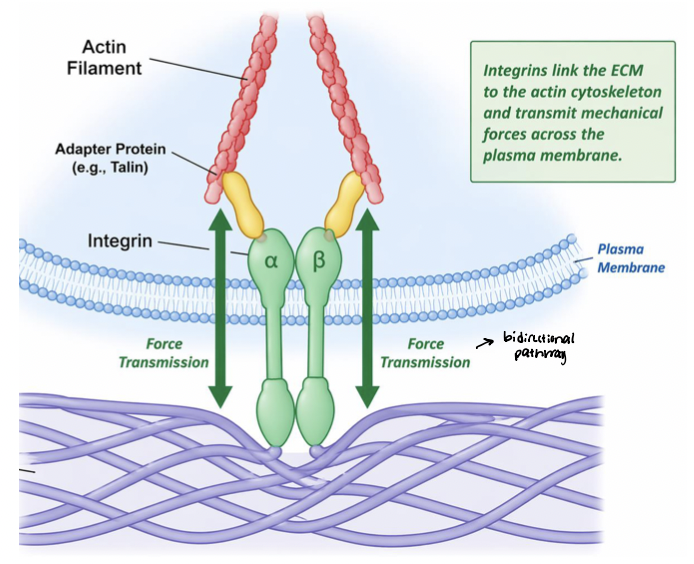
integrin link ECM to cytoskeleton
Integrin signaling is coupled to their physical linkage to the cytoskeleton
integrins physically connect ECM proteins (fibronectin) to actin filaments
physical linkage enables transmission of mechanical forces
adapter proteins link integrin cytoplasmic tails to cytoskeleton
actin filaments terminate at adhesion sites at PM
integrin clustering organizes adhesion complexes
Cells bind the ECM but fail to transmit forces to the cytoskeleton
A mutation prevents integrins from binding to intracellular adapter proteins (talin). Which of the following is the most likely outcome?
a. Cells cannot bind ECM ligands
b. Cells bind the ECM but fail to transmit forces to the cytoskeleton
c. Cells lose cadherin-mediated adhesion
d. ECM proteins cannot assemble properly
It allows many weak interactions to act together to form strong adhesion
Which of the following best explains why clustering of adhesion molecules strengthens cell adhesion?
a. It increases the affinity of individual binding interactions
b. It reduces the number of ligand-binding sites
c. It allows many weak interactions to act together to form strong adhesion
d. It prevents intracellular signaling
Reduced focal adhesion formation
A cell is placed on a surface lacking fibronectin. Which of the following is most likely to occur?
a. Increased integrin activation
b. Reduced focal adhesion formation
c. Increased actin polymerization at the membrane
d. Enhanced cadherin-mediated adhesion
Intracellular adapter proteins induce conformational changes that increase ligand affinity
Which of the following best explains how integrin activation can be regulated from inside the cell?
a. Intracellular adapter proteins induce conformational changes that increase ligand affinity
b. Binding of ECM ligands causes integrins to be internalized
c. Integrins degrade ECM components to expose binding sites
d. Integrins detach from the cytoskeleton to increase mobility
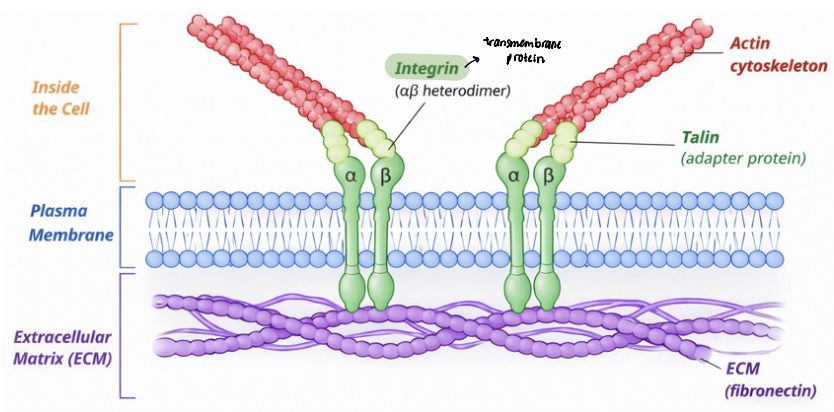
adhesion sites
integrin signaling is coupled to their physical linkage to the cytoskeleton
adapter proteins link integrin cytoplasmic tails to the cytoskeleton
integrins physically connect ECM proteins (fibronectin) to actin filaments
actin filaments terminate at _____________ at the plasma membrane
integrin clustering organizes multi-protein adhesion complexes, establishing a continuous structural linkage across the plasma membrane (signaling)
contractile forces
Cells actively generate ___________ via the actomyosin cytoskeleton
myosin motor proteins contract actin filaments via (+) end directed motility
pull inward toward cell center
force generation is dynamic and regulated by signaling pathways
used to probe the mechanical properties of the ECM
enables cells respond to their physical environment
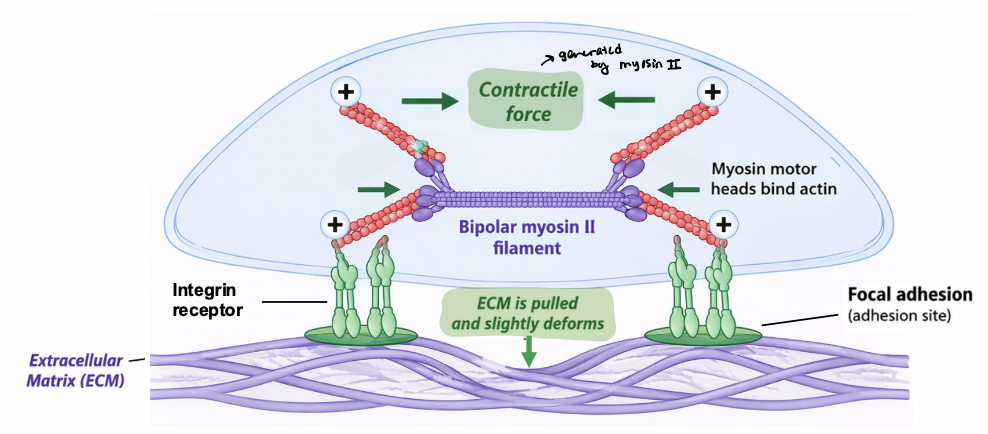
integrins transmit forces
integrins link the ECM to the actin cytoskeleton and transmit mechanical forces across the plasma membrane
contractile forces generated by actomyosin are transmitted to adhesion sites
ECM resists these forces, generating tension
magnitude of tension depends on ECM mechanical properties
establishes physical pathway for bidirectional signaling between cell and its environment
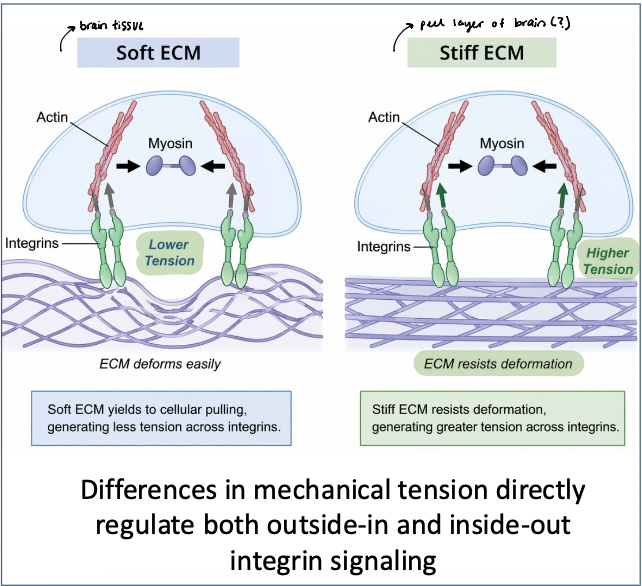
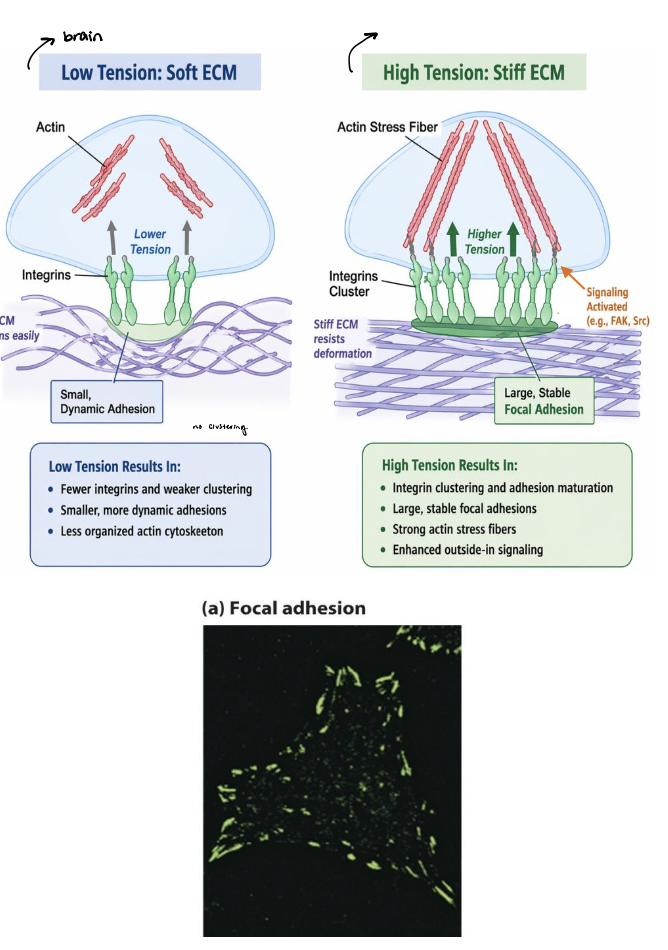
ECM stiffness
soft ECM → deforms easily, leading to lower tension across adhesions
smaller, more dynamic adhesions
stiff ECM → resists deformation, leading to higher tension across adhesion
larger, more stable focal adhesions
integrins and associated proteins respond to tension → mechanosensing
mechanical cues are converted into biochemical signals that regulate cell behavior
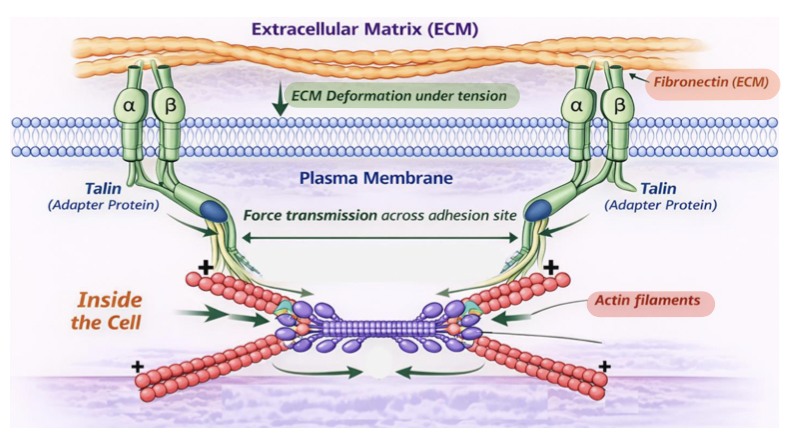
ECM remodeling
Mechanical tension across adhesion sites is used to produce force
this force can stretch ECM proteins such as fibronectin, promoting ECM remodeling
this is how mechanical forces transmitted through integrins are converted into biochemical signals that control cell’s external environment
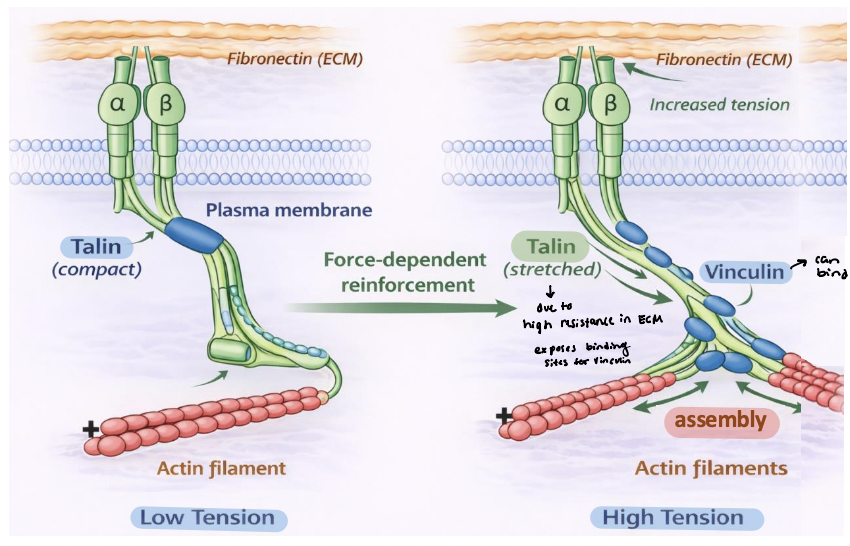
force-dependent adhesion remodeling
Fibronectin stretching causes it to unfold → exposes hidden binding sites (type III domain) that form β sheets with other fibronectin molecules to help promote ECM assembly (outside the cell)
provides molecular mechanism for ________________
Talin stretching at the c-terminus exposes protein interaction sites at adhesions (inside the cell)
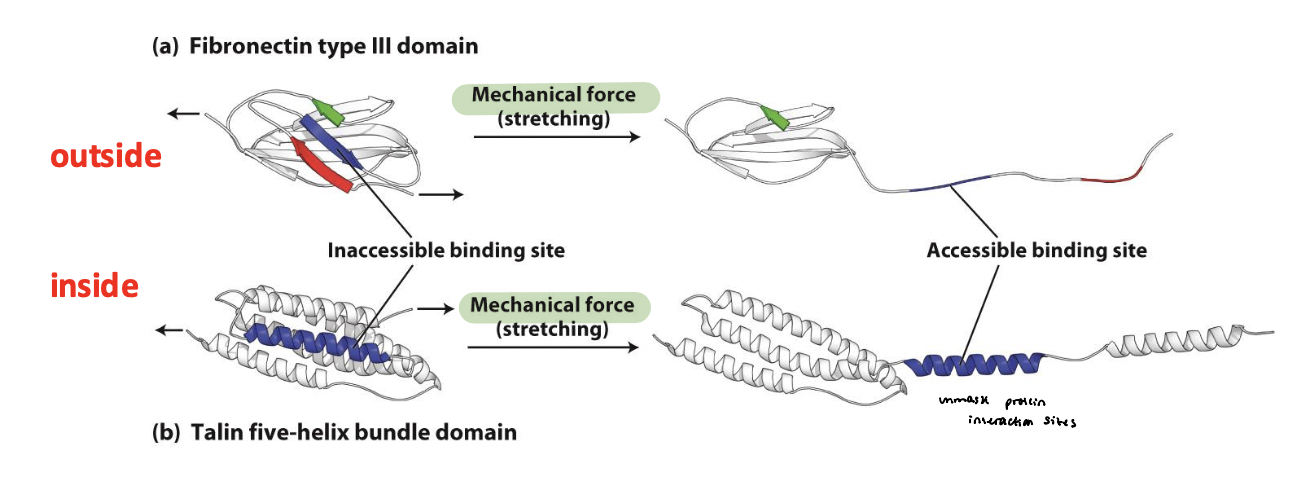
vinculin-binding sites
Mechanical tension stretches talin at integrin adhesions
Talin unfolding exposes cryptic ______________
Vinculin → binds talin and links to additional actin filaments
reinforces the integrin-cytoskeleton connection
promotes assembly and stabilization of actin bundles
drives growth and maturation of focal adhesions
true
T/F: Mechanical tension is the primary driver that controls the organization of adhesion complexes (depends on ECM properties and the environment)
true
T/F: Adhesion structure reflects the mechanical environment of the cell (why the external environment is so critical for cell health and function)
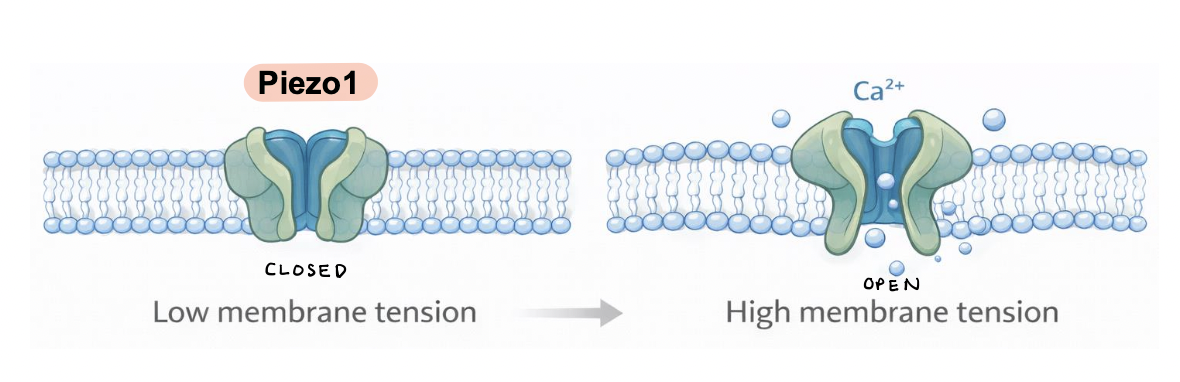
Piezo1
a mechanosensitive ion channel embedded in the plasma membrane
membrane stretch induces channel opening, allowing Ca2+ influx into the cell
Ca2+ signaling regulates cytoskeletal dynamics, gene expression, and cell behavior
proteolytic cleavage
_____________ of ECM liberates growth factors and signaling molecules that were previously sequestered within the ECM
metalloproteases (MMPs)
remodel and degrade the ECM; exist as both membrane tethered and secreted enzymes
3 classes
collagenases
gelatinases
elastases
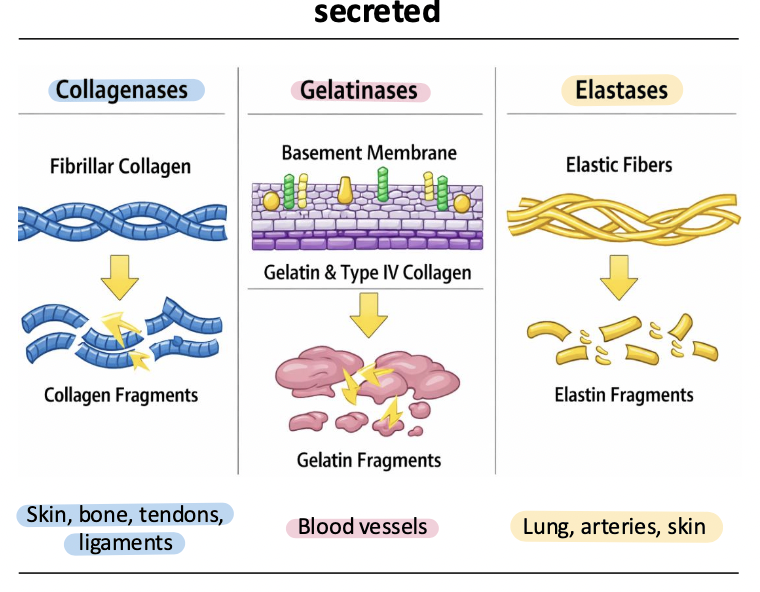
collagenases
MMP-1, MMP-8, MMP-13
cleave fibrillar collagens (types II, II, III)
initiate breakdown of highly structured collagen fibers during tissues remodeling, wound healing, and development
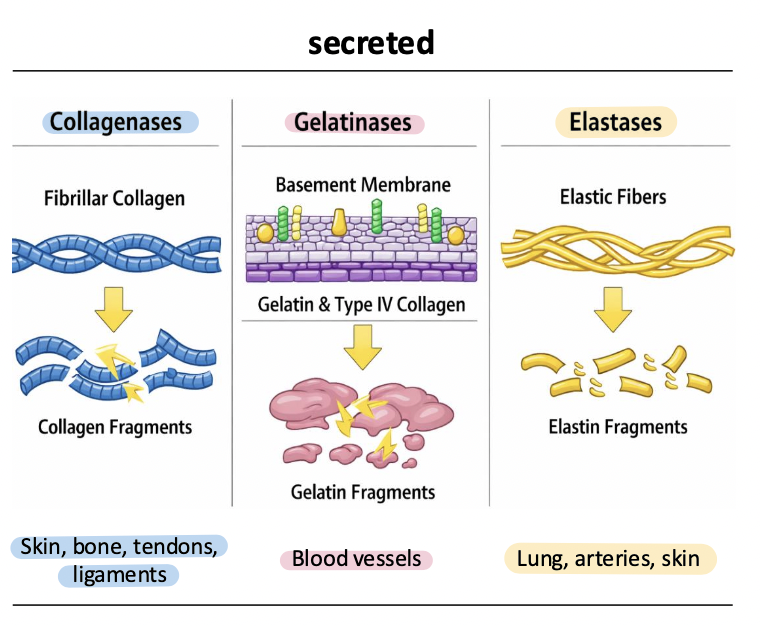
gelatinases
MMP-2, MMP-9
degrade gelatin, Type IV collagen, and basement membrane components
critical for cell migration, angiogenesis, and invasion across basement membranes
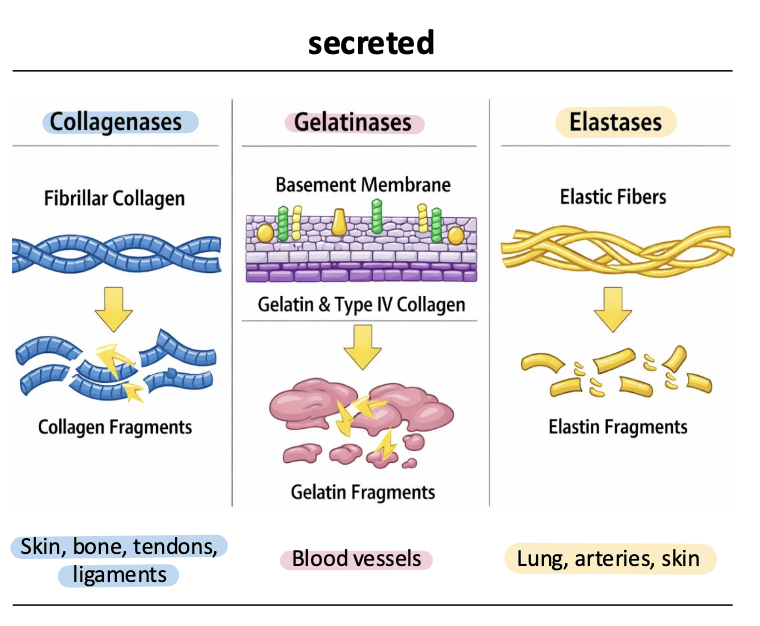
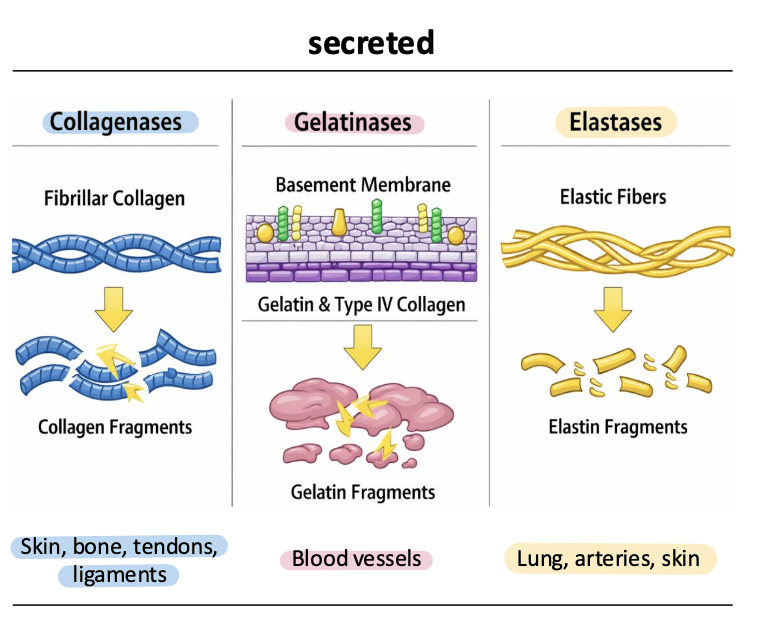
elastases
MMP-12, MMP-7
degrade elastin and elastic fibers
regulate tissue elasticity (smooth muscle) and remodeling, esp in lung, vasculature, and inflammatory responses
ADAMs
membrane-tethered metalloproteases localized to the plasma membrane
mediates ectodomain shedding by cleaving extracellular domains of transmembrane proteins
regulate release and activation of signaling molecules (cytokines, growth factors, receptors, adhesion molecules)
function as key interface between ECM and ICM
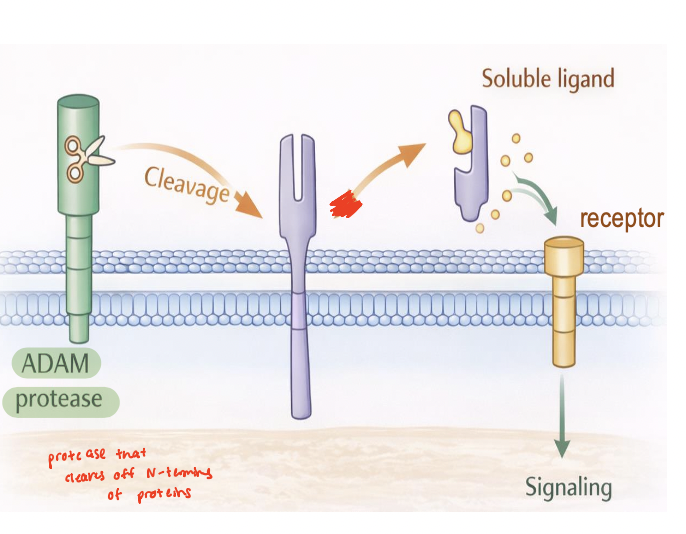
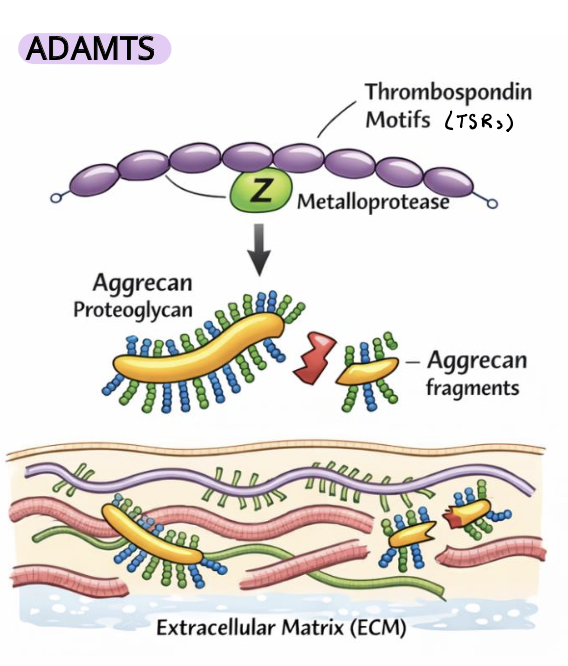
ADAMTs
soluble (not membrane-bound) MMPs that function in the extracellular space to remodel the ECM
regulate ECM turnover and mechanical properties
control tissue stiffness, elasticity, and cell–matrix signaling environments
Thrombospondin motifs (TSRs) mediate ECM interactions and substrate specificity
they cleave key ECM components such as proteoglycans (e.g., aggrecan) and other matrix proteins,
key roles in development and tissue organization, including bone/cartilage and vasculature (affected in disease)
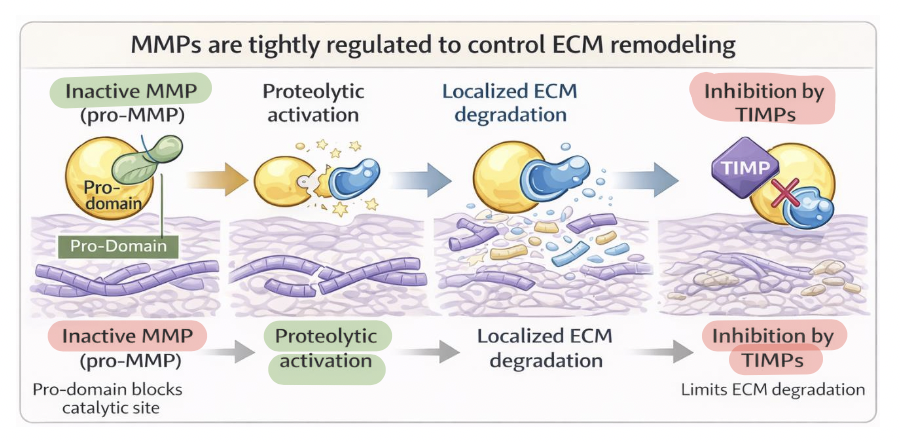
metalloproteases (MMP) regulation
MMP activity reflects a balance between activation and inhibition
synthesized as inactive zymogens (pro-MMPs)
pro-domain blocks the catalytic site to prevent premature ECM degradation
activation → by proteolytic cleavage or conformational change
inhibition → by TIMPs, bind active MMPs to limit proteolysis and maintain ECM balance
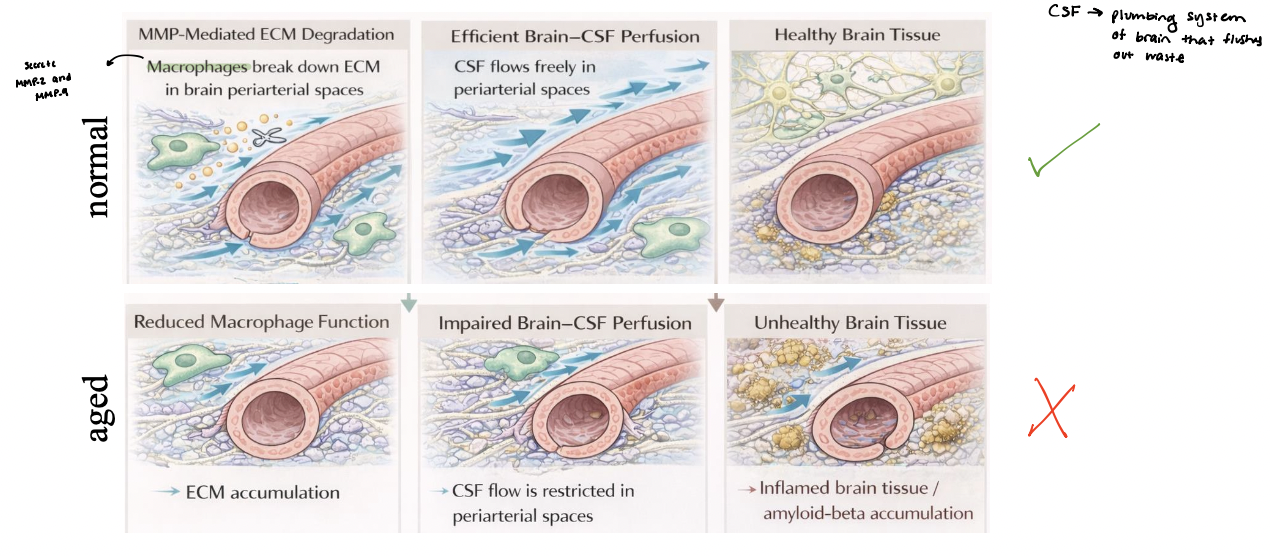
macrophage-mediated ECM remodeling
regulates brain-CSF perfusion
macrophages in periarterial spaces secrete matrix metalloproteases (MMPs) to remodel ECM
MMP-mediated degradation of collagen and laminin maintains an open periarterial pathway
Efficient ECM remodeling supports robust brain–CSF perfusion along periarterial spaces
aging or dysfunction → reduced macrophage activity leading to ECM accumulation and narrowing of flow pathways
impaired CSF perfusion decreases waste clearance, contributing to accumulation of toxic proteins and tissue dysfunction
polarization
Cells that build tissues show ___________, with adhesion molecules generating and maintaining distinct surfaces
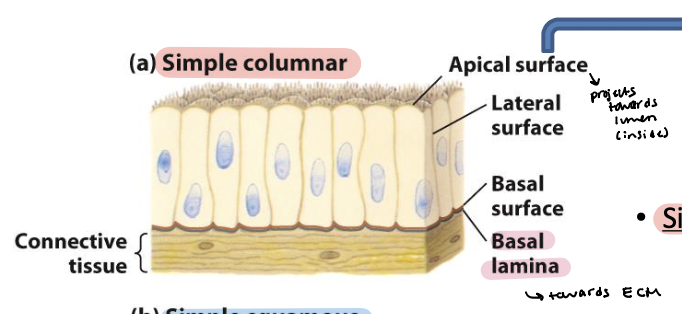
simple columnar epithelia
Elongated cells – including mucus-secreting cells (lining of
the stomach and cervical tract) and absorptive cells (lining
of the small intestine)
Microvilli – on apical surface
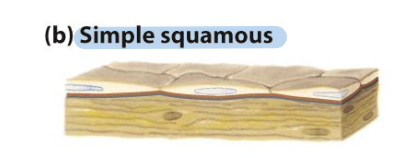
simple squamous epithelia
Thin cells – including cells lining blood vessels (endothelial
cells/endothelium) and many body cavities
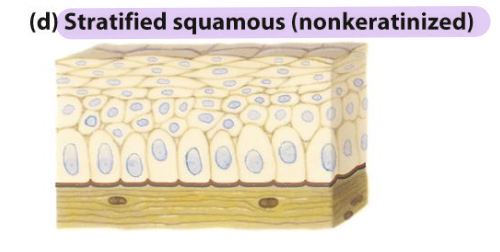
stratified squamous (nonkeratinized) epithelia
Line surfaces such as the mouth and vagina
Resist abrasion
Generally prevent material absorption/secretion into or
out of lined cavity
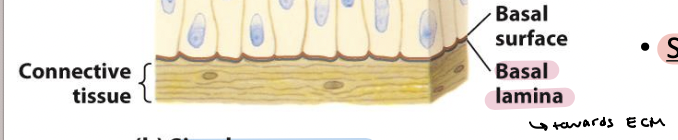
basal lamina
Thin fibrous network of collagen and other ECM components
Connects epithelia to underlying connective tissue
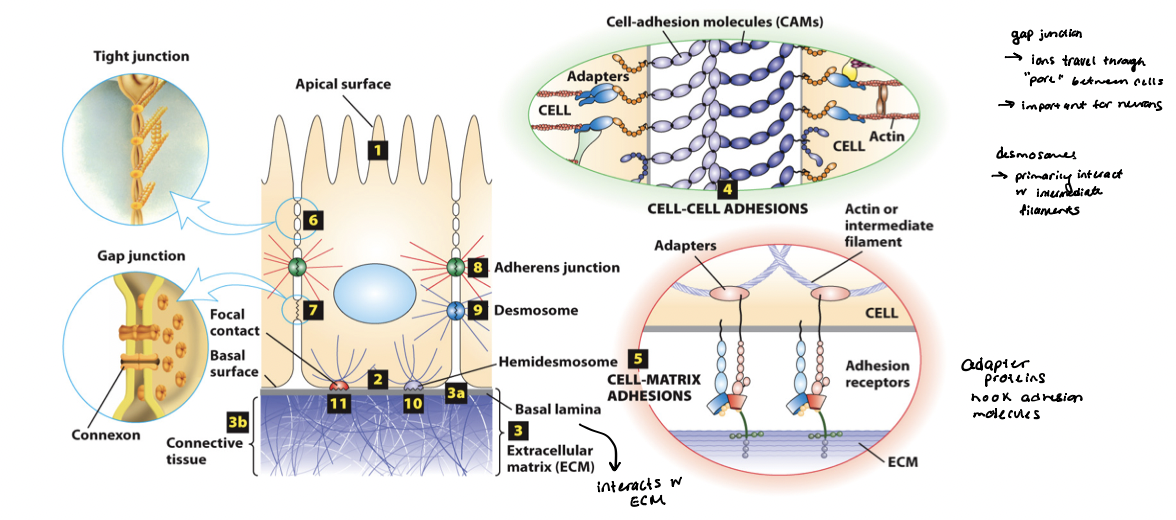
tight junction
Surrounds the cell below the microvilli – connects to all neighboring cells
Regulates paracellular transport of substances between the intestinal lumen and internal body fluids (blood) via the extracellular space between cells
Boundary between apical and basolateral regions of the plasma membrane
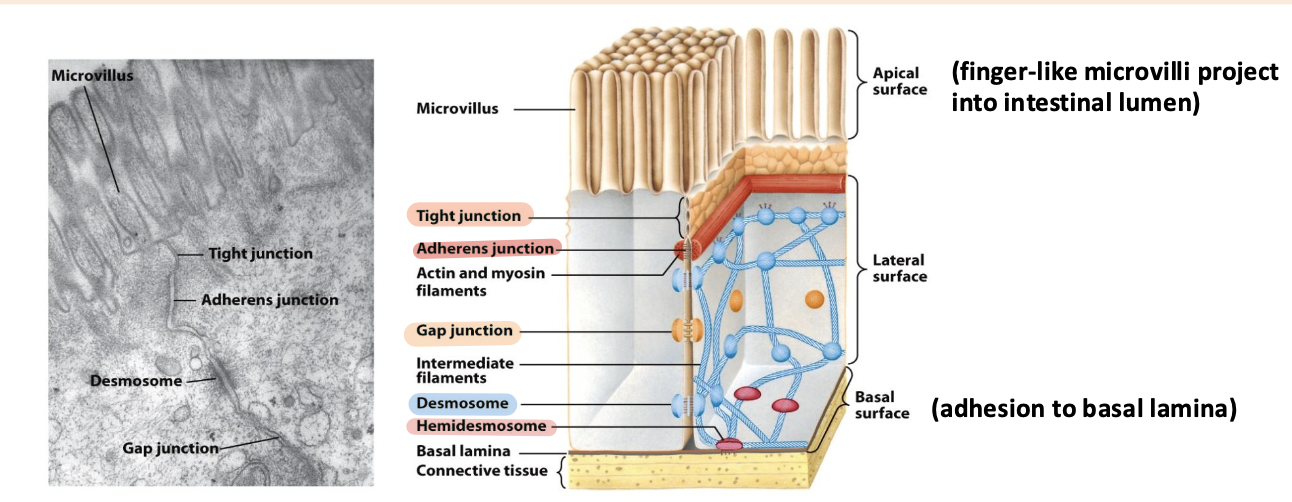
gap junctions
allow movement of small molecules and ions between cytosols of adjacent cells
form pores for cells of certain size diffuse into cell
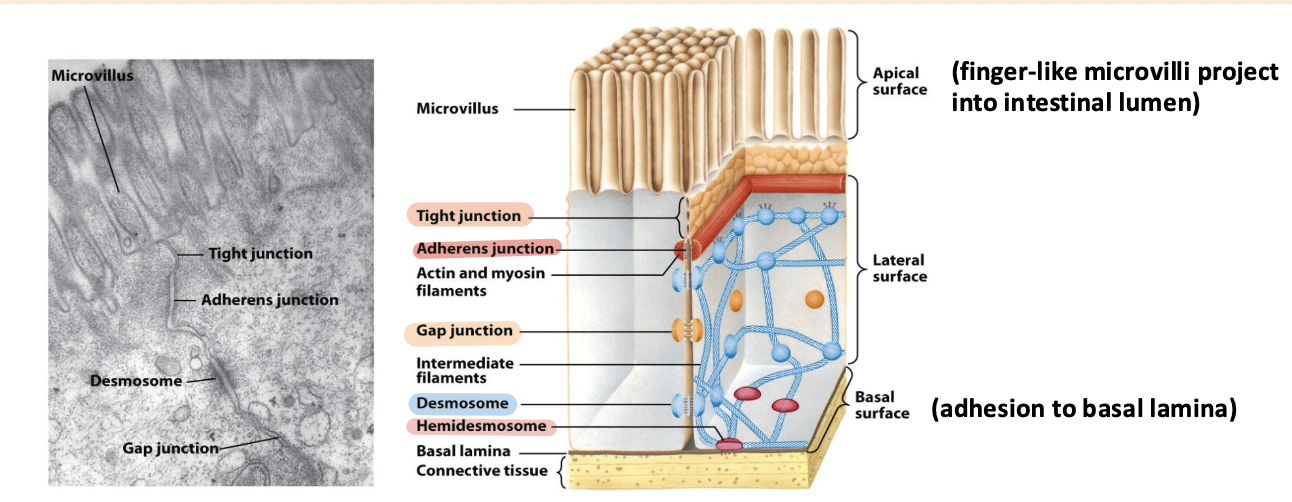
adherens junction
Continuous junction with all neighboring cells
Circumferential belt of actin and myosin filaments associated with the adherens junction – functions as a tension cable that can internally brace and control cell shape
desmosomes and hemidesmosomes
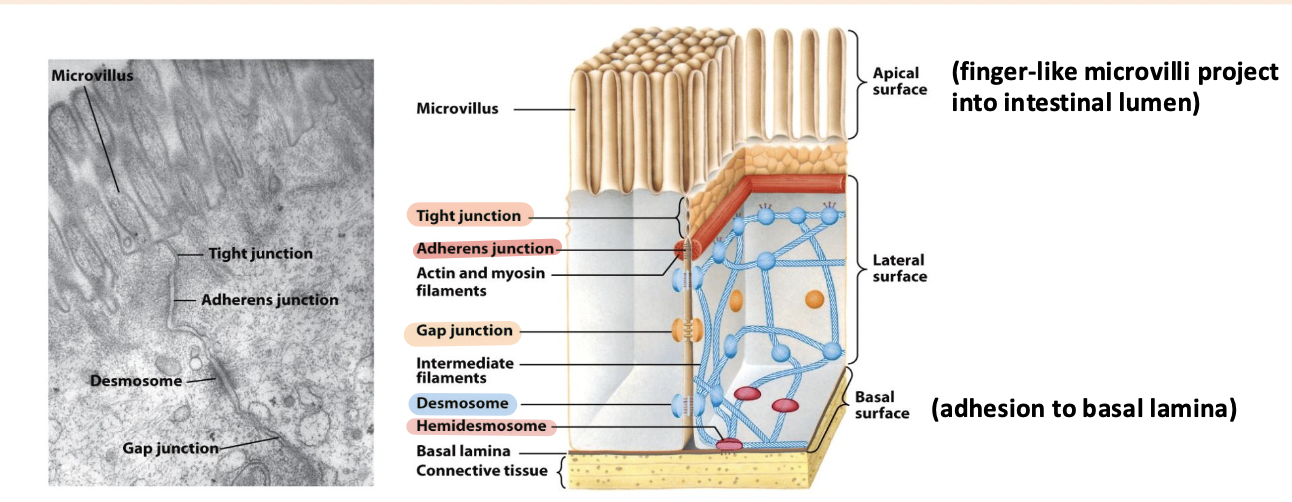
desmosomes
spot cell-cell junctions
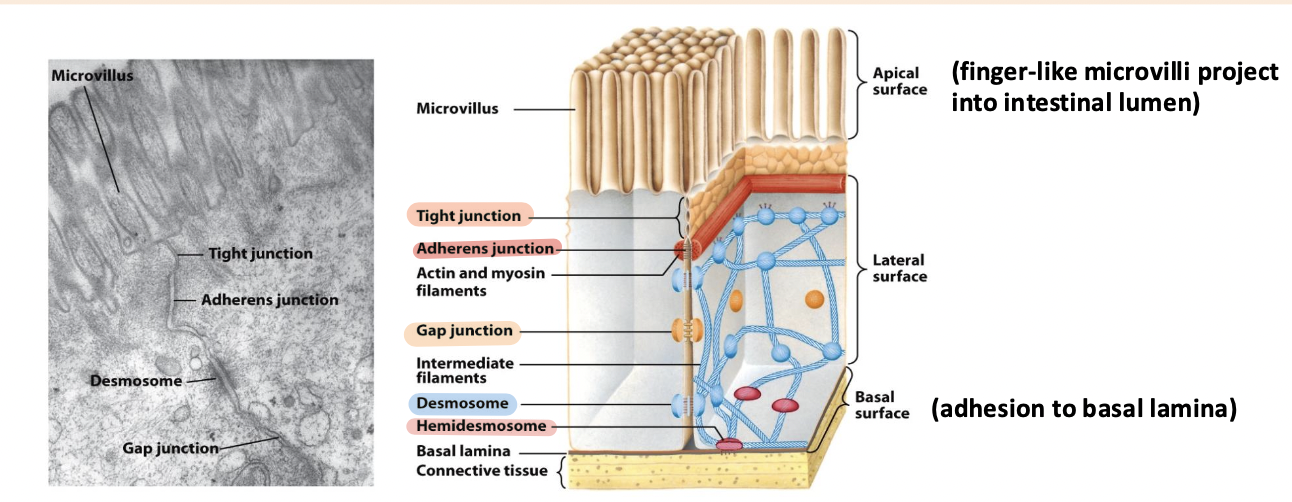
hemidesmosomes
spot cell-ECM junctions, similar to focal adhesion
anchor epithelium to underlying ECM
basal surface (ECM)
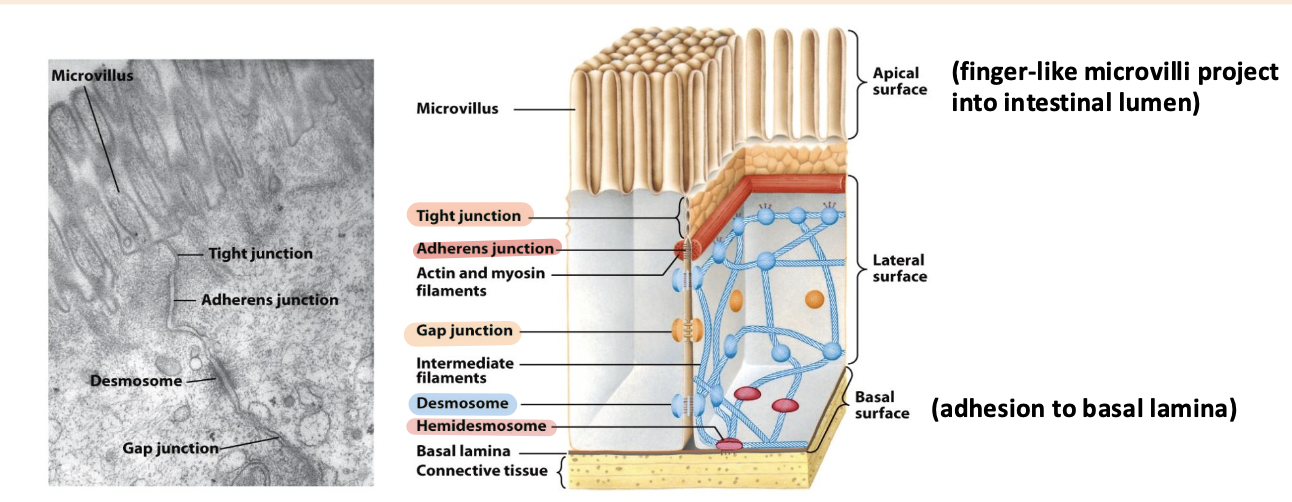
cadherins
mediate cell-cell adhesions in adherens junctions and desmosomes
preferentially mediate homophilic adhesion (E-cad/E-cad)
require Ca2+ for binding
in presence of calcium, mouse fibroblasts do NOT self adhere
E-cadherin transgene, calcium added → cells adhere and clump together
cadherin, no calcium → cells fail to adhere together
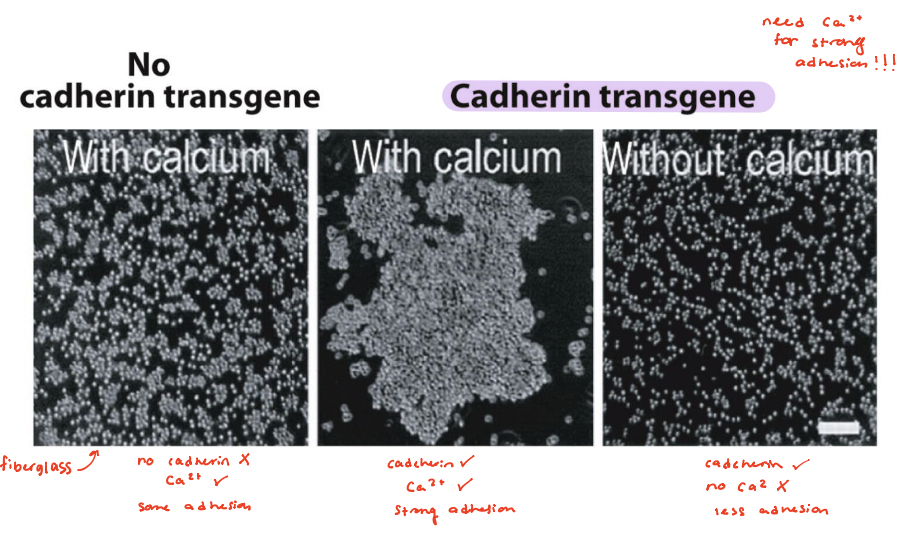
E-cadherin
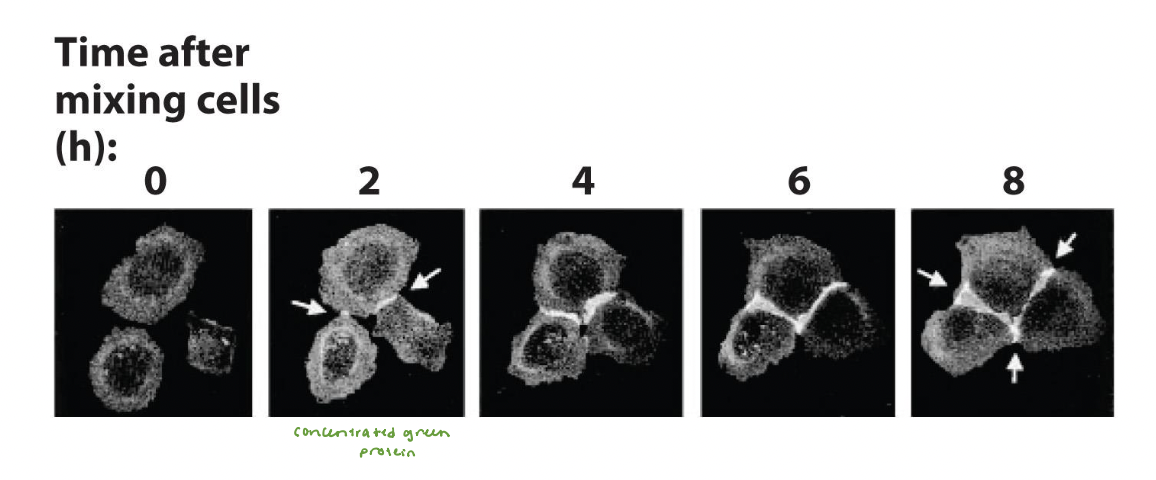
mediates adhesive connections in cultured MDCK epithelial cells
clusters mediate initial attachment of cells into sheets
experiment results:
mediates initial attachment and subsequent zippering together of the epithelial cells
forms bicellular junctions and tricellular junctions
exop