CHEM 315 Exam 3 Conceptual
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
What is Adsorption Chromatography?
The solute is adsorbed on the surface of the stationary phase.
What is Partition Chromatography?
In an open tube, the solute is in the stationary phase of the column surface
What is Ion-Exchange Chromatography?
Mobile phase ions are covalently attracted to the stationary phase ions
What is Molecular Exclusion Chromatography?
Small particles get stuck in column pores, and large molecules pass through
What is Affinity Chromatography?
Specific molecules are covalently bound to the stationary phase, allowing other components to pass through.
What are the stationary phase(s) of Adsorption Chromatography?
Solid
What are the stationary phase(s) of Partition Chromatography?
Liquid
What are the stationary phase(s) of Ion-Exchange Chromatography?
Solid
What are the stationary phase(s) of Molecular Exclusion Chromatography?
Solid
What are the mobile phase(s) of Adsorption Chromatography?
Liquid or gas
What are the mobile phase(s) of Partition Chromatography?
Gas
What are the mobile phase(s) of Ion-Exchange Chromatography?
Liquid
What are the mobile phase(s) of Molecular Exclusion Chromatography?
Liquid or gas
What is a resolution that displays good separation?
R > 1.5
What is retention time?
The time between injection and the arrival of a solute at the detector.
What is partition coefficient?
The ratio of solute concentrations in the mobile and stationary phases.
What is retention factor?
The amount of time a sample spends in the stationary phase relative to the mobile phase
What is separation factor?
Ratio of retention factors (bigger value on top)
How does more plates affect resolution?
Increase resolution.
How does separation factor affect resolution?
A greater separation factor increases resolution.
How does plate height affect resolution?
A shorter plate height increases resolution.
What does the coefficient 'A' stand for in the van Deemter equation?
Multiple flow paths (EddyDiffusion); smaller stationary-phase particles are more ideal
What does the coefficient 'B' stand for in the van Deemter equation?
Longitudinal diffusion; faster flow through the column = less longitudinal diffusion
What does the coefficient 'C' stand for in the van Deemter equation?
Mass transfer between phases
What does the coefficient 'u' (mu sub x) stand for in the van Deemter equation?
Flow rate
What does 'H' stand for in the van Deemter equation?
Plate height
True or False: The van Deemter equation describes peak broadening by quantifying variables as the compound travels through the column.
True.
What increases resolution in Gas chromatography?
Increasing column length,
decreasing temperature,
increasing column film thickness,
decreasing column diameter (narrower columns)
In normal-phase HPLC chromatography, what would elute first?
The least polar compound would elute first.
In reversed-phase HPLC chromatography, what would elute first?
The most polar compound would elute first.
In normal-phase HPLC chromatography, what are the polarities of the mobile and stationary phase?
Mobile: Nonpolar, Stationary: Polar.
In reversed-phase HPLC chromatography, what are the polarities of the mobile and stationary phase?
Mobile: Polar Stationary: Nonpolar
In normal-phase HPLC, which mobile phase would give the best retention?
Low polarity
In reversed-phase HPLC, which mobile phase would give the best retention?
High polarity
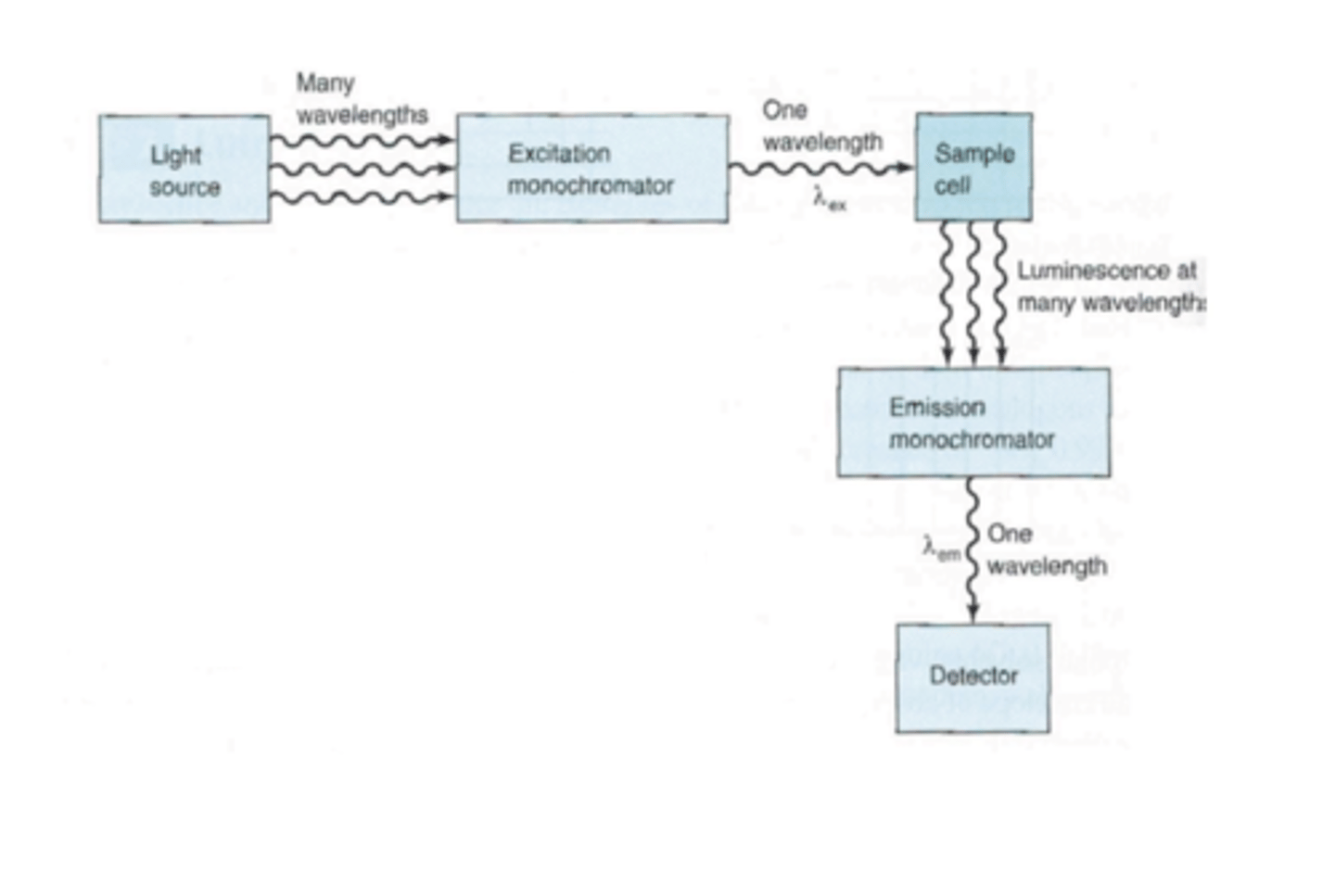
For the following experimental setup, what is being measured?
Emission.
True or False: Reduction describes the gaining of electrons.
True.
True or False: Oxidation describes the loss of electrons.
True.
The oxidizing agent is:
Reduced.
The reducing agent is:
Oxidized.
How does oxidation and reduction affect oxidation number?
Oxidation increases oxidation number; Reduction decreases oxidation number.
True or False: A galvanic cell is where electricity is generated through spontaneous reactions.
True.
True or False: By using the Nernst equation, a linear plot can be generated to determine the concentration of an ion using an ion selective electrode.
True.
How does an ion selective electrode measure ion concentration?
It measures the potential differences between ions in solution.
How can you find the potential of a cell?
Cathode - Anode.
The cell is spontaneous if:
E > 0.
RT/F is equal to 0.05916 under what specific conditions?
T = 25 C.
For an electrochemical cell, what is the line notation?
| Anode || Cathode |
What happens to the anode in an electrochemical cell?
It gets oxidized.
Redox Titration
A method used to determine the concentration of an oxidizing or reducing agent in a solution by reacting it with a solution of known concentration.