Science Skills and Ethical Guidlines- Psych
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
Independant Variabele
the factor that is deliberately manipulated, changed, or selected by the experimenter in a scientific investigation
Dependant Variable
The factor being measured or observed in an investigation.
Extraneous Variables
any variable other than the IV that can cause a change in the DV and therefore affects the validity or accuracy of the experiment.
- if not controlled, extraneous variables become confounding variables.
Confounding Variable
A confounding variable is a variable other than the IV that has had an unwanted affect on the DV, making it impossible to determine which of the variables has produced the predicted change in the DV
Hypothesis
It is hypothesied that "population who is "exposed to the IV" would have a "impact on the DV- direction" than "population" not exposed to the "IV"
Within Experimental Design
Each participant or subject is in both experimental and control groups, exposed to all treatment conditions
Strengths of Within Experimental Design
The conditions are identical, therefore it can effectively control the unwanted influence of variables arising from individual participant differences.
This design requires a smaller amount of participants as they are in all conditions.
Limitations of Within Experimental Design
Although this design keeps individual participant differences constant, it does not necessarily control all participant variables that can influence the results.
Between Experimental Design
Uses two groups often one group as the experimental group and the other the control group (not exposed to the IV)
Strengths of a Between Experimental Design
Time efficent has both the experimental and control groups can be conducted at the same time. There is also no order effects between conditions to control
Limitations of a Between Experimental Design
There is often a need for a larger amount of participants to help ensure the spread of participant variables within the sample will match the distribution within the population.
Allocation Sampling Method
The method of assigning participants from a sample into different experimental conditions (e.g., control or experimental groups) after they have been selected.
Random Sampling
Sampling technique that ensures every memeber of the population has an equal chance of being selected to be part of the sample

Stratified Sampling
The process of selecting a sample from a population comprised of various subgroups, then selecting a separate sample from each subgroup.
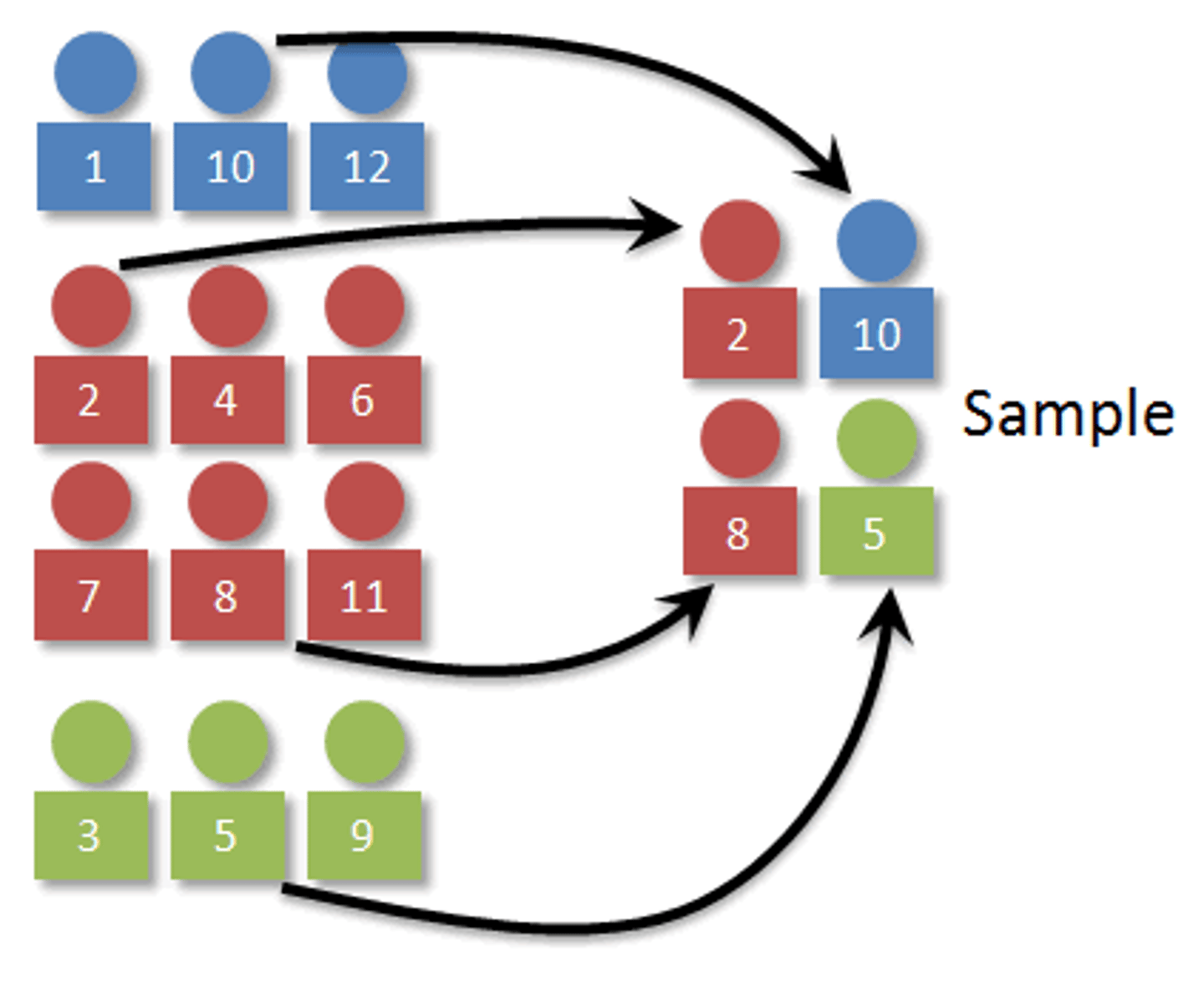
Correlational Study
A research project used to investigate the relationship that exists between variables without any control over the setting in which the relationship occurs.
- reference may be made to the direction of the relationship between the variables and the strength of the relationship
Positive Correlation
Two variables vary in the same direction
- as one variable increases, the other tends to increase
Negative Correlation
Two variables change in opposite directions
- as one variable increases, the other tends to decrease
Zero Correlation
There is no relationship between the two variables
Strengths and Limitaions of Corelational Studies
Although, Correlational Studied examine how variables are naturally related they do not permit the researcher to draw firm conclusions about its cause and affect relationship.
Controlled Experiment
An experimental study examining the connection between the IV and DV while controlling all other variables
Fieldwork
The practical application of psychological theories in real-world settings outside the lab, such as schools, hospitals, or communities
- It involves direct observation, data collectiona and engagement with individuals to understand behaviour in a real-world setting
- does not control external variablers like weather
Simulation Modelling
A model to imitate a real or theoretical psychological system, allowing them to study complex behaviours, processes, or systems
- Because simulations are artificial, they may not perfectly replicate real-life, leading to lower ecological validity compared to actual observations.
Difference between Quantitative and Qualitative Data
Quantitative data is numerical, measurable, and answers "how many" or "how often," while qualitative data is descriptive, interpretative, and answers "why" or "how"
Primary Data
Raw information that is collected directly by researchers through methods like interviews, experiments, observations and surveys.
- tailored to answer specific reseach questions however can require significant amount of time effort and costs
Secondary Data
Information collected by someone other than the user, such as previous research studies, government statistics, or archival records.
- it is generally a cheaper, faster and more convenient way to gather data
- The researcher has no control over the data collection process, which may lead to invalidity and outdated data.
Measures of Central Tendency
Mean, median, mode
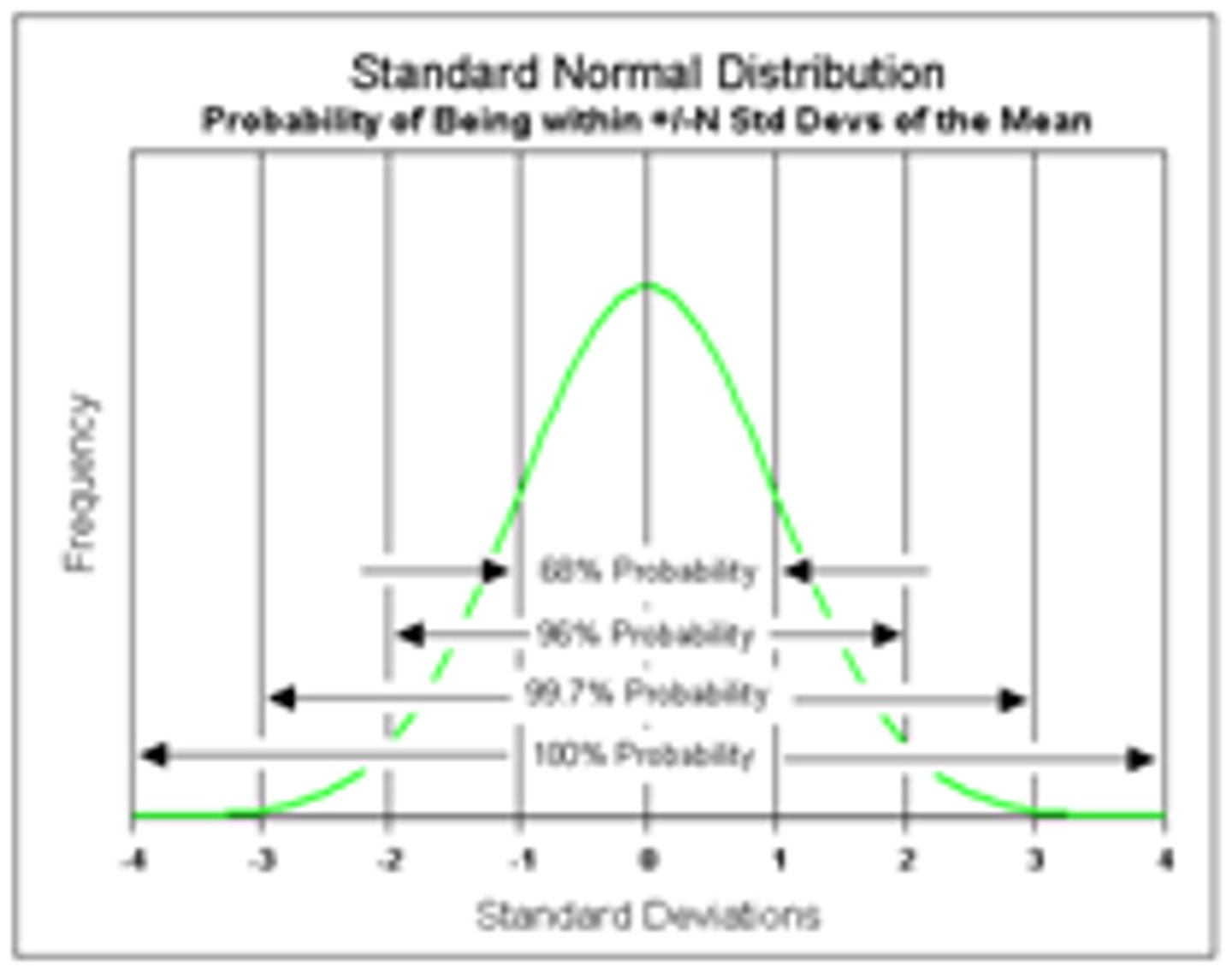
Standard Deviation
Indicates that the widely distributed scores are distributed around a central point
- a higher standard deviation indicates a greater spread of scores from th mean
Low standard Deviation
the data points in a dataset are clustered tightly around the mean (average) value, inclduing high consistency.
Repeatability
The degree in which a specific research investigation obtains similar results when it is conducted again under the same conditions on all occasions
Reproducibility
How close the results are to one another when an investigation is replicated under changed conditions such as different day, temperature, etc.
Validity
refers to the extent to which the procedures used for a research study measure what it is intended to measure.
Beneficence
Minimise risks and ensure benefits in society
Integrity
Accurate and responsible reporting of findings
Justice
There is a far distrubution of costs and benefits of the investigation
Non Maleficence
Avoiding the causations of harm
Respect
Recognise that all individuals, both human and non human have values and importance.
Voluntary Participation
Participants must be willing and not feel pressured to take part in the study
Informed Consent
If appropriate, the participant must be informed of the nature and purpose of the study. A form or document must be signed to participate in an experiement.
Confidentiality
Participants have a right to privacy, and the details of their involvement should not be published in a way that enables them to be identified.
Withdrawel Rights
Participants have the right to decline to participate the study without giving reason and without receiving any negative consequences
Debriefing
On completion of the study, the participant must obtain information about the study, including its true purpose, the procedure, reuslts and conclusions
Decpetion
Information is withheld from the participant or they are deliberately misinformed as their knowledge of certain information may influence the results.