E.2 Quantum physics
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
What is the photoelectric effect and what does it prove?
It is the emission of electrons from a metal surface when light shines on it, providing evidence for the particle nature of light.
What is the threshold frequency in the photoelectric effect?
It is the minimum frequency of incident light required to release a photoelectron from a specific metal surface.
How is the maximum kinetic energy of a photoelectron calculated?
Maximum kinetic energy equals the energy of the photon (Planck constant times frequency) minus the work function of the metal.
What is the work function of a metal?
The minimum energy required for an electron to escape from the surface of the metal.
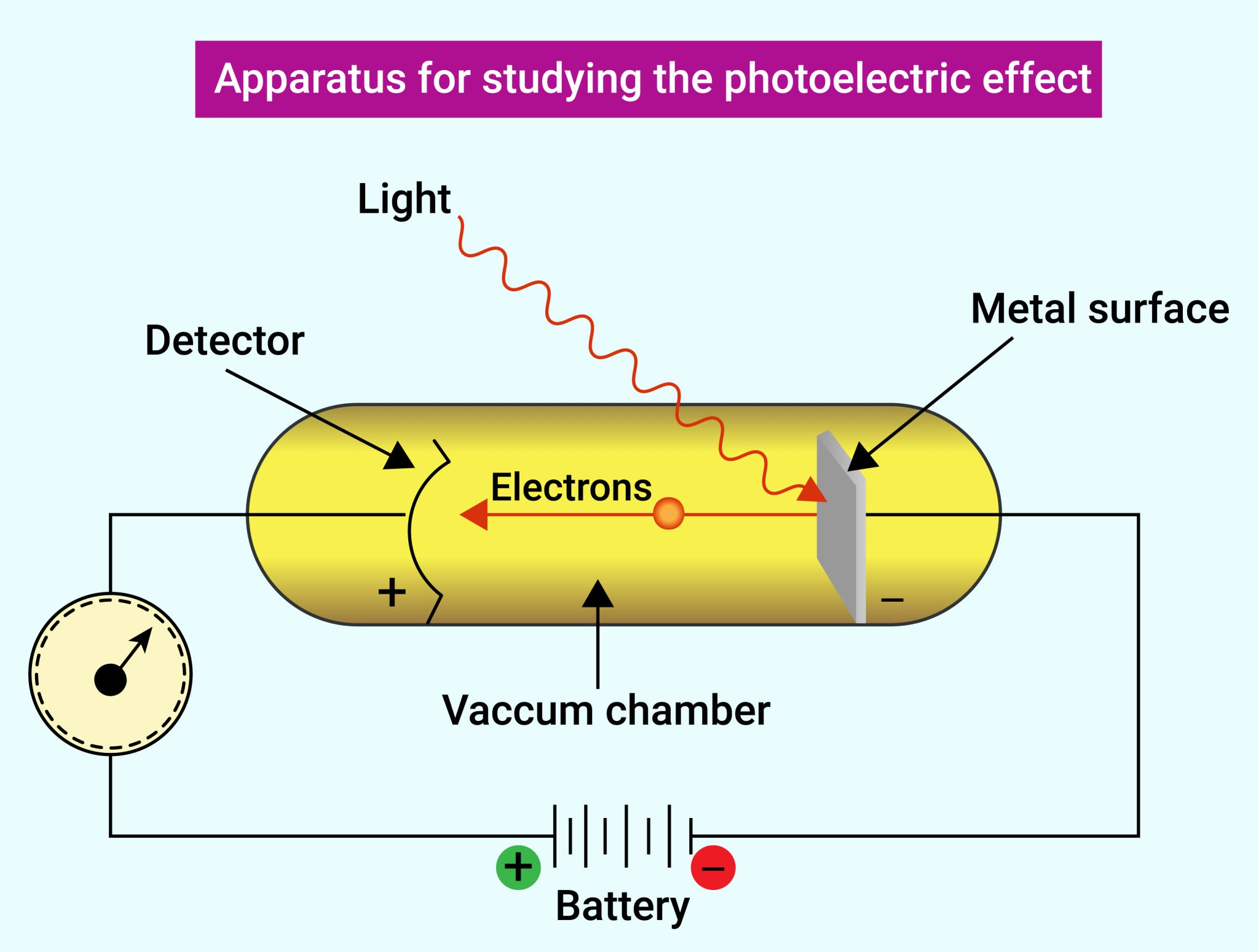
Why can't the classical wave theory explain the photoelectric effect?
Wave theory predicts that any frequency of light should eventually eject electrons if the intensity is high enough, but in reality, it only depends on frequency.
What provides evidence for the wave nature of matter?
The diffraction of particles (like electrons passing through a crystal lattice) shows that matter can exhibit wave-like behaviors.
What is the de Broglie wavelength formula in words?
Wavelength equals the Planck constant divided by the momentum of the particle.
What is meant by wave-particle duality?
The concept that both light and matter exhibit both wave-like and particle-like properties depending on the experiment being performed.
What happens during Compton scattering?
A photon collides with an electron and scatters with a longer wavelength, proving that photons carry momentum like particles.
What is the formula for the Compton wavelength shift in words?
The change in wavelength equals the Planck constant divided by the quantity (mass of the electron times the speed of light) times the quantity (one minus the cosine of the scattering angle).
In a particle diffraction experiment, what determines the location of the minimum intensity?
The location is determined by the de Broglie wavelength of the particles and the width of the aperture they pass through.
Why is Compton scattering considered "more convincing" than the photoelectric effect for the particle model?
It demonstrates a clear "billiard ball" style collision where both energy and momentum are conserved between a photon and an electron.