mineralogy exam 1
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
What are the five characteristics of a mineral?
Naturally ocurring
Inorganic
Definite chemical composition
Crystalline structure
Solid
What are the 8 most common elements that make up minerals?
Si, O, Al, Ca, Na, K, Mg, Fe
Write electron configurations for Ca, Mg, Si, Fe, K and their ions.
Ca: 1s2 2s2 2p6 3s2 3p6 (4s2)
Mg: 1s2 2s2 2p6 (3s2)
Si: 1s2 2s2 2p6 (3s2 3p6)
Fe: 1s2 2s2 2p6 3s2 3p6 (4s2 3d6)
Find the percent ionic character for H-Cl, Ti-O, and Na-Cl.
20%; 60%; 70%
Describe the differences between ionic, covalent, intermediate, metallic, and non-valence dependent bonds.
Ionic: donating electrons, between atoms with extreme electronegativity differences (electrostatic attraction), brittle deformation
Covalent: sharing electrons, between atoms with high but equal electronegativities, stronger than ionic (shorter bond)
Intermediate: between ionic and covalent
Metallic: free flow of electrons, between atoms with low but equal electronegativities, ductile deformation
Non-valence dependent: relies on weak electrostatic intermolecular forces (Van der Waals bonds, hydrogen bonds)
What is electronegativity? What is its trend across the periodic table?
A measure of the tendency of an atom to attract a bonding pair of electrons; increases across a period and decreases down a group
What are the two kinds of ions? How do their sizes change?
Anion (-): gain electrons, increase in size
Cation (+): lose electrons, decrease in size
What is the trend of atomic radii across the periodic table?
Decreases down a group, inreases across a period
What is effective radius vs. bond length?
The distance from the center of an atom or ion to the edge; the distance between the centers of two adjacent ions
Name the four types of packing of atoms.
Simple cubic: stack directly on top of each other
Cubic close: fills new void spaces, ductile
Hexagonal close: re-covers old void spaces, brittle
Body centered cubic: surrounds a center atom
What is coordination number? How do you calculate it?
How many other atoms are touching an atom; radius of cation/radius of anion
What are Pauling’s Rules?
Radius Ratio Rule: Coordination number of a cation depends on relative size of cation and anion
Charge balance must be achieved
Coordination polyhedra don’t like to share edges or faces.
In crystals with multiple different cations, those with large valence and small coordination number tend not to share polyhedral edges or faces.
Nature tends towards simplicity.
Prewitt’s Addendum: Sum of cation CN = sum of anion CN.
How does ionic radius change with oxidation state (charge) and coordination number?
As charge increases, radius decreases; as CN increases, radius increases; as charge increases, CN decreases
How is bond strength calculated? Calculate for C (+4, CN=3) and Ca (+2, CN=6).
S = charge of cation/CN of cation; 4/3; 1/3
What is the law of parsimony?
Nature keeps the number of unique sites in a crystal structure small.
What are isostructural minerals? What are polymorphs?
Same structure, different composition; same composition, different structure
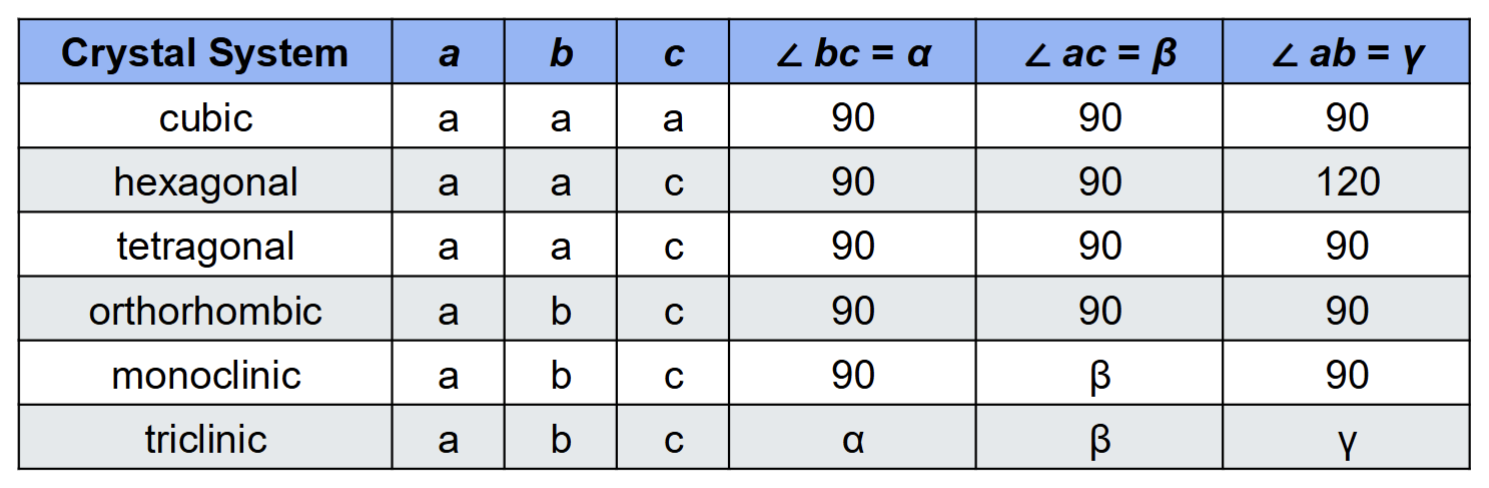
What are the seven crystal families?
Isometric/Cubic: all equal sides, all angles = 90
Tetragonal: two equal sides, all angles = 90
Hexagonal: two equal sides, two 90 and one 120 angle
Trigonal: two equal sides, two 90 and one 60 angle
Orthorhombic: no equal sides, all angles = 90
Monoclinic: no equal sides, two 90 and one obtuse angle
Tricliinic: no equal sides, no equal angles
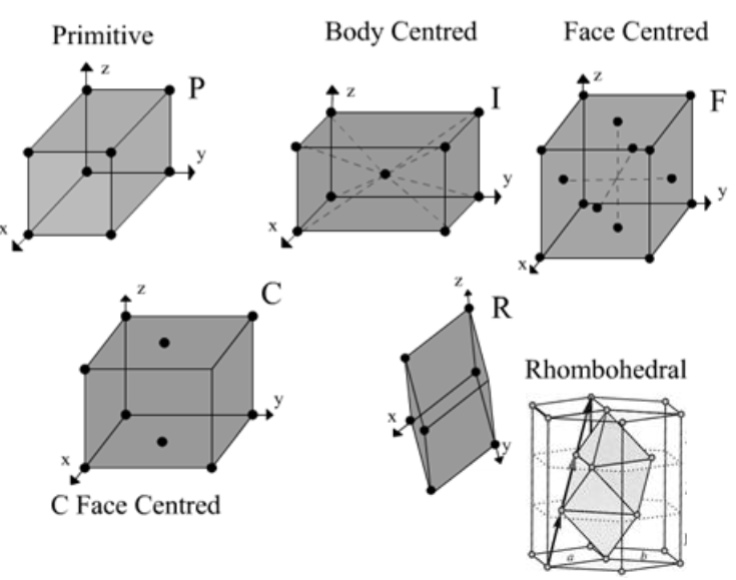
What are the types of unit cell? How are they formed?
Primitive, body-centered, face-cenetered, centered, rhombohedral; produced by translation in 2D
What are the three types crystal face development?
Euhedral: good crystal faces; grown in open cavity
Anhedral: no crystal faces; grown in tight space
Subhedral: intermediate
What is the Law of Bravais?
A crystal face is more commonly developed in a crystal if it intersects a larger number of lattice points.
What are the 2D plane lattices?
Square: a = b, y = 90
Rectangle: a =/ b, y = 90
Diamond: a =/ b, cosy = a/2b, center
Hexagonal: a =/ b, y = 120
Oblique: a =/ b, y =/ 90
How are minerals grouped?
Divided into classes based on chemical composition and structure
What are orbitals? What do subshells represent?
Areas of greatest probability of finding electrons; energy levels
What are the four ways a mineral is formed?
Crystallize from a melt
Solid-solid reactions due to pressure and temperature
Precipitate from a fluid (ex. evaporites)
Condensation from a vapor
How does slow cooling compare to fast cooling?
Less nuclei, bigger crystals; more nuclei, smaller crystals
What is homogeneous vs. heterogeneous nucleation?
Homogeneous: mineral nuclei form in a liquid and overcome metastable state to continue growing
Heterogeneous: mineral nuclei form on a substrate (surface) present in the liquid giving some stability and allowing for easier growth
What is the nucleation barrier?
Melts may cool below the liquidus without crystallizing; nuclei must grow large enough to overcome barrier to continue growing
What happens at an eutectic point?
The first melt to form will always be at the eutectic composition, regardless of the bulk composition of the system
The system remains at the eutectic until all the liquid is consumed and then drifts back to the starting composition (if in equilibrium).
What happens when water is added to a system with a solvus?
Lowers melting temperature
Lowers liquid field into solvus