1) Asymmetric Synthesis Terminology, Houk Model, Allylic Epoxidation and Re & Si Prochiral Carbons
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
What is a stereospecific reaction?
A reaction in which the stereochemistry of the reactant completely determines the stereochemistry or the product.
i.e. starting material and mechanism completely determines the outcome
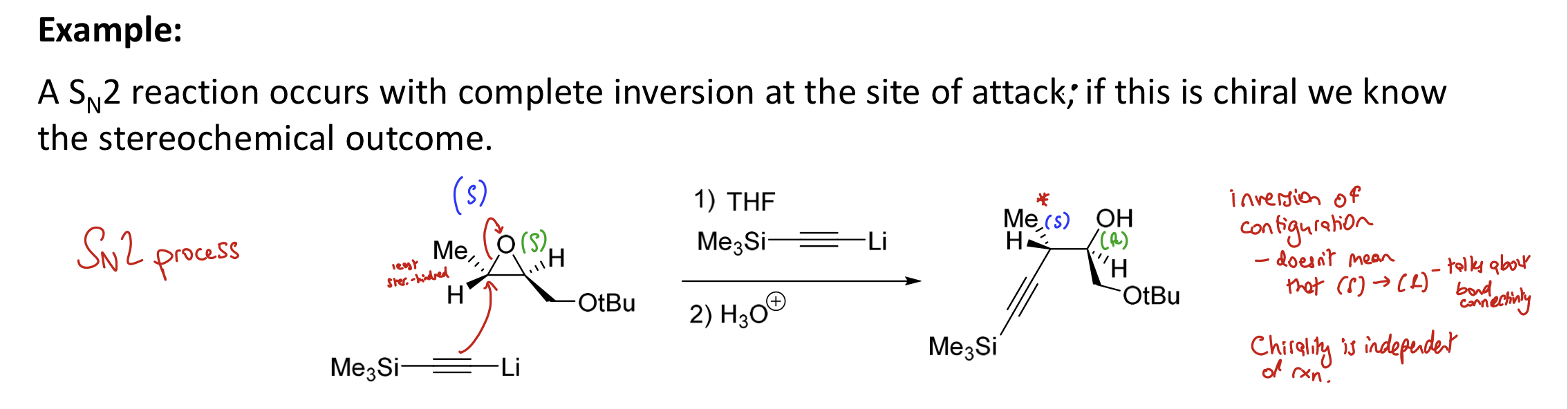
Is Sₙ2 stereoselective or stereospecific?
Stereospecific
complete inversion of stereochem if starting material is chiral
Diastereospecific
Reaction which forms only one diastereoisomer (control of relative stereochem and not absolute stereochem)
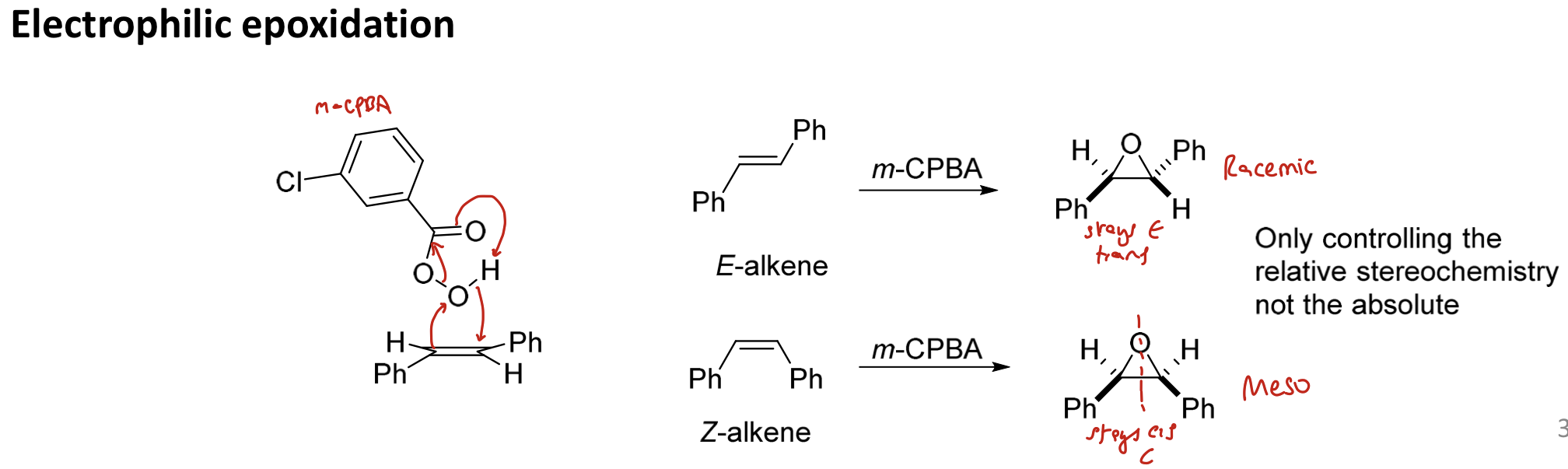
Give reagent(s) for electrophilic epoxidation. Is the reaction stereoselective or stereospecific?
m-CPBA
diastereospecific
Is hydroboration stereospecific or stereoselective? Why?
Diastereospecific
HBR₂ will always add H to the more substituted end of the alkene and BR₂ to the less substituted end
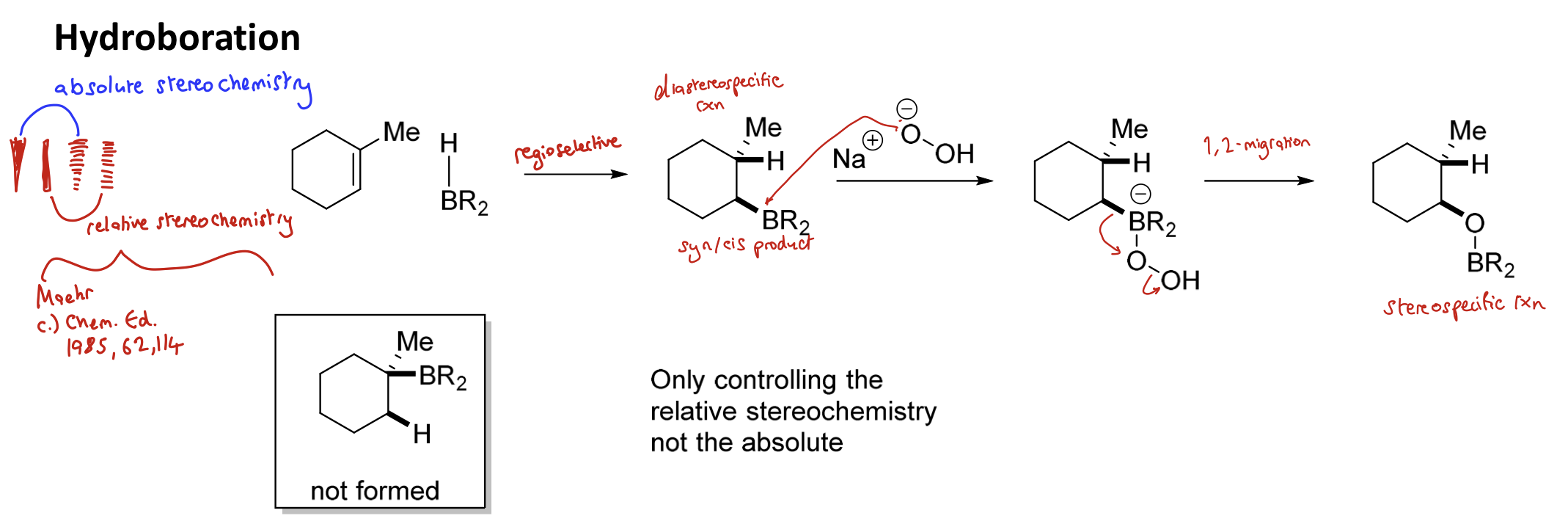
What do the filled square and dashed square signify in comparison to the filled triangle and dashed triangle in relation to stereochemistry?
The square signifies relative stereochemistry and the triangle signifies absolute stereochemistry
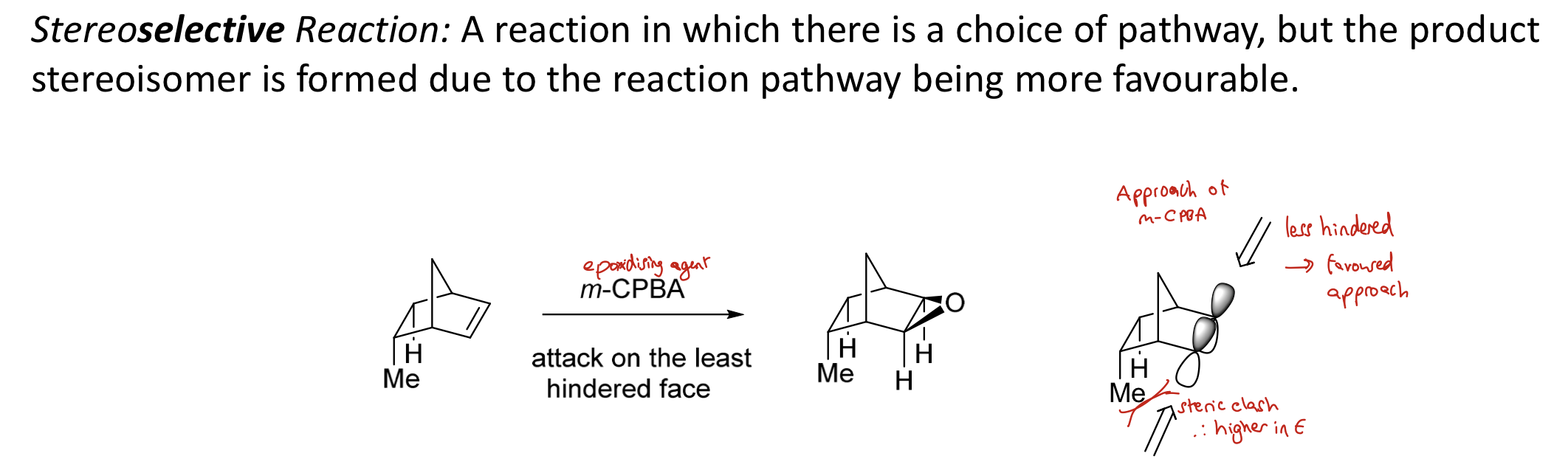
What is a stereoselective reaction? Give an example.
A reaction in which there is a choice of pathway but the product stereoisomer is formed due to the reaction pathway being more favourable
e.g. epoxidising agent attacking on the least hindered face
How do you calculate enantiomeric excess?
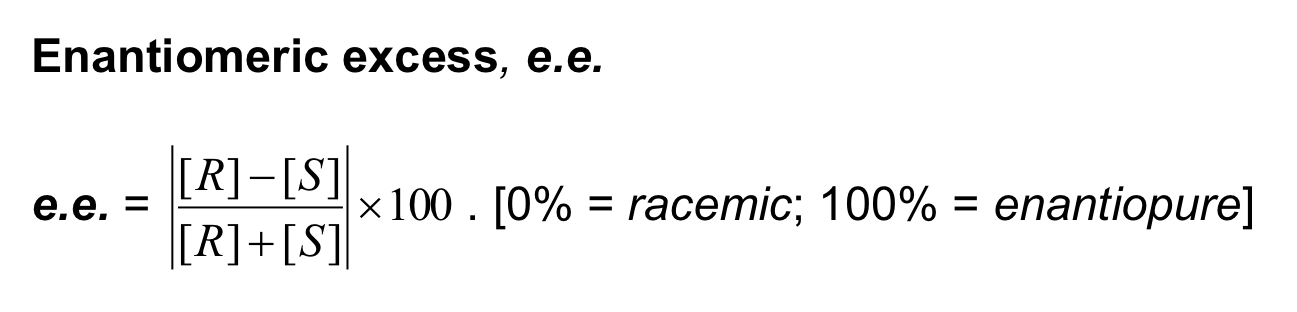
What is enantiomeric excess?
Mixtures in between optically pure and racemic are described by their optical purity or enantiomeric excess or enantiomers (er)
expressed as % enantiomeric excess (%ee).

What is diastereomeric excess? How do you calculate it?
A measure of the selectivity of a diastereoselective reaction.
0% = no selectivity, 100% = fully selective
d.e. = ([major] - [minor] / [major] + [minor]) x 100
e.g. major = 76%, minor = 24%
d.e. ( 76 - 24 / 76 + 24 ) x 100 = 52 %
![<p><span>A measure of the selectivity of a diastereoselective reaction. </span></p><p><span>0% = no selectivity, 100% = fully selective</span></p><p><strong>d.e. = ([major] - [minor] / [major] + [minor]) x 100</strong></p><p>e.g. major = 76%, minor = 24%</p><p>d.e. ( 76 - 24 / 76 + 24 ) x 100 = 52 %</p>](https://assets.knowt.com/user-attachments/cdca7694-9889-4cb9-a974-d9e2275e1cb9.png)
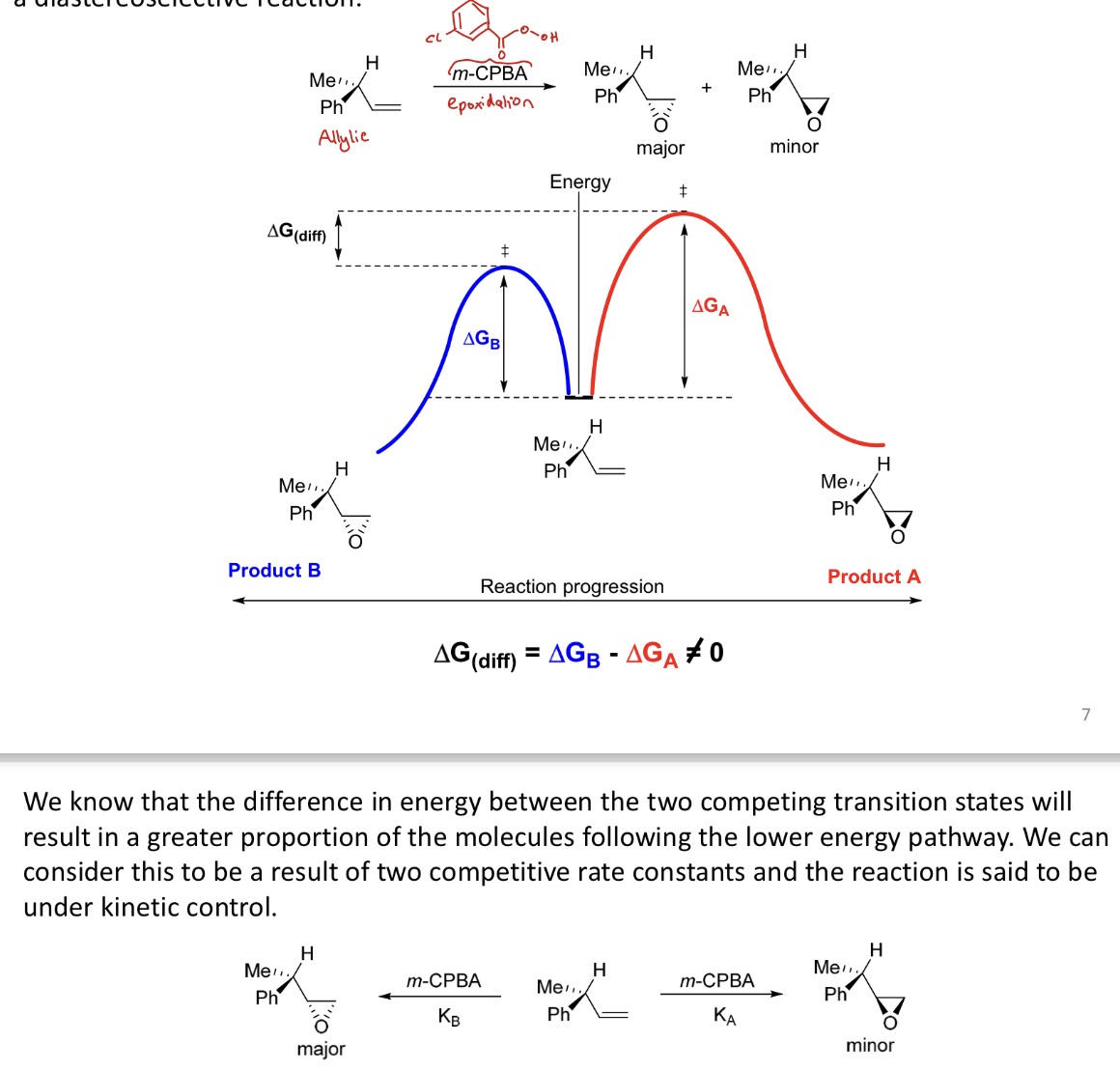
How does diastereoselectivity arise with a reaction under kinetic control?
The transition states are diastereoisomeric so have different energies. The major product is derived from the one with lowest energy.
Why do we see diastereoselectivity in the addition of a hydride to a ketone?
Hydride + ketone is irreversible and is kinetic,
ratio of the products (diastereoisomers) is determined by the relative rates of the facial additions
Syn product approached from underside → less steric clash (B) → kinetic product
Anti product approached upper side (more sterically hindered) via higher energy TS → TD product
They are different because the TS are diastereoisomeric and have unequal energies
Steric factors determine the TS energy and then the product distribution
What feature is an inherent control of stereoselectivity?
Substrate with chiral centre
→ it is possible to make a single isomer product from a diastereoselective reaction
Diff in E between the 2 competing TS⁺s → more molecules following lower E pathway
this is the result of two competitive rate constants → rxn under kinetic control
What is an allylic system?
A system with a chiral centre α to an alkene
How can the selectivity of electrophilic addition to alkenes be rationalised?
Combo of allylic strain and molecular orbital overlap → Houk’s rule
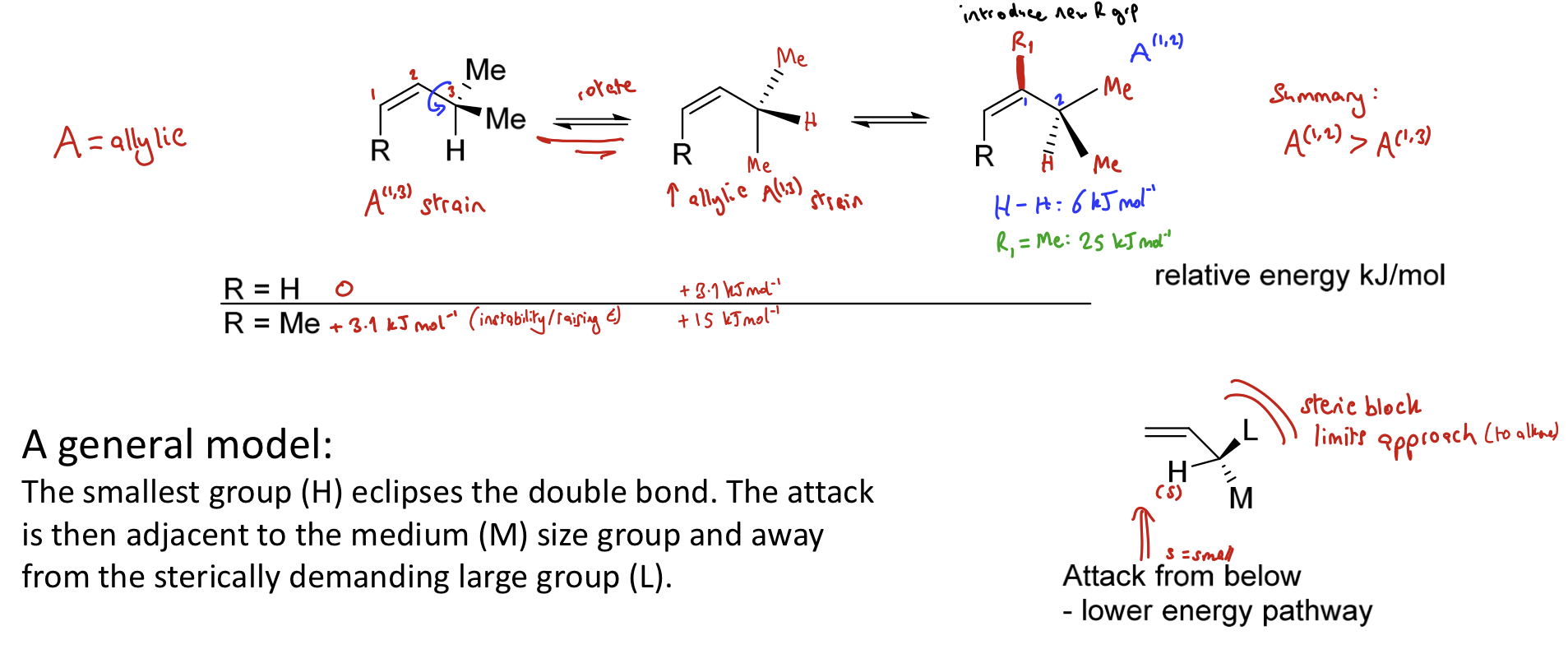
What is Houk’s rule/Allylic Strain Model?
Used to model selectivity of electrophilic addition to certain alkenes.
Smallest group eclipses the double bond → minimises A(1,3) strain.
Attack then comes from the face with smaller substituent.
Which strain is more dominant: A(1,2) or A(1,3) strain?
A(1,2)
Draw general model of electrophilic attack of an alkene
The smallest group (H) eclipses the double bond. The attack is then adjacent to the medium sized group and away from the sterically demanding large group
Steric block limits approach so the attack from below is favoured (the lowest energy pathway)
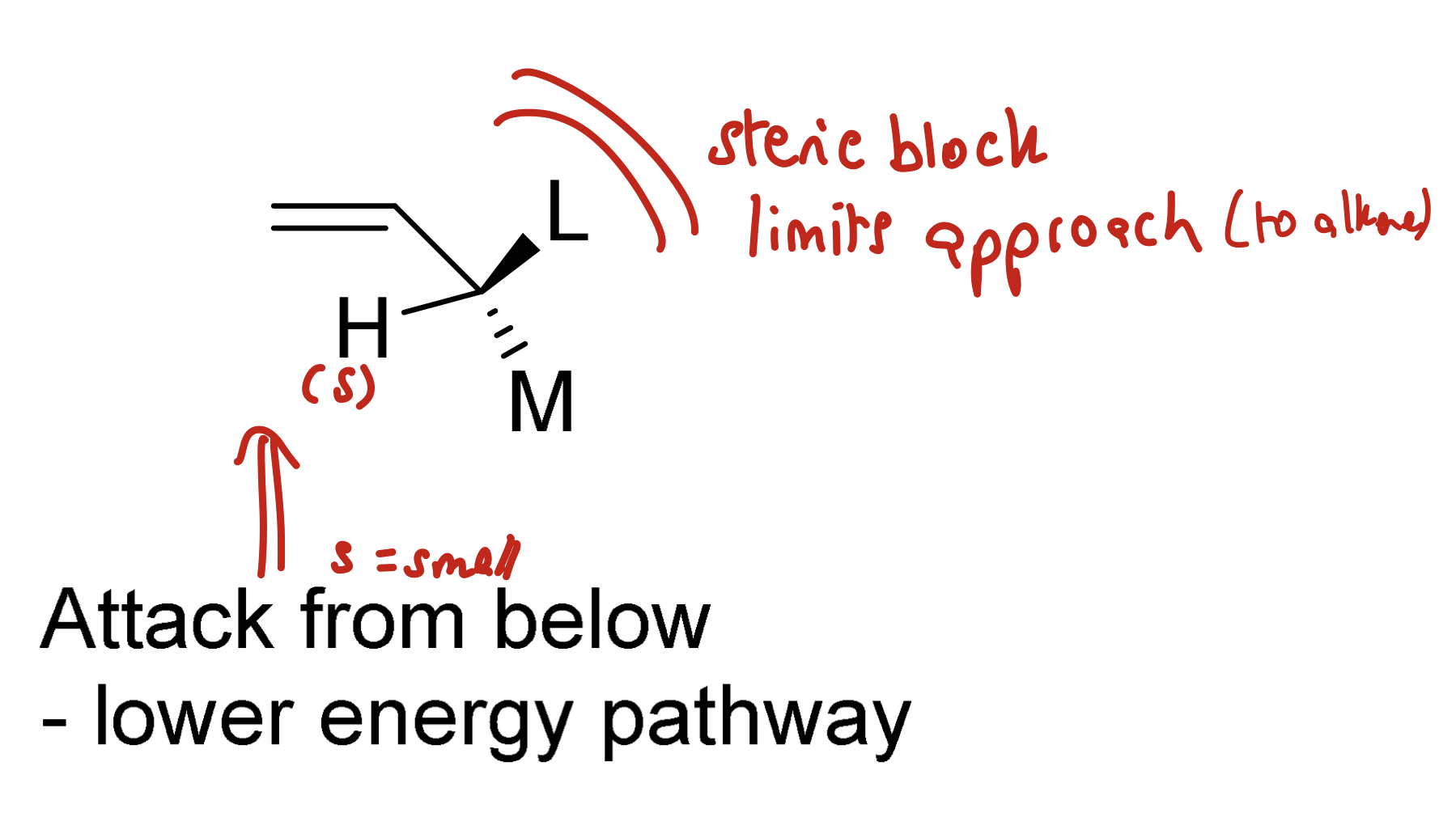
Which clash has the most energy?
Me-Me (MOST)
Me-H
H-H (least)
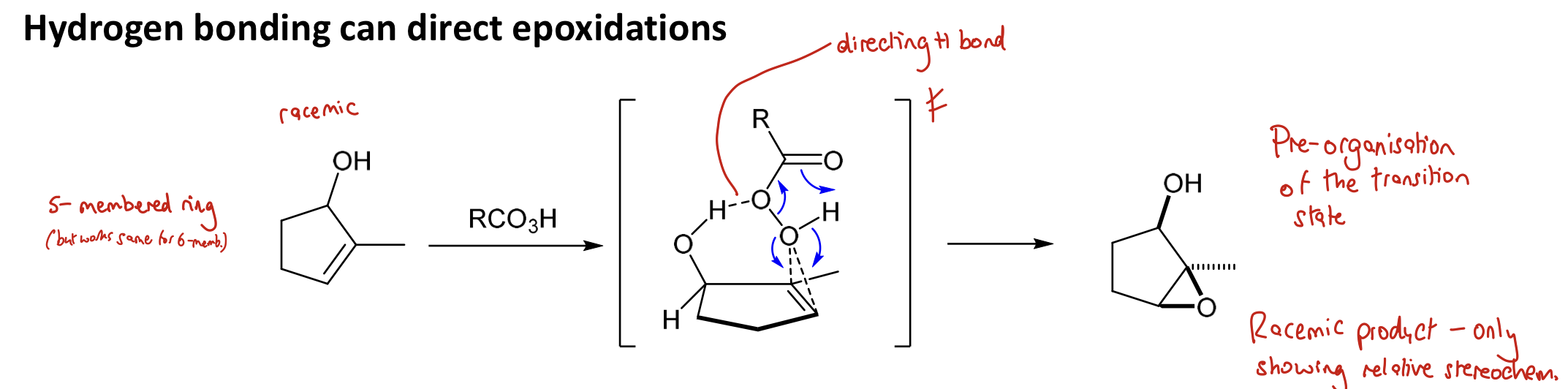
How can H bonding direct epoxidations?
(there is pre-organisation of the TS): A H bond from something such as an alcohol to the peracid will cause epoxidation on the same face as the bonding group
When there are multiple options for H bonding to occur, think about what will happen if a large group goes up or down and do whatever option which minimises sterics
→ syn product
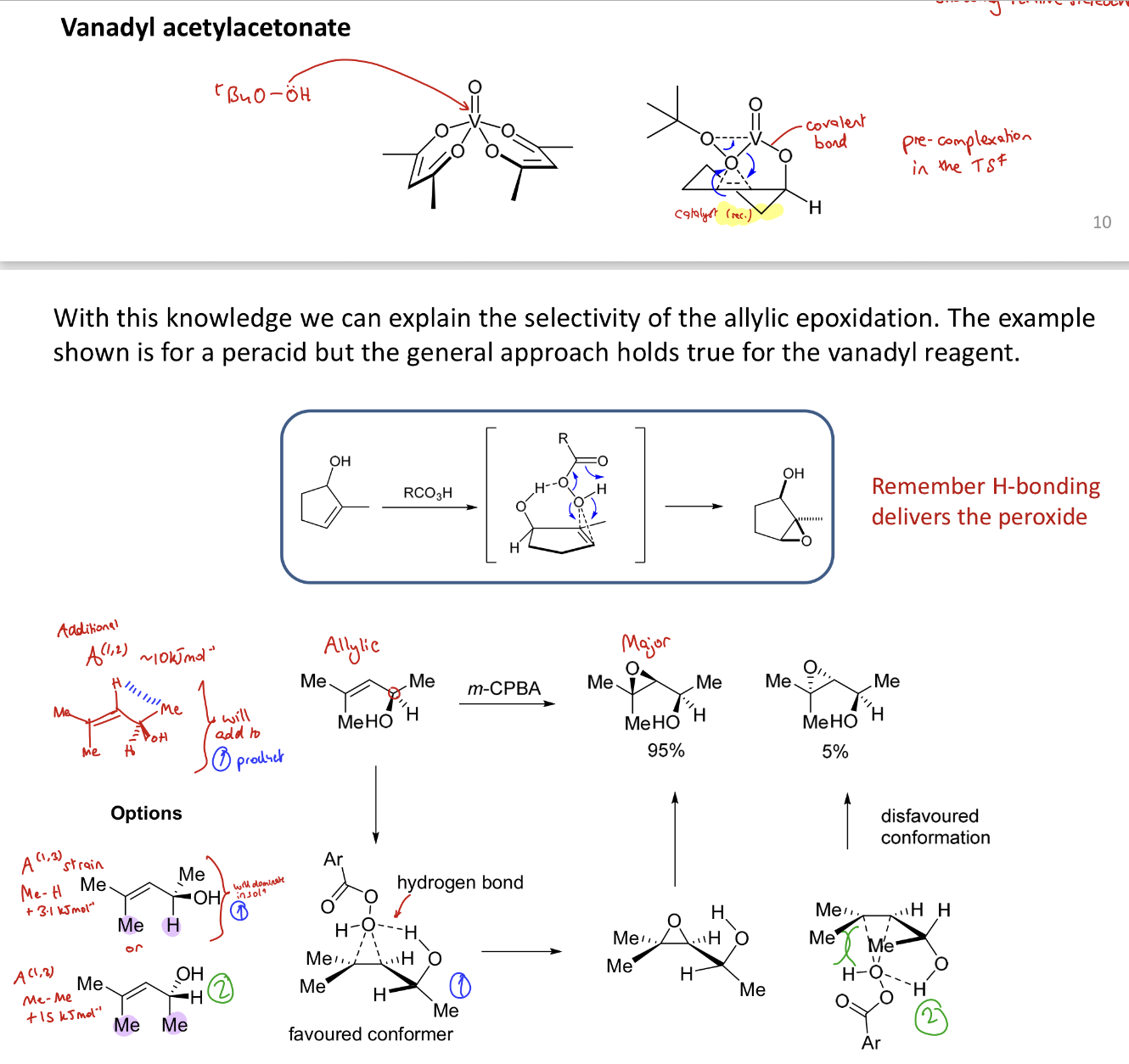
How can vanadyl acetylacetonate (tBuO₂H, VO(acac)₂ )direct the direction of epoxidation?
Similarly to a peracid, covalent bonding causes pre complexation to an O group and the epoxide is formed on the same face as the O containing group
→ syn product
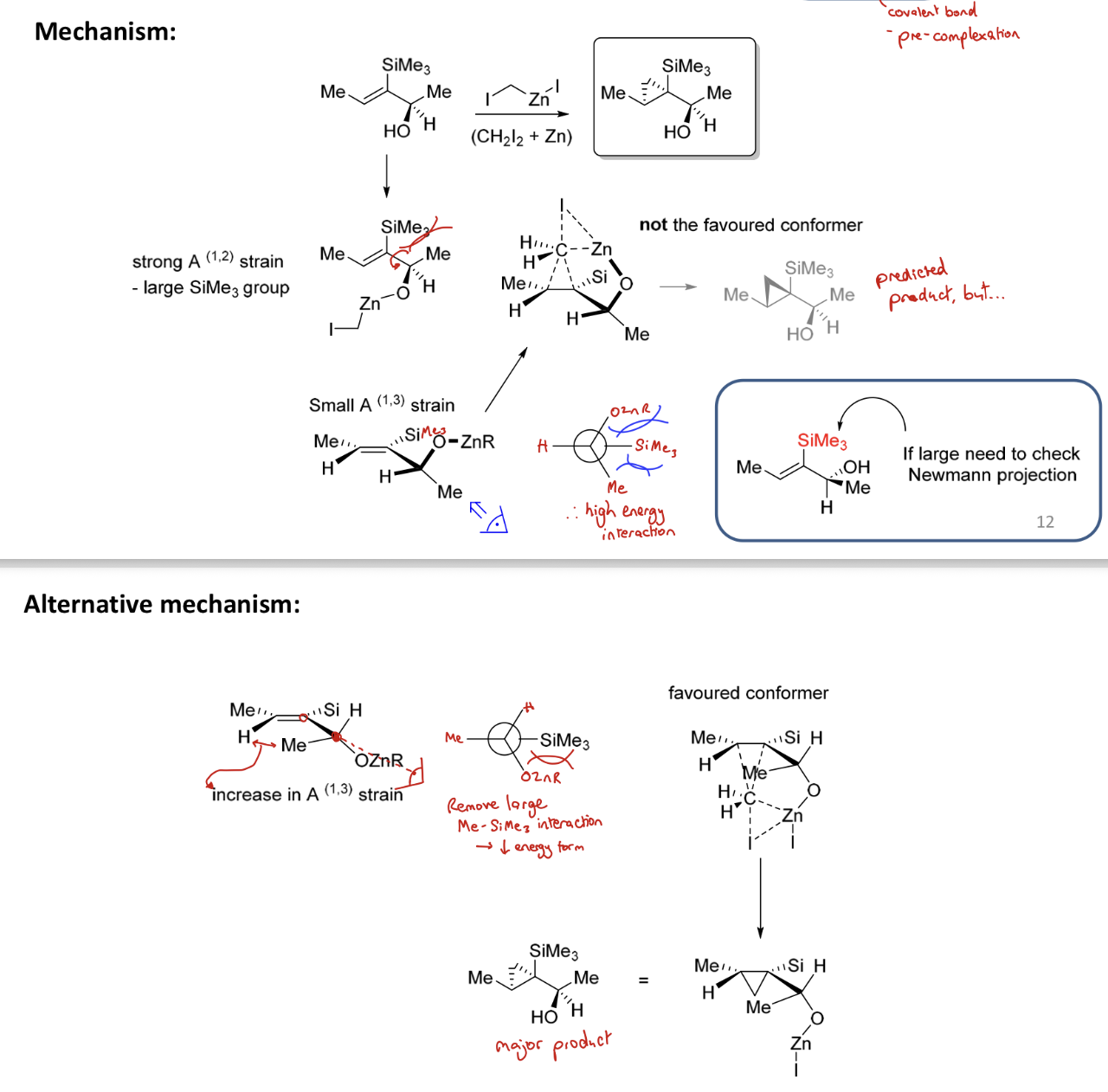
How do you achieve cyclopropanation of an alkene with an α OH group? How is the reagent formed?
Using Simmons-Smith reagent: IZnCH₂I - organozinc
IZnCH₂I formed from CH₂I₂ and Zn
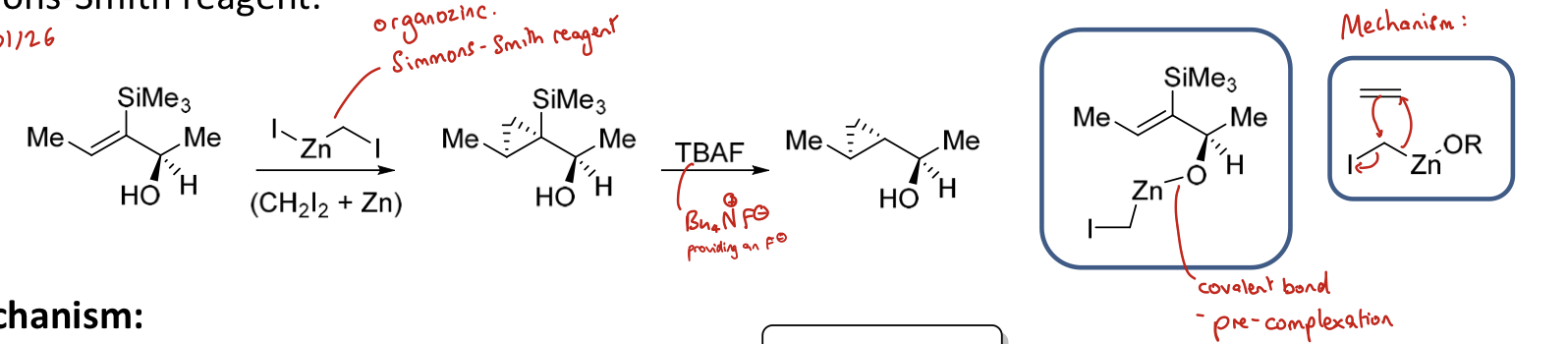
What is the mechanism for cyclopropanation of an alkene using IZnCH₂I (and TBAF)?
The Simmons-Smith reagent complexes to the alcohol group to form OZnCH2I before a pericyclic Sn2 reaction with the alkene (opening up double bond)
TBAF removes SiMe3
3 large groups next to each other can cause large clash so Houk model can break down and H doesn't eclipse
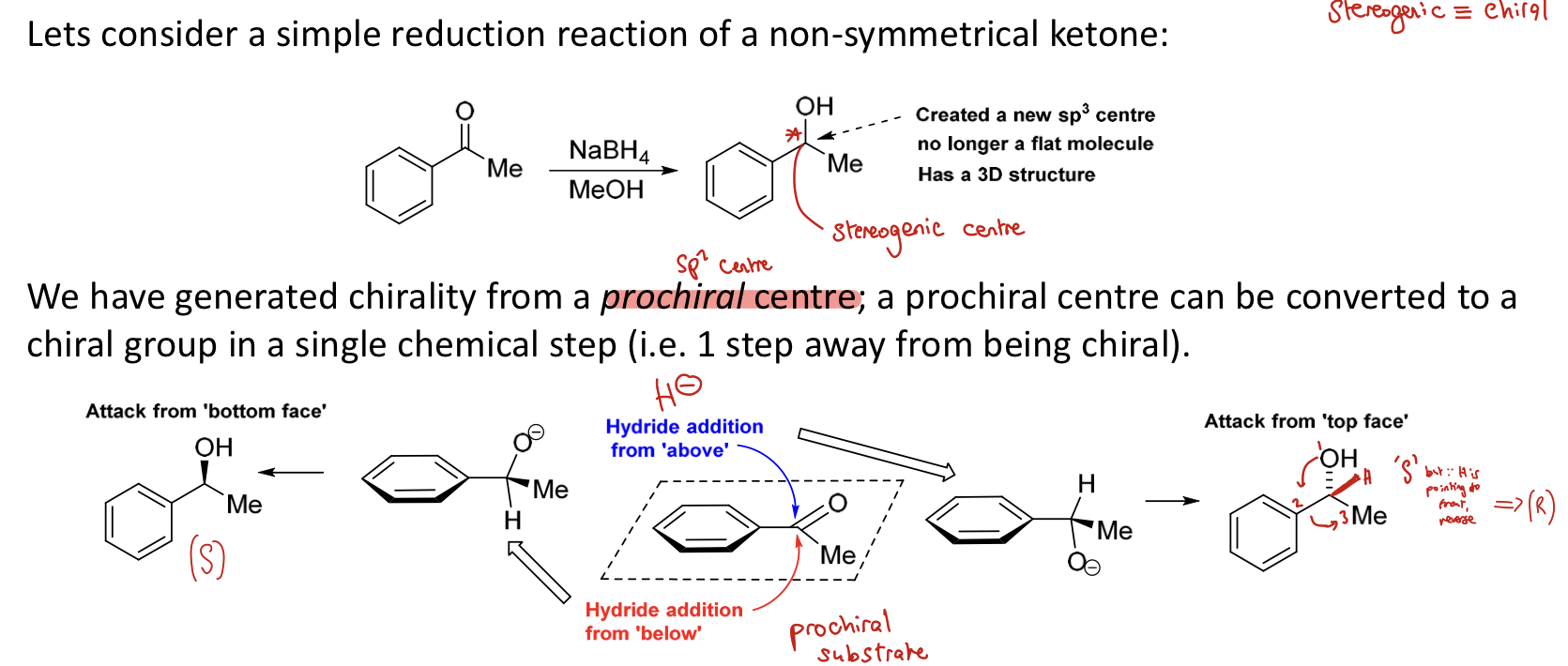
What is a prochiral centre?
sp2 centre that can be converted into a chiral group in a single chemical step (1 step away from being chiral)
e.g. 1 bond forming or 1 transformation
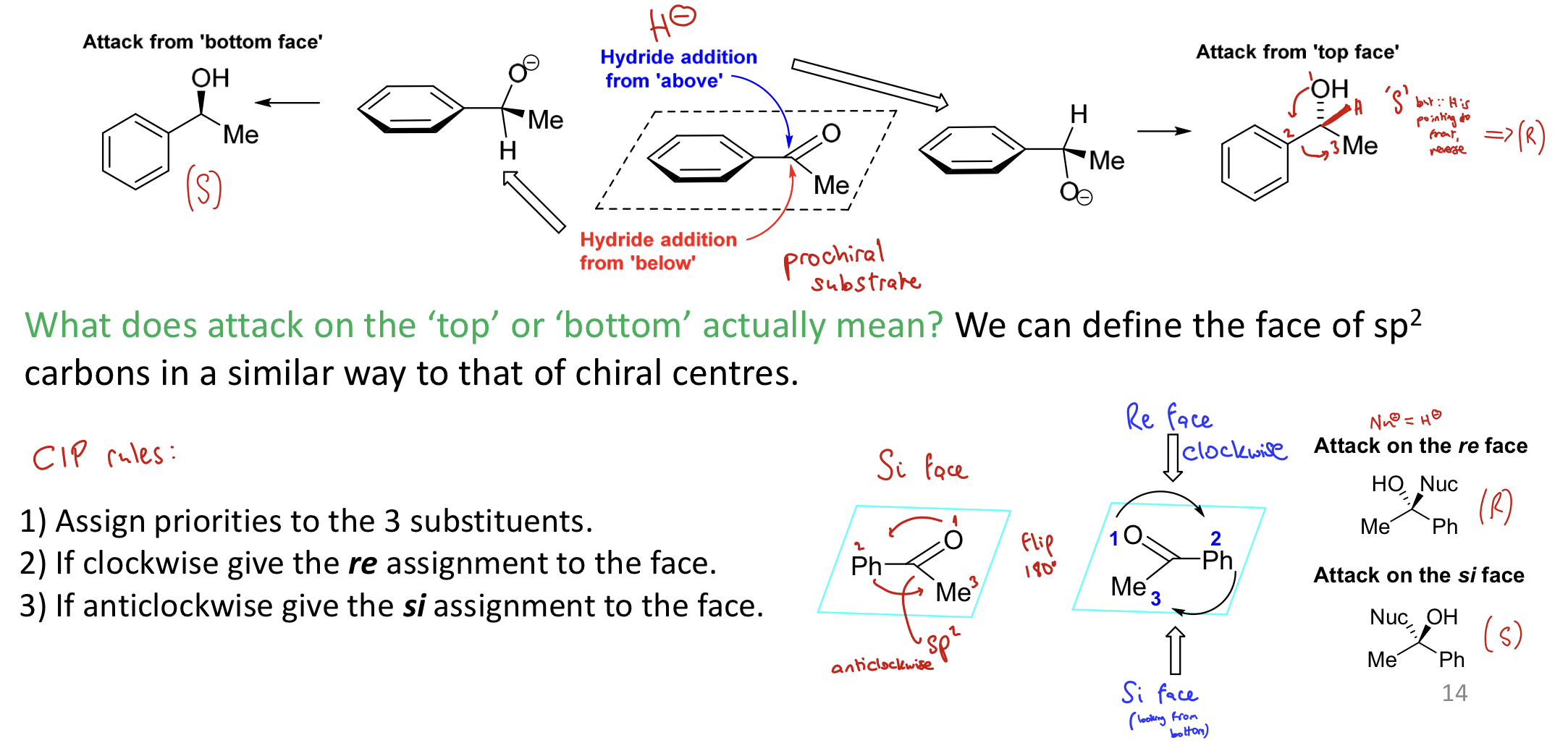
How to define Re and Si faces
Put the molecule in the plane of the board
Assign priority to the groups
Clockwise = re
Anti-clockwise = si
Attack on re gives R and attack on si gives S
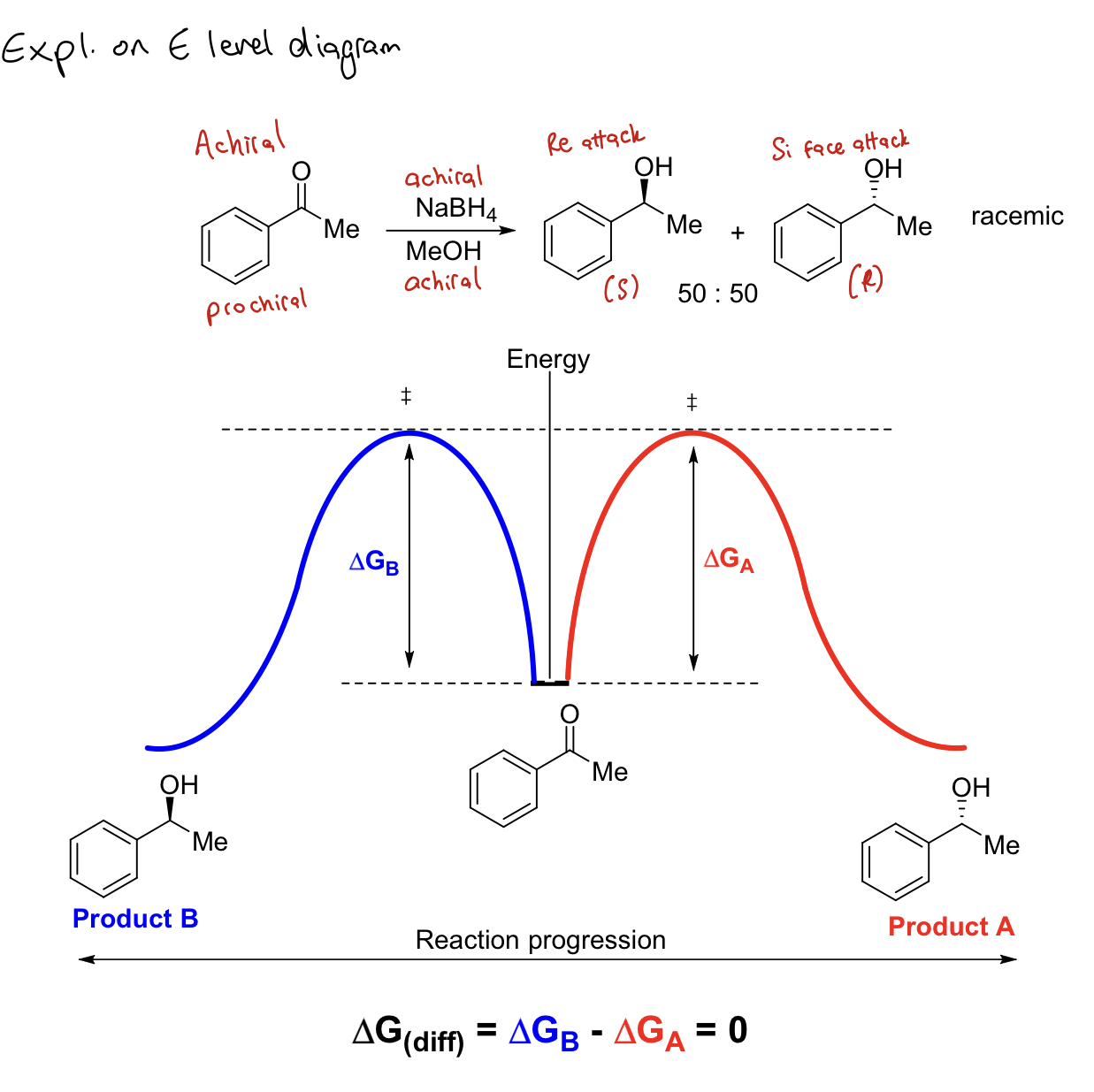
Can optically active products be produced using achiral (or racemic) starting materials and reagents in an achiral solvent? Draw E level diagram to explain your answer.
No! Products will also be achiral or racemic.
The energies of the 2 pathways are equal so there is no favoured pathway
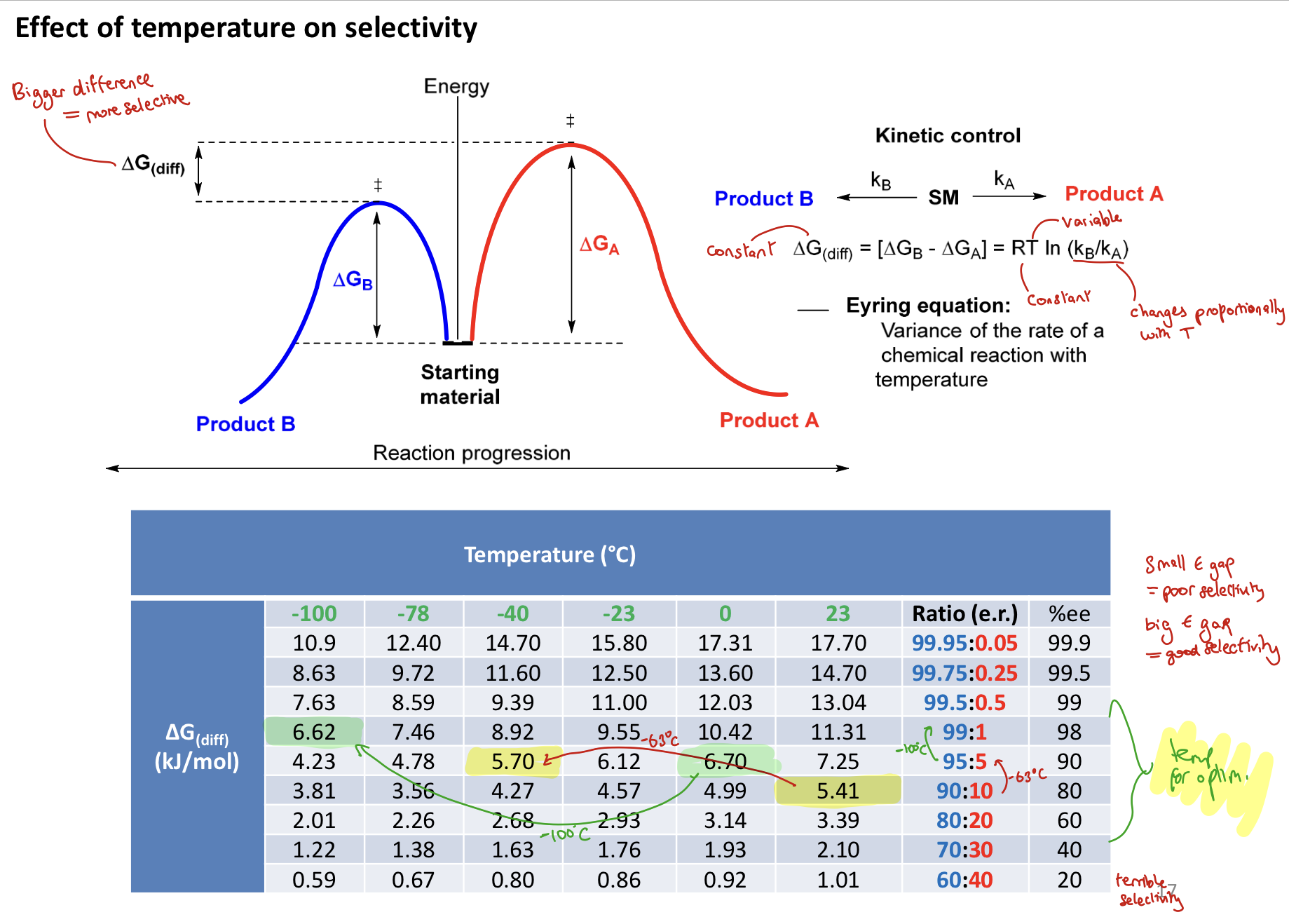
How does temperature impact the selectivity/enantiomeric excess?
Depends on the ∆G(diff) between the 2 competing pathways
small E gap = poor selectivity → low e.e. (nearer racemic)
large E gap = good selectivity → high e.e. (nearer enantiopure)
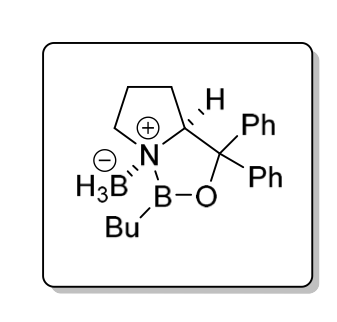
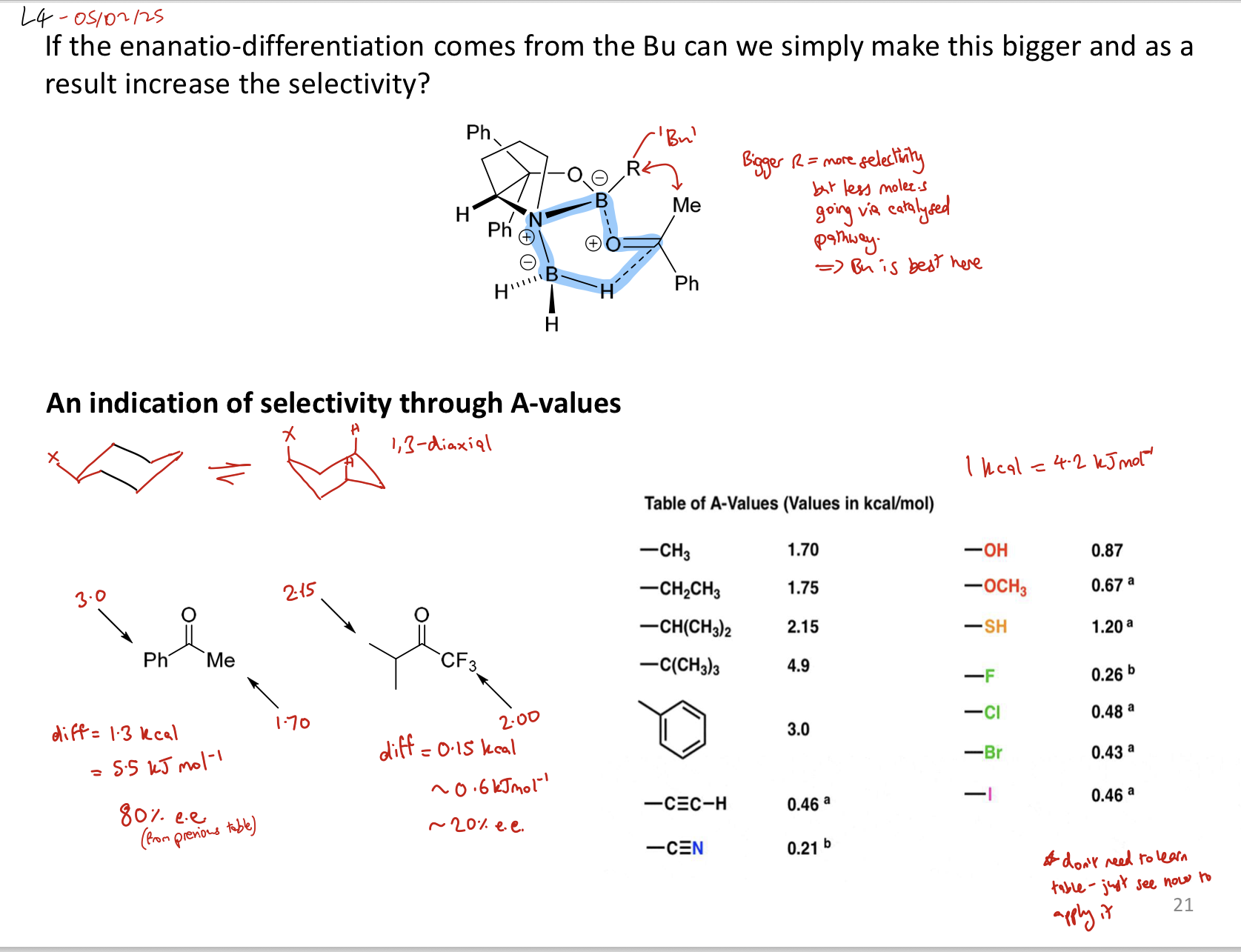
What’s this:
CBS catalyst - selectively reduces a carbonyl to an R alcohol by attacking si face
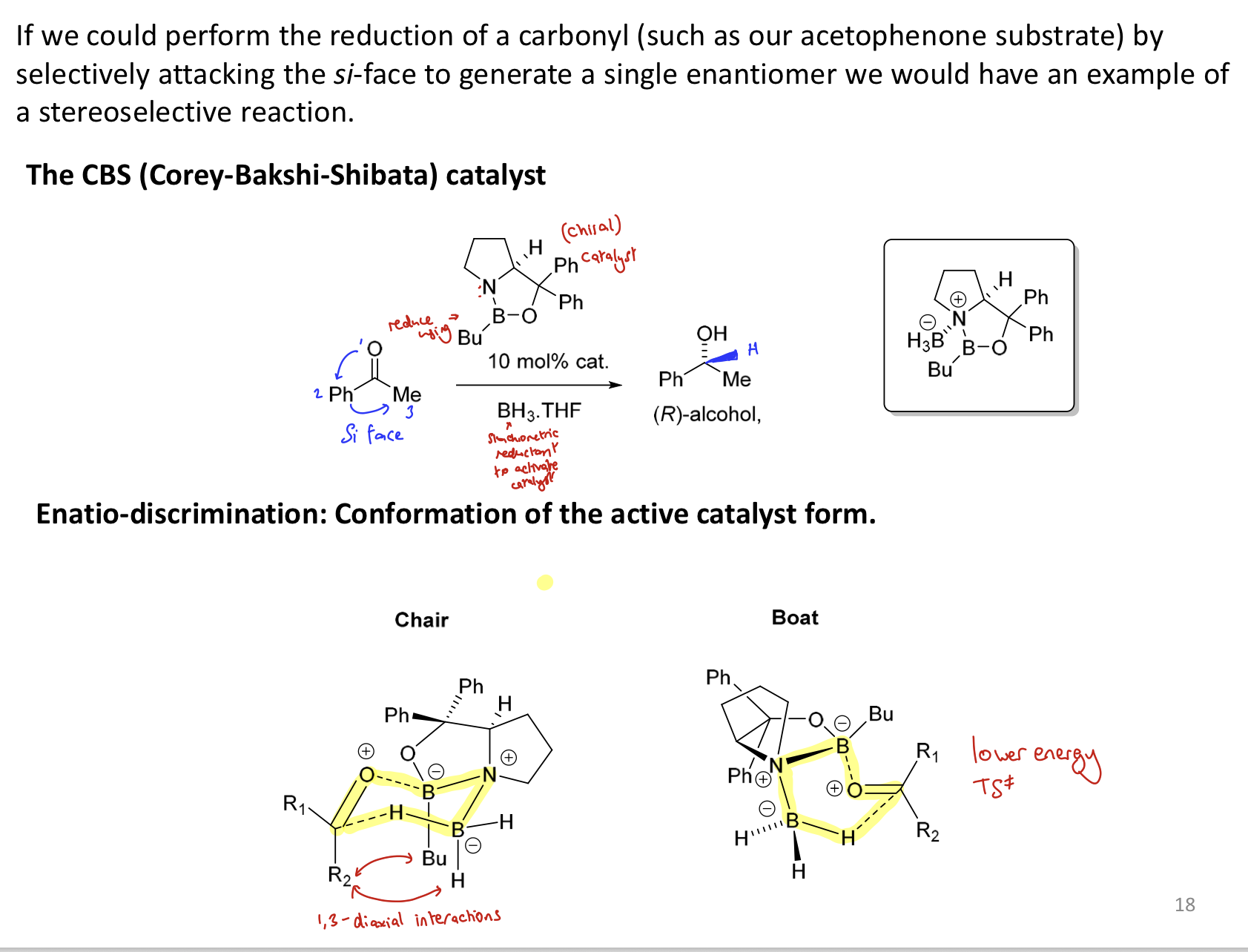
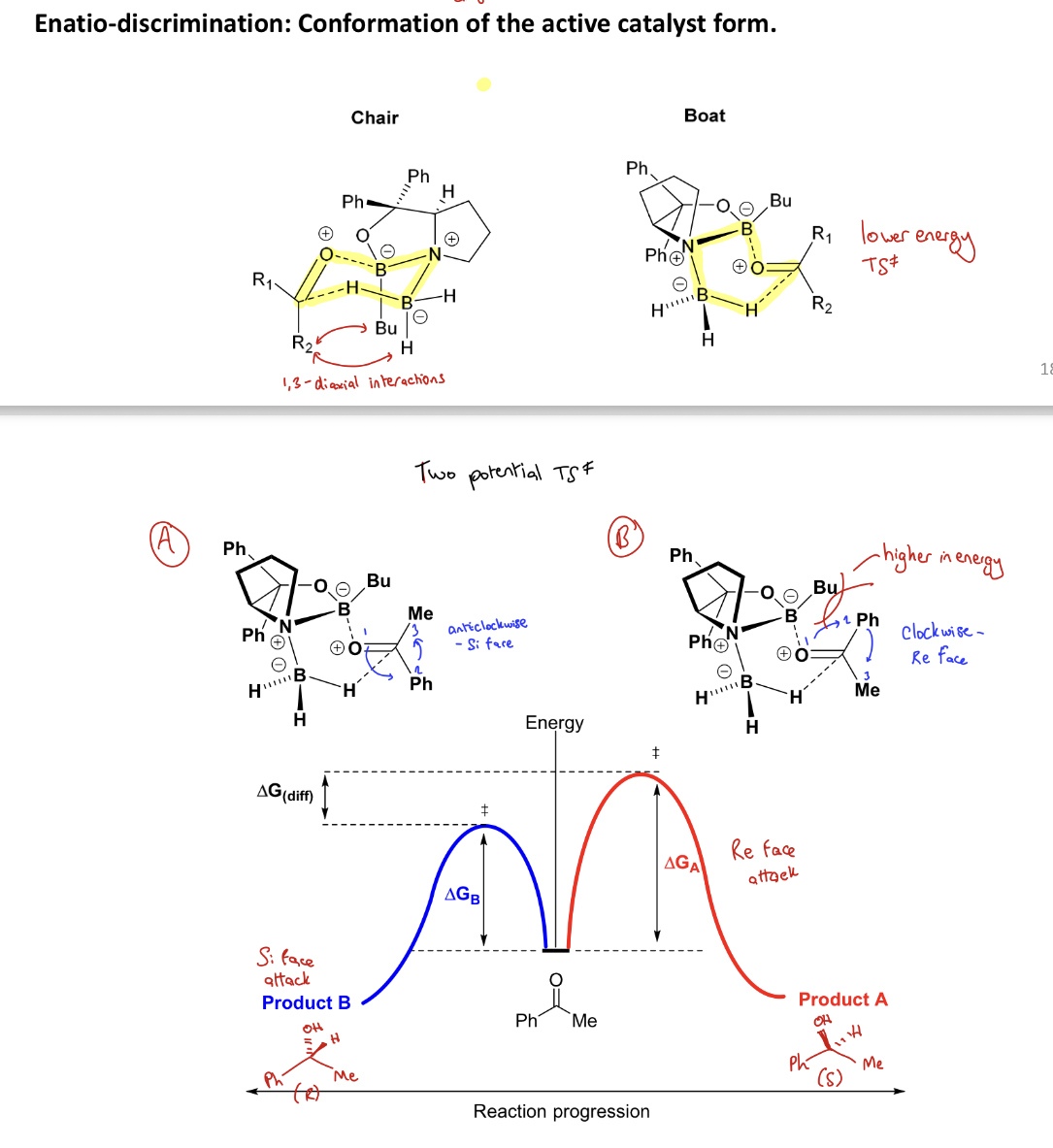
How does CBS catalyst give the R alcohol selectively?
It will complex in a boat transition state (B to O of ketone and H to C of ketone) to avoid the 1,3 diaxial interaction between a ketone R group and the Bu. CBS complexes to the si face as there is less interaction between the bigger R group and Bu
i.e. CBS introduces steric clash, which makes the pathway that has steric clash in it unfavourable
= stereoselective rxn
What is the relationship between temperature and ee if a catalyst is present?
↑ catalyst → ↑ yield → ↑ e.e and selectivity because more likely to go via catalytic rxn than background rxn
If temp ↑ but catalyst amount stays same then, yield is good but enantio selectivity ↓ due to ↑ competition between catalytic and background rxns
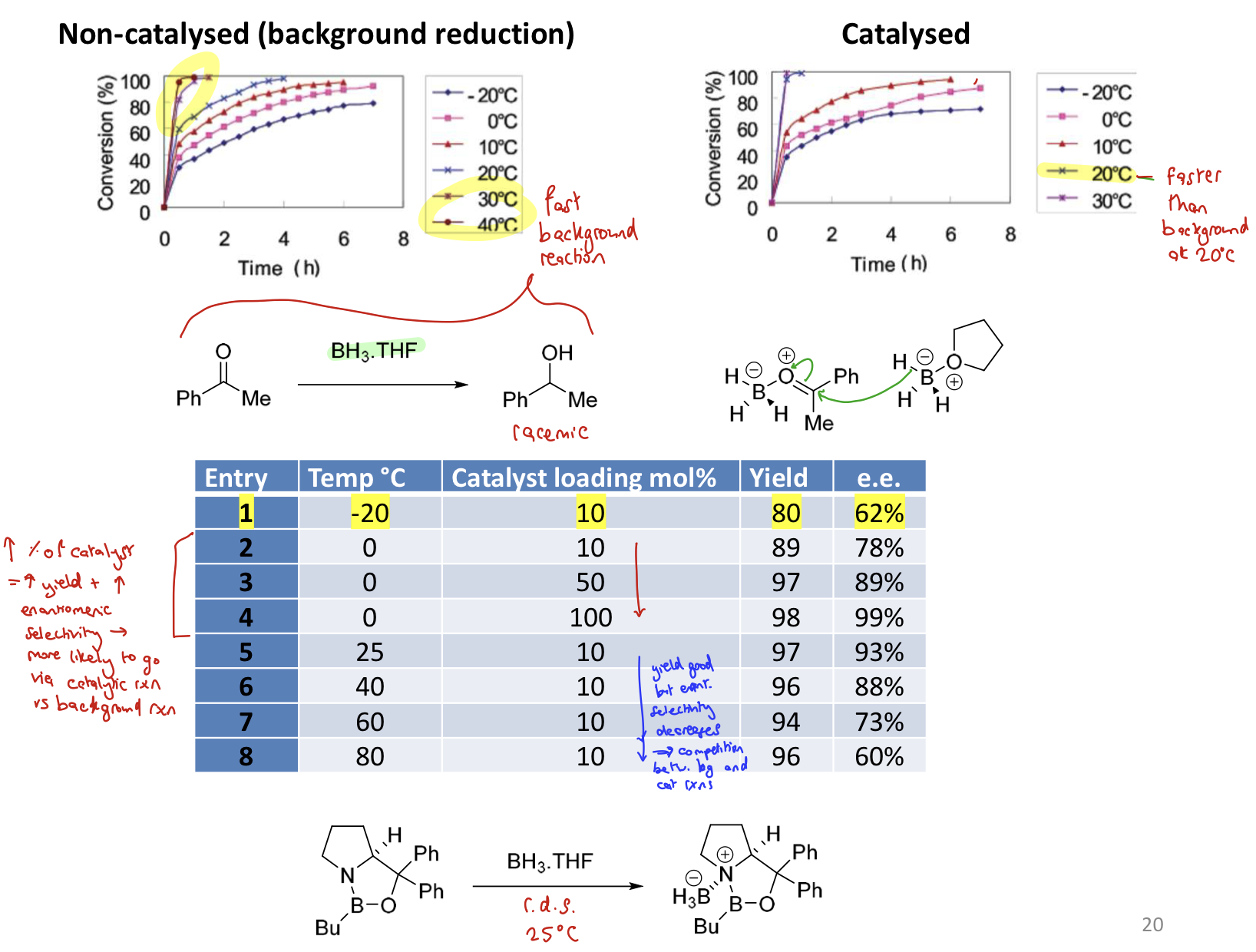
How can selectivity of a ketone reduction be determined through A values?
The larger the difference in energy between the two R groups, the higher the e.e.