Lab 5: Reduction of Aldehydes and Ketones
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
is lithium aluminum hydride gentle or strong? and what does this mean?
lithium aluminum hydride is gentle. it will not reduce esters, amides or carboxylic acid under normal conditions
how does sodium borohydride react with protic solvent?
it will yield hydrogen gas at room temp because boron and H share comparable electronegativities
what are common common solvents to use with sodium borohydride
methanol and ethanol
why do we microwave in this reaction
it increases the reaction rate
techniques used
microwave heating, liquid-liquid extraction, rotary evaporation, IR, and HNMR
table of reagents
Acetophenone (structure, MW, density, BP)
Propiophenone (structure, MW, density, BP)
Isobutyrophenone (structure, MW, density, BP)
4-Methoxybenzaldehyde (structure, MW, density, BP)
P-tolualdehyde (structure, MW, density, BP)
4-Fluoroacetephenone (structure, MW, density, BP)
4-Ethoxybenzaldehyde (structure, MW, density, BP)
4-Ethylbenzaldehyde (structure, MW, density, BP)
4-Ethylacetophenone (structure, MW, density, BP)
Sodium borohydride (structure, MW)
Ethanol (structure, MW, density, BP)
Dichloromethane (structure, BP)
Sodium sulfate (structure
broadly explain the procedure
take sodium borohydride and place into a microwave with ethanol
add unknown aldehyde or ketone and seal vessel
once microwave is done, put mixture into sp funnel
add water to sep funnel then add two portions of ethyl acetate (aka you will keep adding ethyl acetate to your aqueous)
combine the organic layers and wash the organic layer with water four times (aka keep adding water to the organic)
add magnesium sulfate to organic solution to dry and then filter
rotovap to get remaining ethyl acetate out
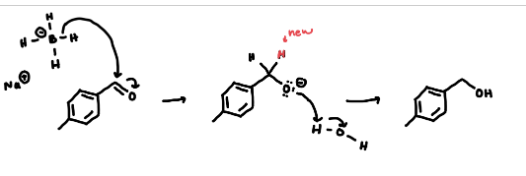
how do the nucleophile and electrophile interact in this interaction?
the nucleophile will donate electron pair to electrophilic carbon, forming a new carbon nucleophile sigma bond to push the pi electrons onto the oxygen (single bond instead of double bond)
describe the geometric change in carbons
the carbonyl will rehybridize from sp2 (planar) to sp3 (tetrahedral)
what kind of alcohols does the reduction of aldehydes and ketones result in?
primary and secondary alcohols
why is sodium borohydride so much better to use?
sodium borohydride is safer, less reactive, and can be used with water and/or alcohol solvents
lithium aluminum hydride is more difficult to handle safely, will be more reactive, sensitive to water, protic solvents, and heat. it also can only be used with intert, non-protic solvents like ether
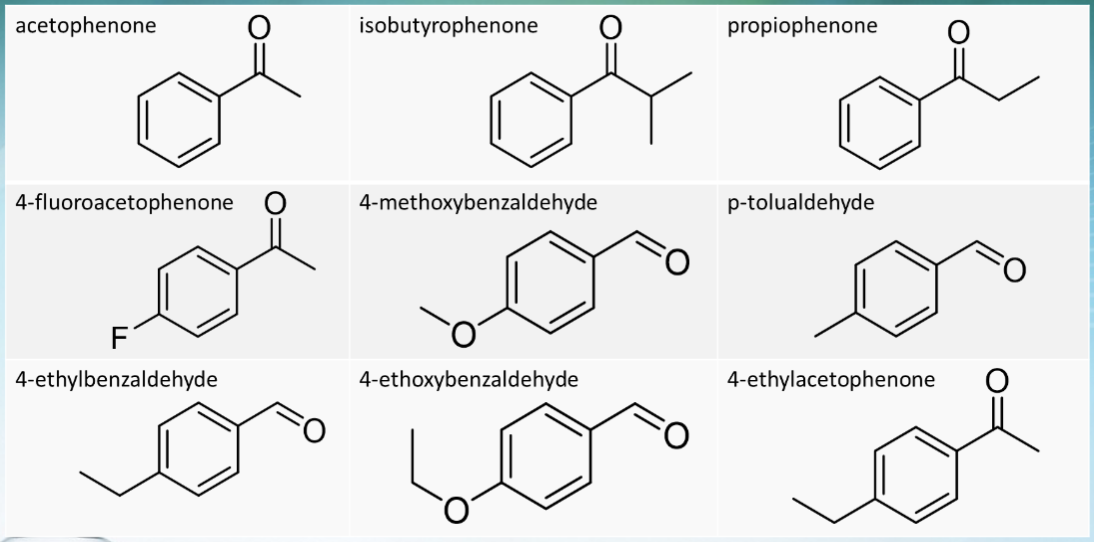
what are the 9 possible aldehydes and ketones used in this experiment?
see photo of acetophenone, isobutyrophenone, propiophenone, 4-fluoroacetophenone, 4-methoxybenzaldehyde, p-tolualdehyde, 4-ethylbenzaldehyde, 4-ethoxybenzaldehyde, and 4-ethylacetophenone
ethanol boiling point
78 ºC
ethyl acetate boiling point
77 ºC
sodium borohydride melting point
400 ºC
magnesium sulfate melting point
1,124ºC
name all SDS things
acutely toxic, chronically toxic, flammable, corrosive/caustic
what is the reducing agent
sodium borohydride
what solvent will you be using to do extraction
ethyl acetate
how will you be heating your mixture
microwave and microwave vessel (Teflon vessel)
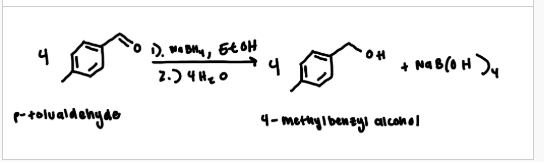
general balanced equation
1:4 ratio (1 NaBH4 and aldehyde/ketone)
describe the general reaction mechanism
B-H bond attacks carbon of carbonyl. pushes electrons to O. then the O is protonated with water
what was the limiting
unknown aldehyde/ketone
could you identify your unknown with your IR?
No, there were not any differences to help you determine your unknown but there were differences to show the reaction went to completion (change from carbonyl to OH). the only way the IR helped me was it helped me eliminate options that had distinct functional groups like Cl
The unknown aldehydes/ketones and sodium borohydride are all more soluble in acetone than ethanol. Would acetone therefore be a more suitable solvent for this experiment?
No because acetone is a ketone. sodium borohydride will want to react with it instead of the unknown (see reaction scheme attached)
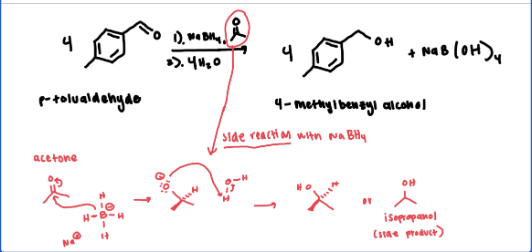
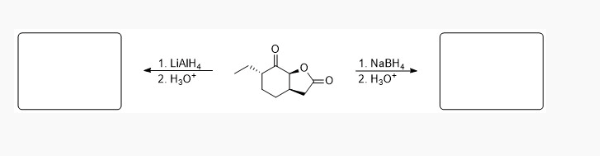
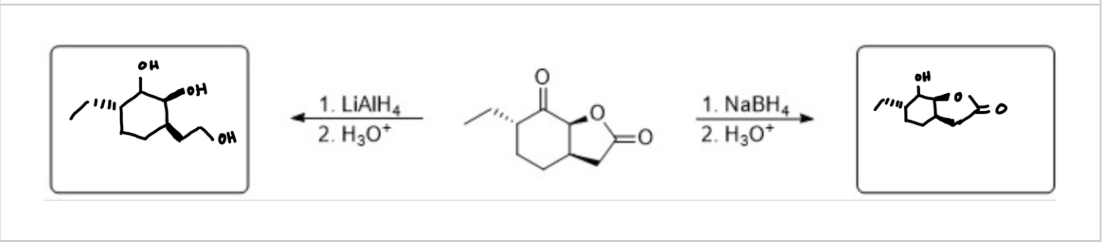
predict the major products
see photo (only difference is lithium alumnium hydride can react esters)
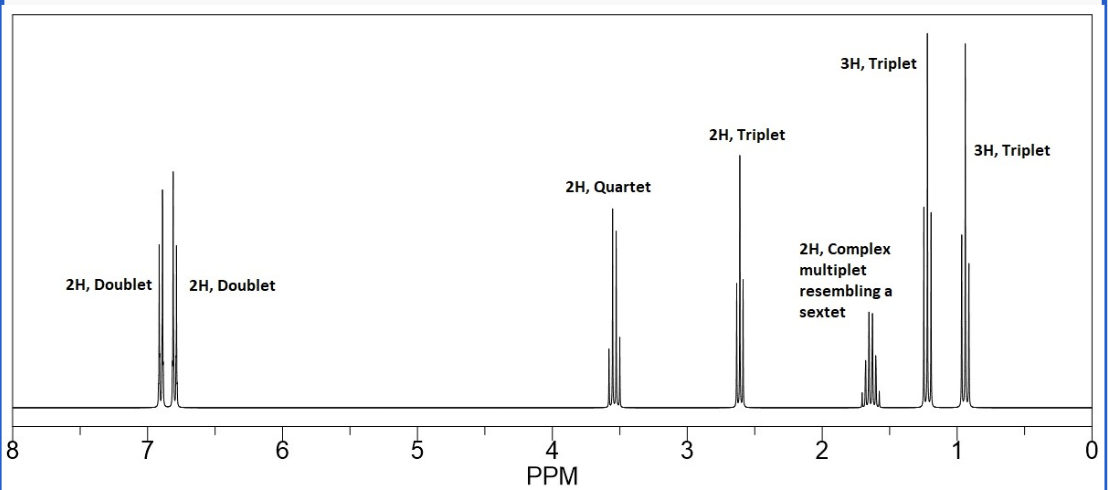
Provide the structure of the unknown ketone/aldehyde starting material that would produce the following product HNMR spectrum:
see photo
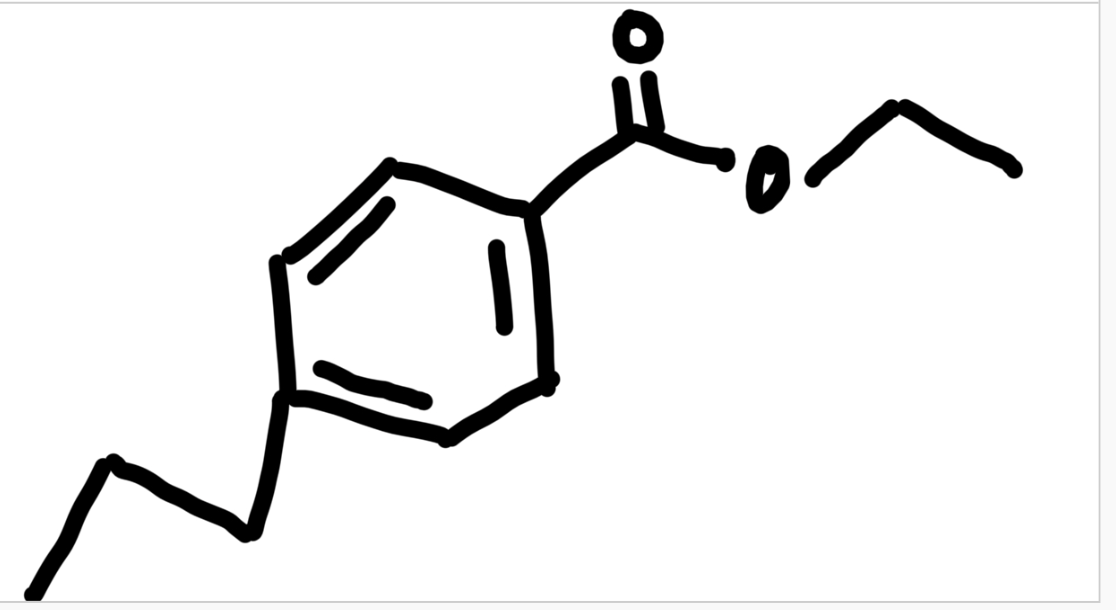
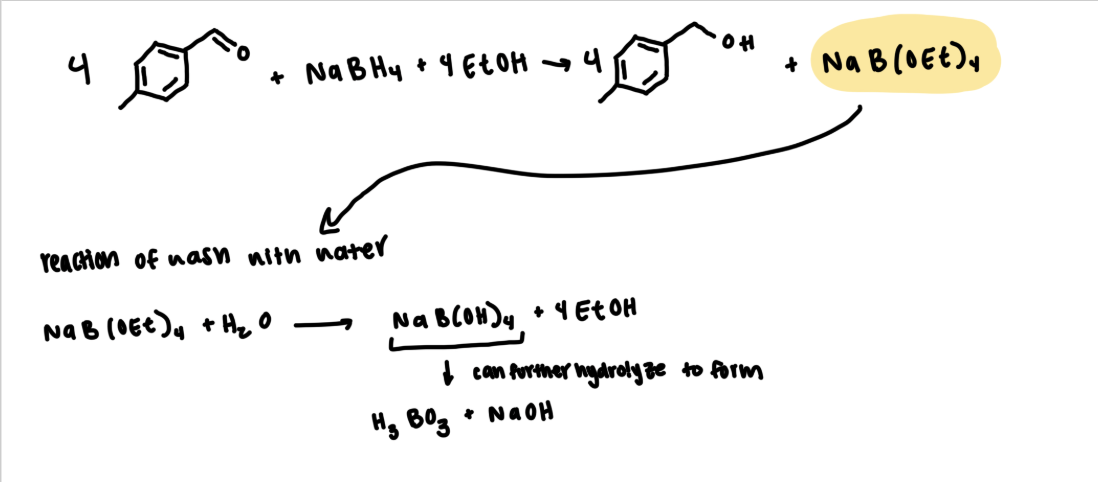
Students are asked to “wash” their reaction solution, as well as, the organic extraction layers with deionized water. What are the most likely impurities that are removed during these washing steps?
The purpose of the "washing" step is to remove any water-soluble byproducts from our product-containing organic layer. Some of the water-soluble impurities are sodium borate, excess sodium borohydride, sodium hydroxide, and ethanol.