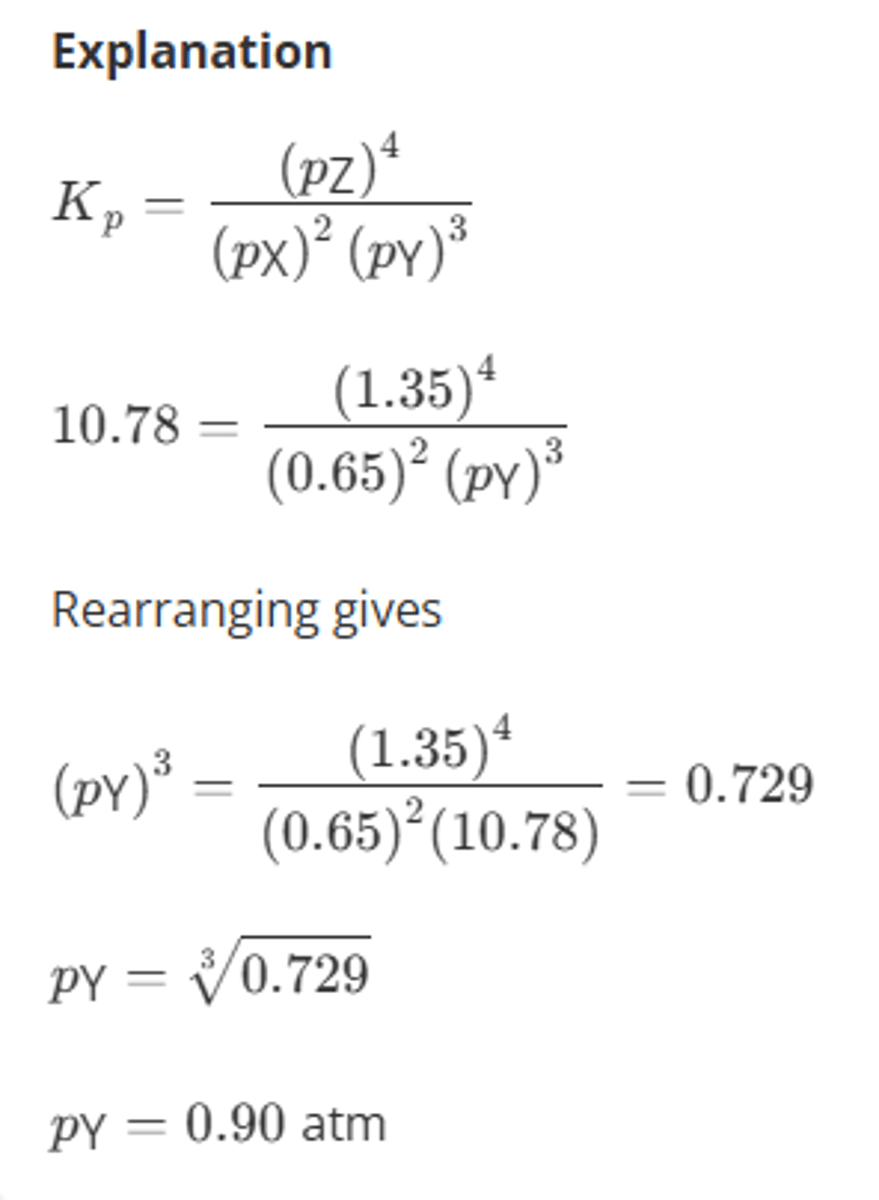10. Equilibrium constant Kp for homogenous systems
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
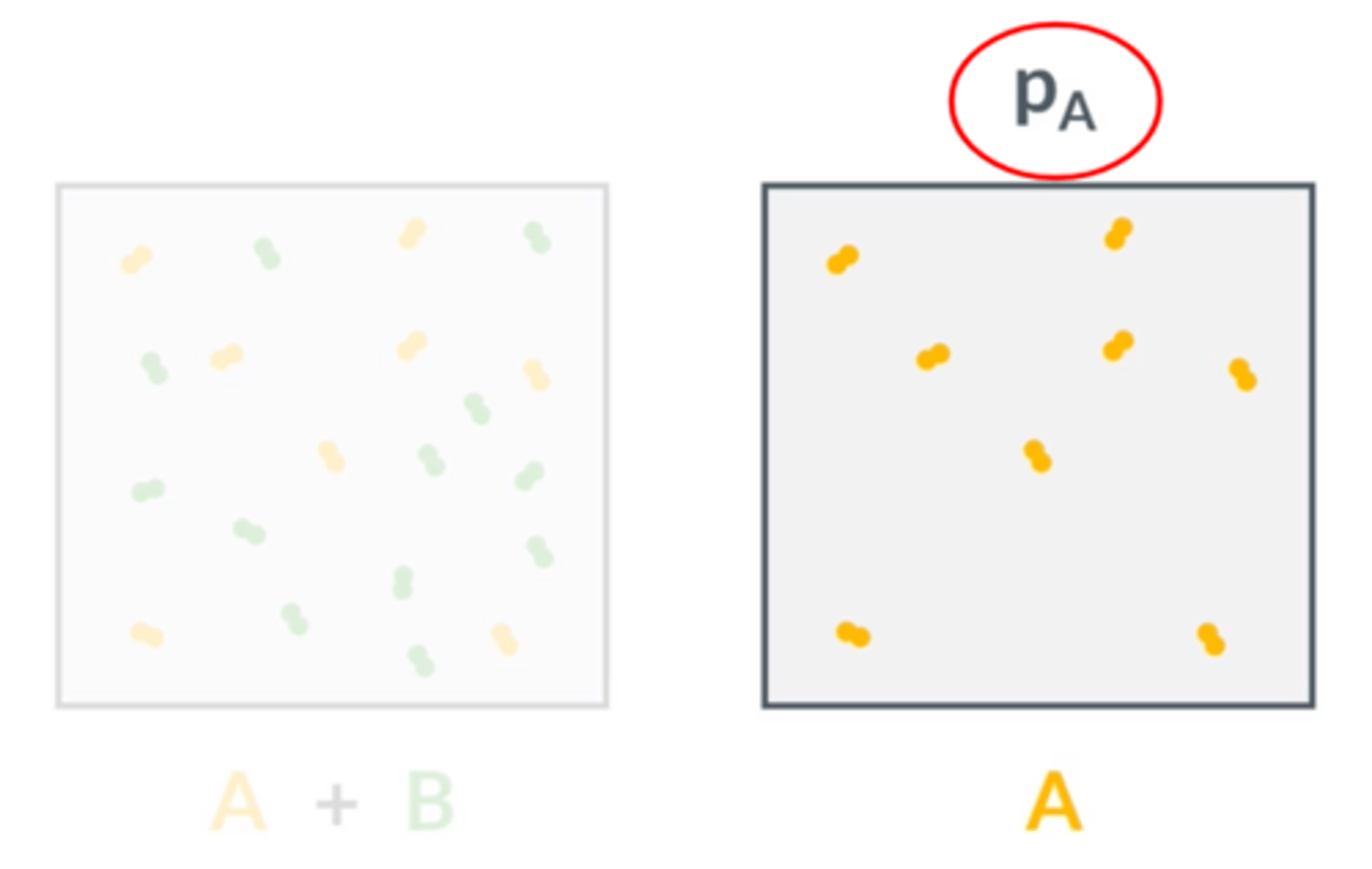
What is partial pressure?
The pressure that the gas would exert if it alone occupied the volume of the mixture.
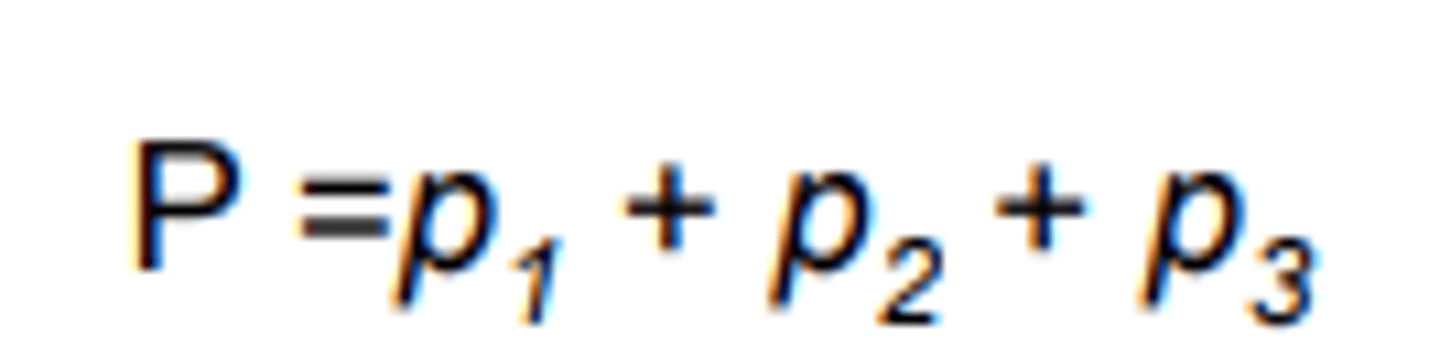
How would you represent the partial pressure of gas A?
Equilibrium constant of pressure (Kp)
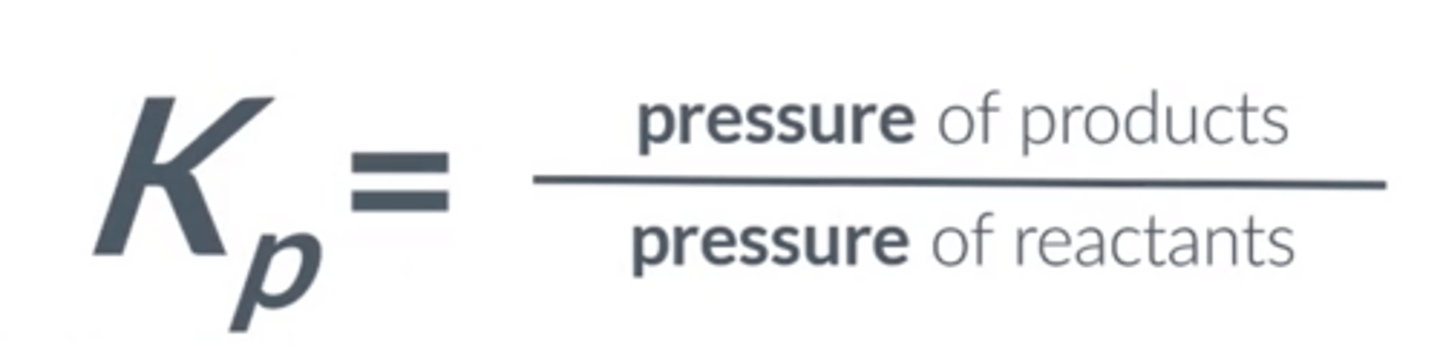
Give the Kp equation for aA + bB -- cC + dD
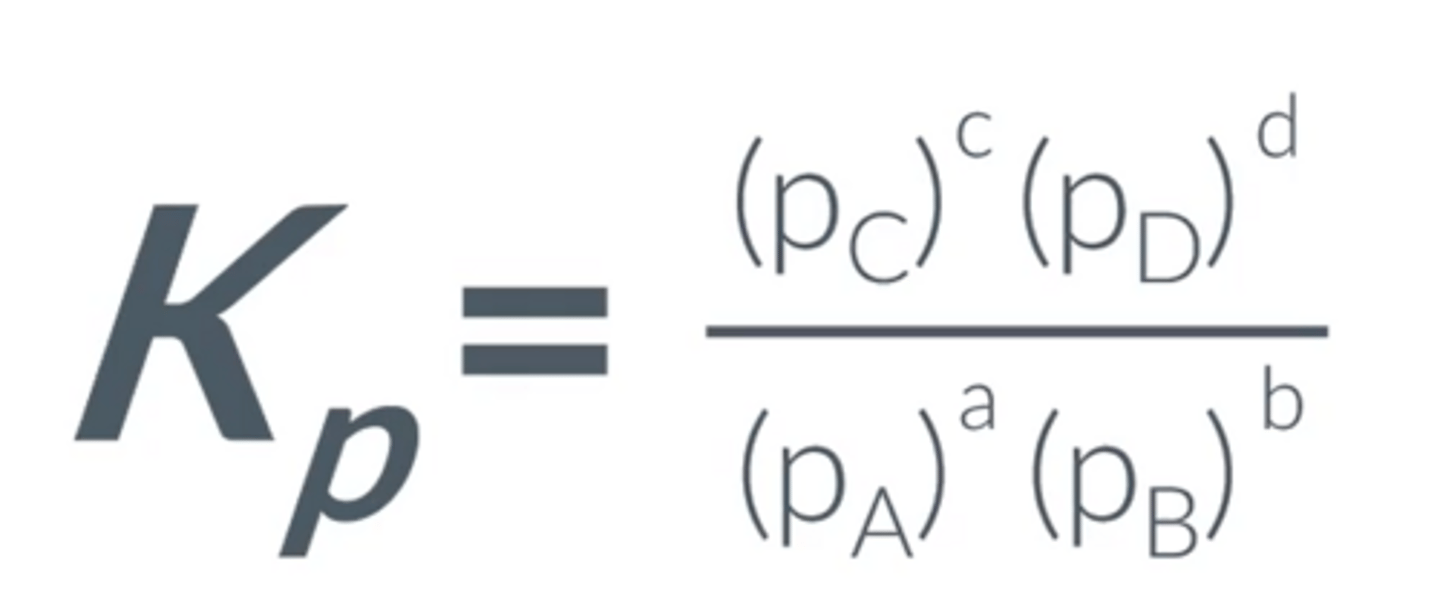
Total pressure of a gas
The sum of the partial pressures of each (individual) gas

How would you calculate the partial pressure of a gas?
Partial pressure p = mole fraction x total pressure
How is partial pressure calculated in a lab?
pA = total pressure x mole fraction of A
May not know the mole fraction of each gas in a mixture. This means they need a different way to measure the partial pressure of each gas and one of the most common ways they do this is by using a mass spectrometry.
Consider the following reaction: 2NO (g)+O2(g) ⇌ 2NO2(g)
At equilibrium, the mole fraction of each gas present is as follows: NO=0.364, O2=0.231 and NO2=0.405. The total pressure of the system is 85kPa.
Calculate the partial pressure of each gas in the system.

Consider the following reaction: SO2 (g) + O2 (g) ⇌ 2SO3 (g)
At equilibrium, the mole fraction of each gas present is as follows: SO2=0.185 and O2=0.174. The total pressure of the system is 210kPa. Calculate the partial pressure of each gas in the system.
Give your answer to 3 significant figures

What is the mole fraction?
The sum of the mole fractions for each gas in a mixture will always equal 1
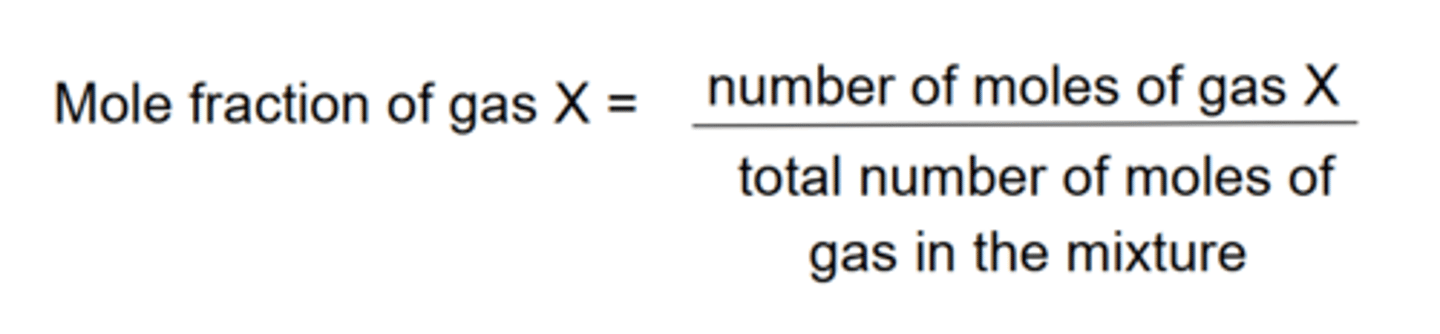
Consider the following reaction: 3O₂ (g) ⇌ 2O₃ (g)
At equilibrium, 16 moles of O₂ and 24 moles of O₃ are present. Calculate the mole fraction of each gas.

A reaction is represented by aA (g) + bB (g) ⇌ cC (g) + dD (g), what Kp for the system?

How do you calculate the units for Kp?
Write out the units for the partial pressures in the same arrangement as the Kp equation and cancel out/multiply together.
Usually in Pa, kPa, atm etc. DO NOT CHANGE UNITS
Determine the units for the equilibrium constant for this reaction if pressures are measured in units of atmospheres (atm).
N₂(g) + 3H₂(g) ⇌ 2NH₃(g)
atm⁻²
What is the effect of increasing temperature on Kp for an endothermic reaction?
Equilibrium shifts to the right, so partial pressures of products increase, so Kp increases
When we increase the temperature of a reaction, the value for Kp...
increases if the forward reaction is endothermic.
decreases if the forward reaction is exothermic.
What is the effect of increasing the overall pressure on Kp for this reaction?
Pressure does not affect Kp as, if moles of gas are not the same on each side), either top or bottom of Kp expression will have a total pressure term that does not cancel.
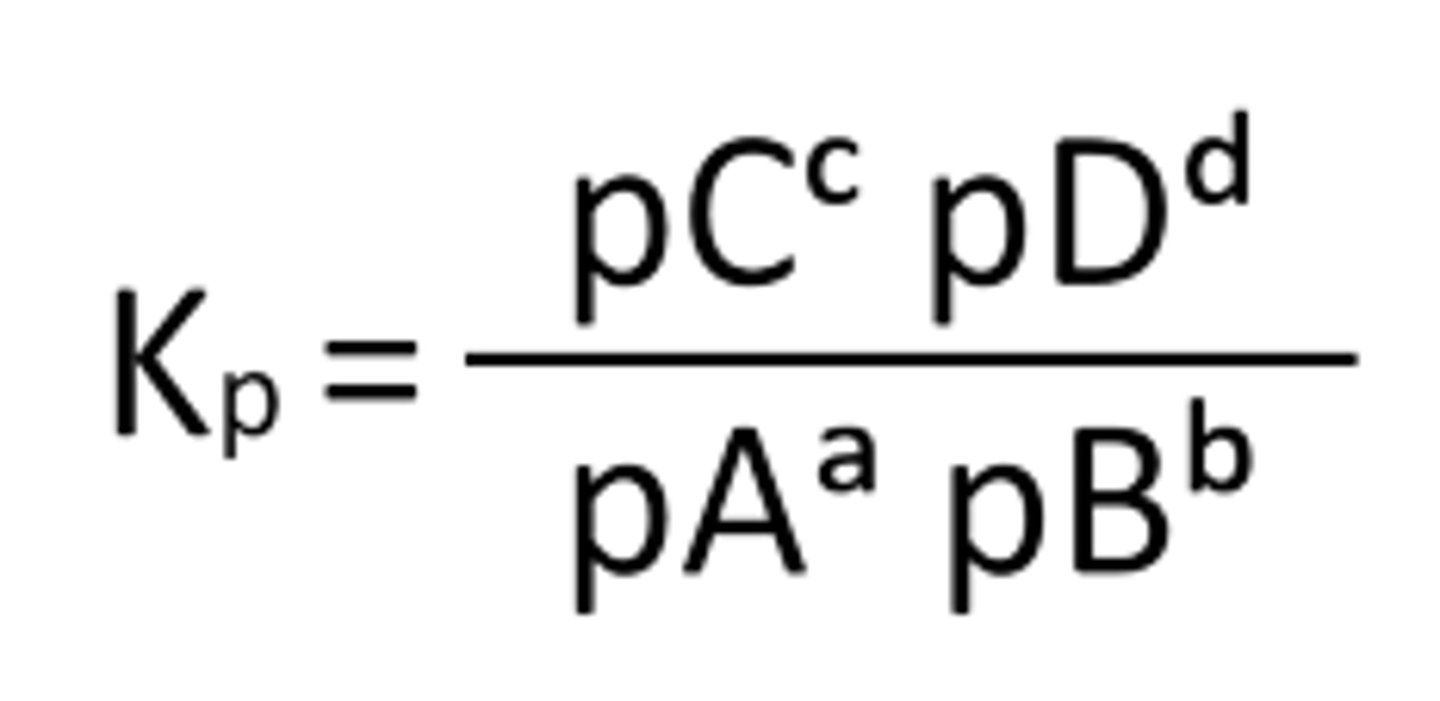
What will be the kinetic effect of increasing the temperature and pressure for any reaction?
Increasing both will increase the rate of reaction as: Temperature - many more particles have energy greater than or equal to the activation energy → more successful collisions per second
Pressure - more particles in the same volume → more successful collisions per second.
Consider the following reaction:
2X(g) + 3Y(g) ⇌ 4Z(g)
At equilibrium, the partial pressures of X and Z are 0.65 atm and 1.35 atm respectively. The equilibrium constant in terms of pressure is 10.78 atm⁻¹.
Calculate the partial pressure of Y in the equilibrium mixture with appropriate units.