Mistakes to go over
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
What are the two reagents that indicate robinson annulation?
Draw them out
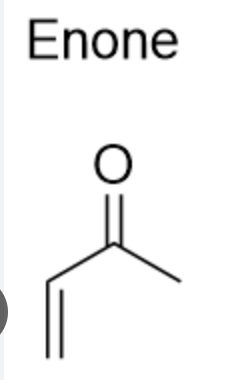
A enone + 1-3 dicarbonyl

why?
because for aldol condensation you need TWO H+
Baeyer– Villiger Oxidation
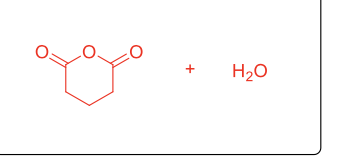

What reaction is this?
Robinson Annulation
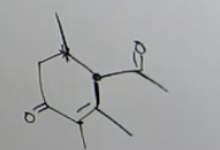
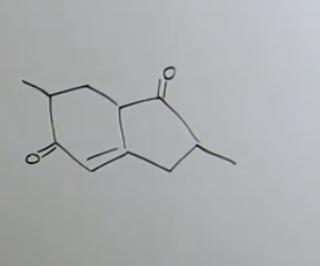
How do you know this is robinson annulation?
It has two reactions 1-3 functional groups and 1-5 so we know it was michael + aldol condensation
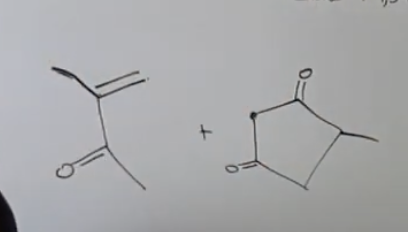
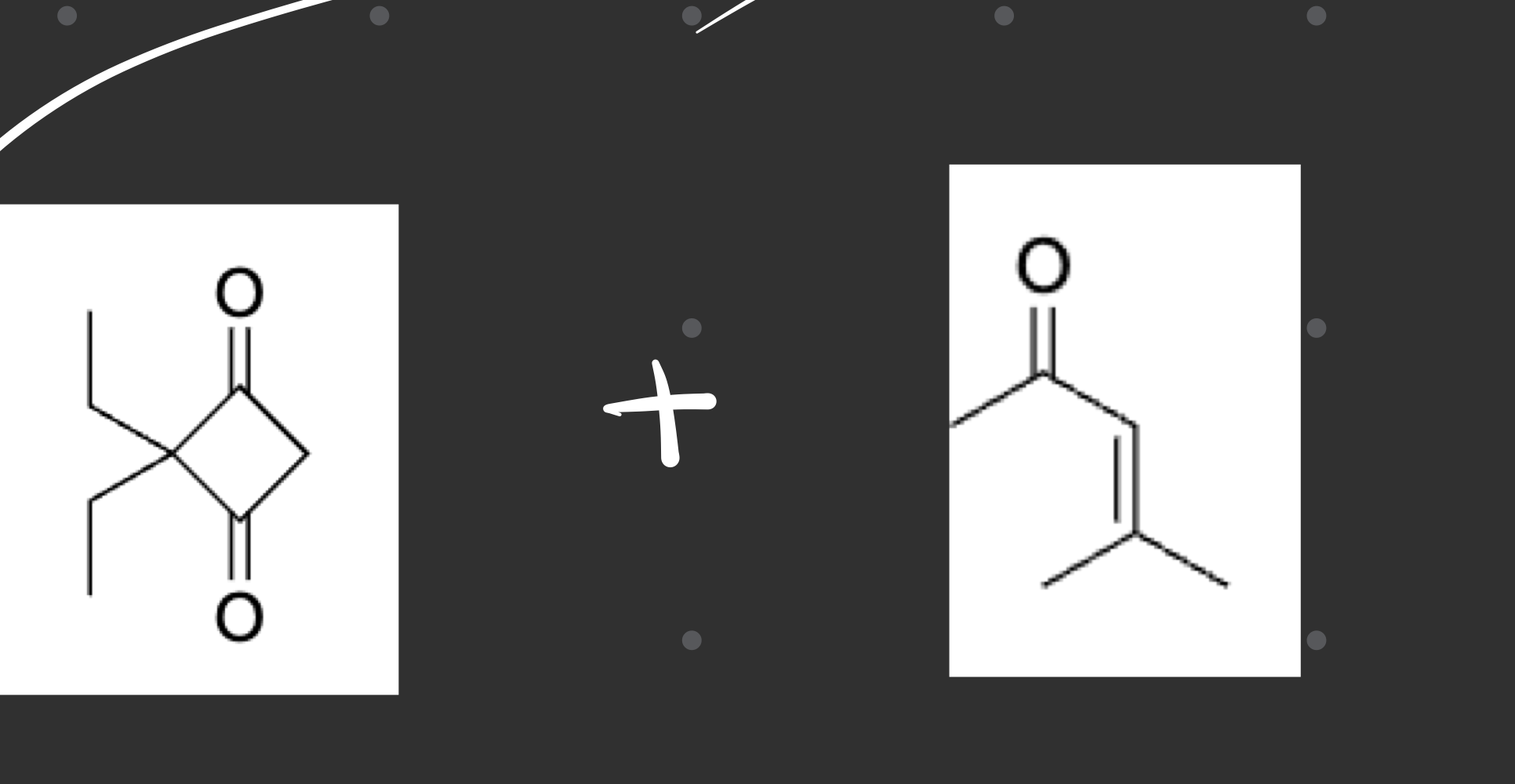
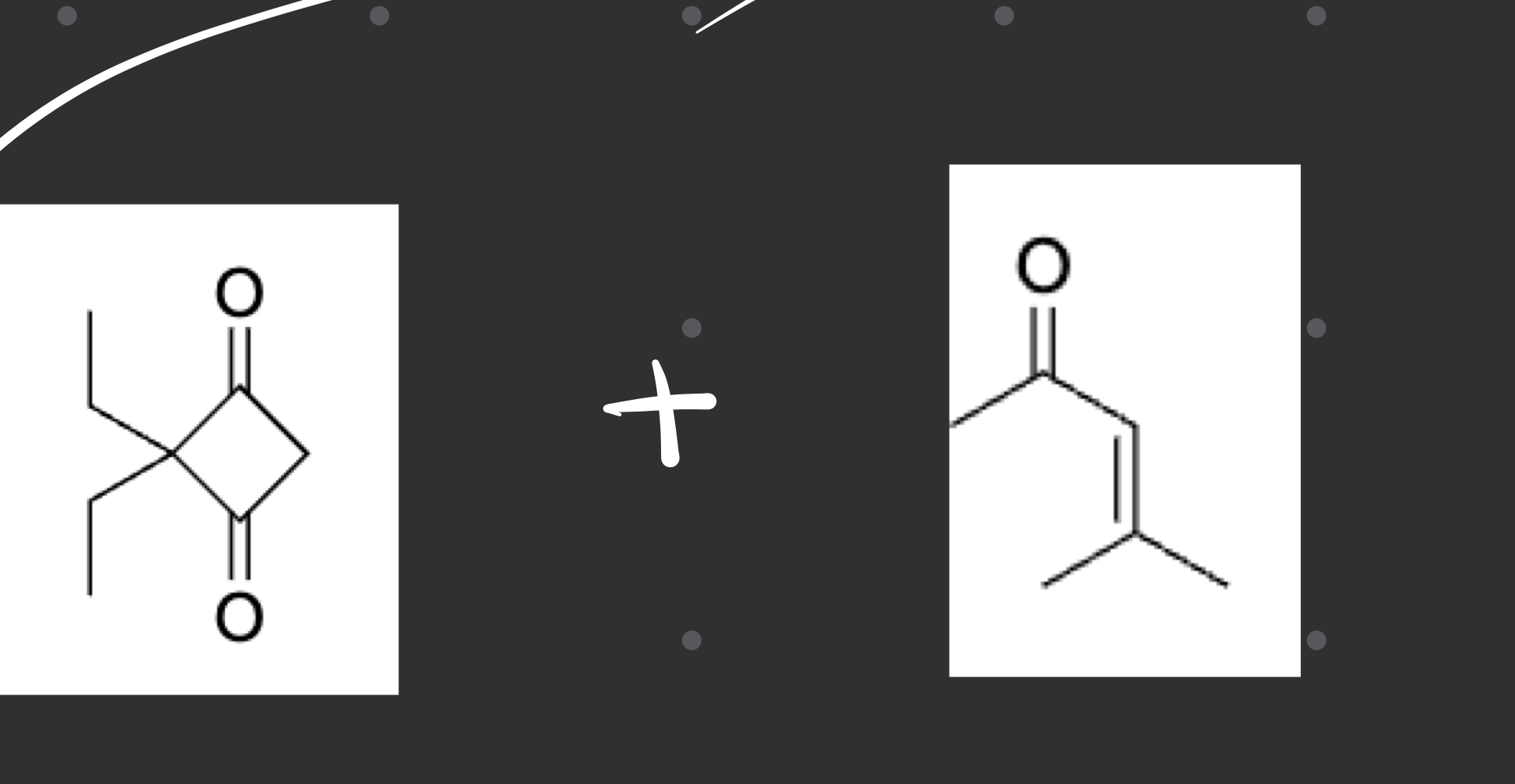
what type of reaction was this and whats the evidence
1,3 diketone
enone (1,3)
Robinson Annulation
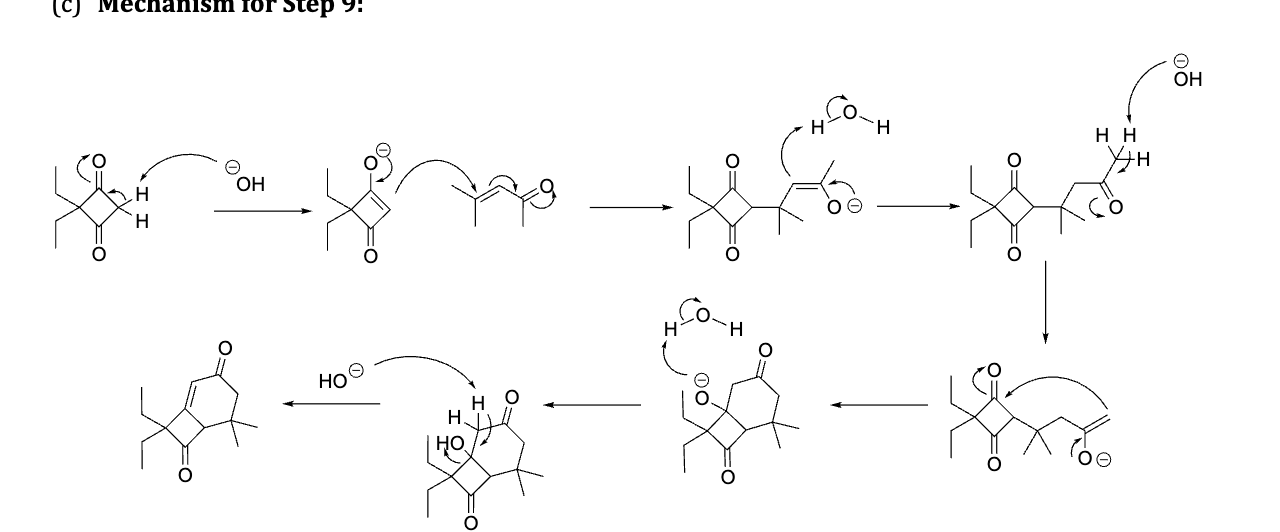
provide the Mechanism for the product of this reaction
What reagents are needed for a robinson annulation?
You are adding a base to remove acidic hydrogens, adding heat for condensation for aldol and then H20
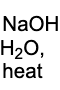
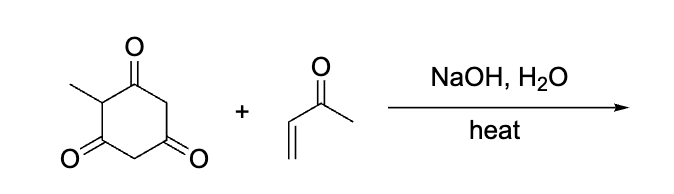
what was the product of this?
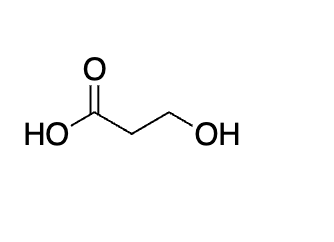
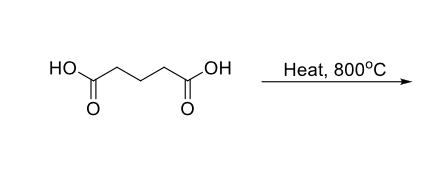
The H2S04, H20 reflux tells you that you make the COOH and it leaves as CO2
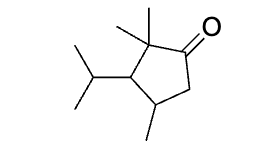
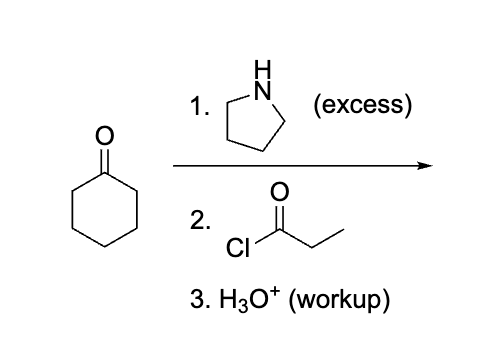
explain what is happening here, don’t do product yet rather draw the process ?
Although we are used to seeing R2NH for stork it will create an enamine which will react with the acyl cl to create that double bond ketone after the work u.
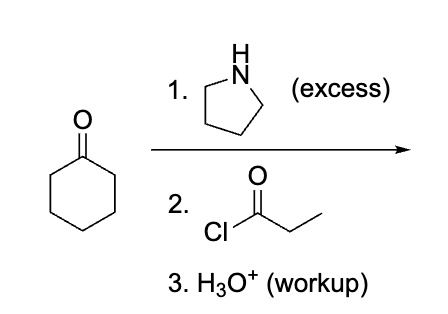
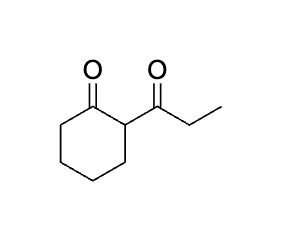
Product?
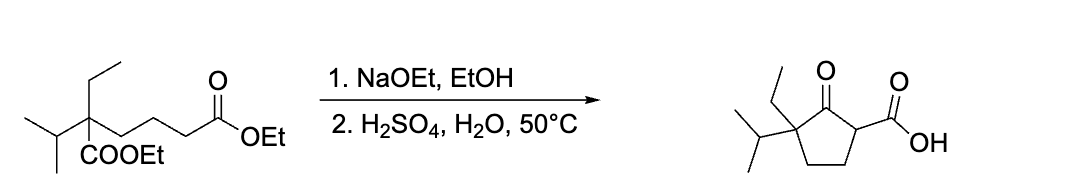
why does the COOH stay?
not hot enough for decarboxylation look for the word REFLUX or 100 degrees
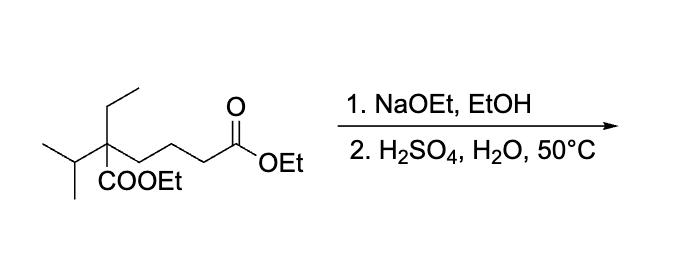
product
product?
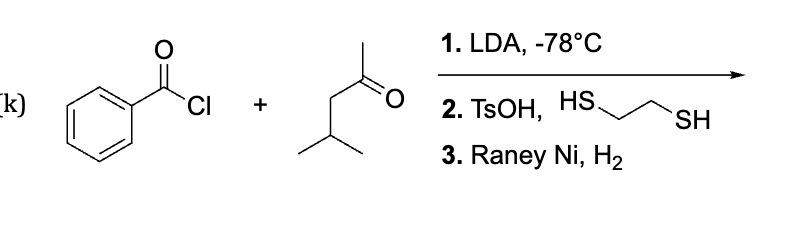
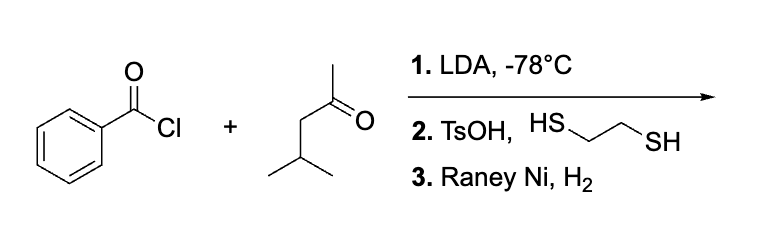
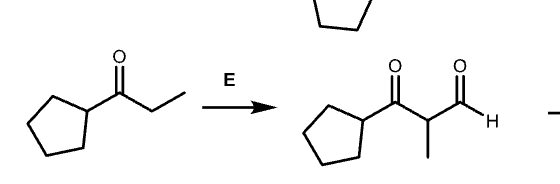
This has LDA which side will u use to attack
less substituted side
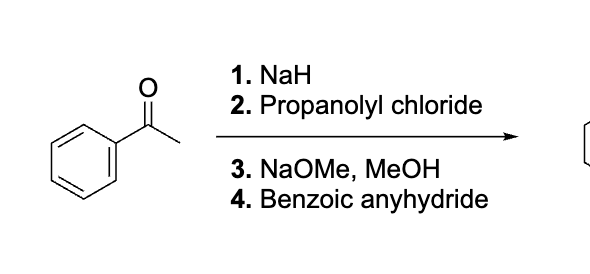
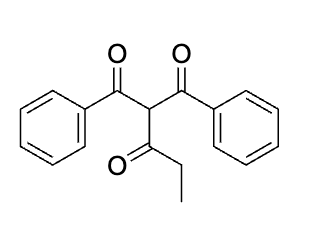
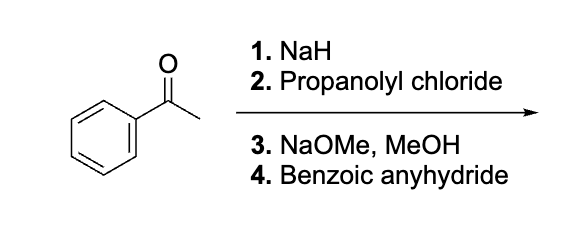
Draw each step for this
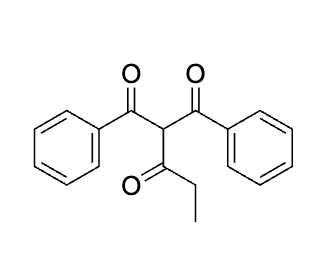
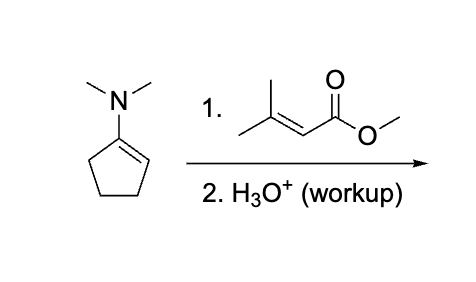
explain why this works before doing dont do it yet
emaine= same as aldol, hydrolyze to get rid of it
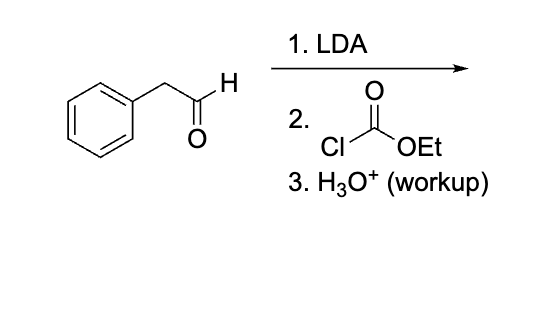
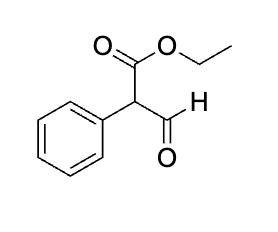
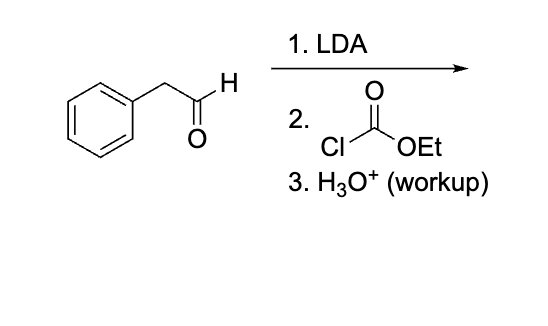
what is the concern ab this question?
because it is h30+ and specfically says workup and theres no step after just leave it orginlaly and dont make the COOH
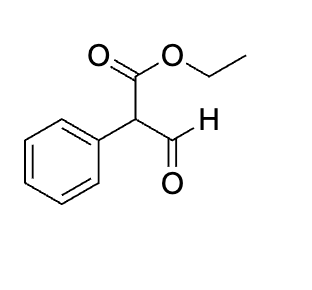
draw mechanism for step one
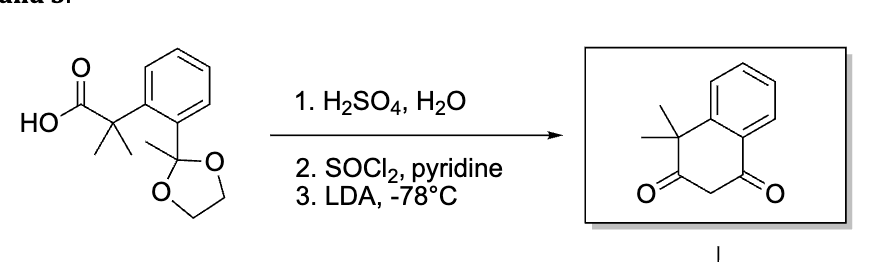
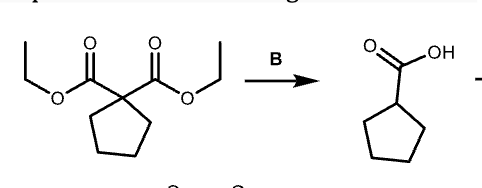
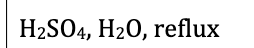
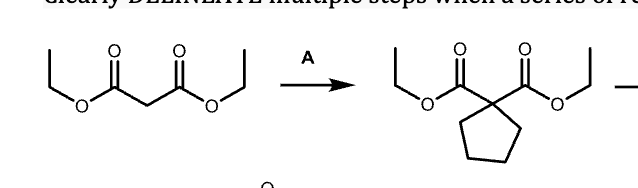
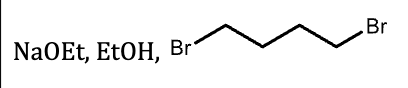

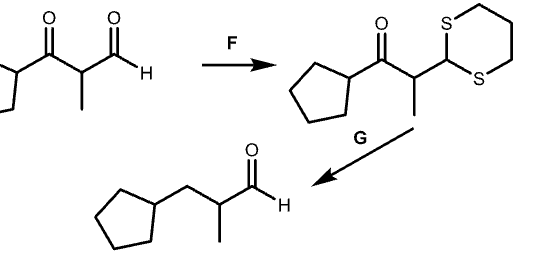
G?
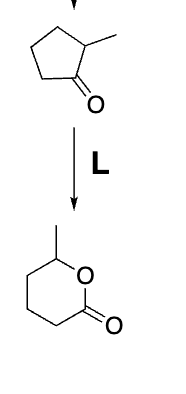
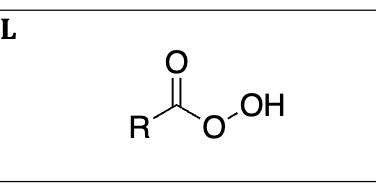
peracid so mcpba is right
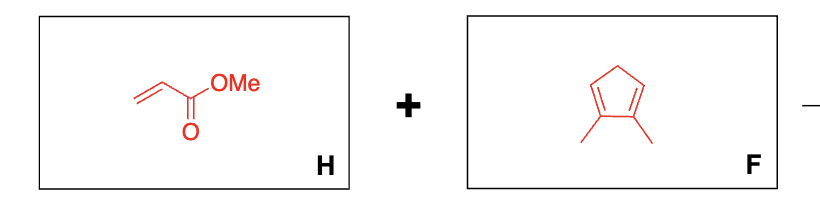
before answering what reaction is this?
diels-alder
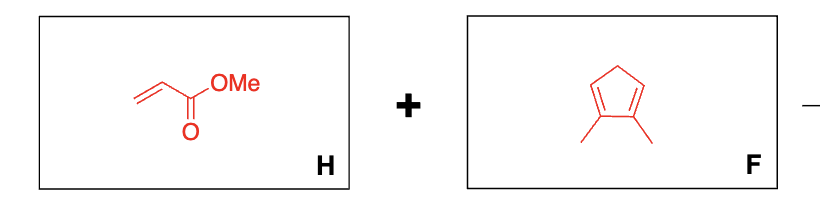
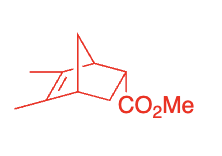
What reagent is needed for diels-alder?
heat
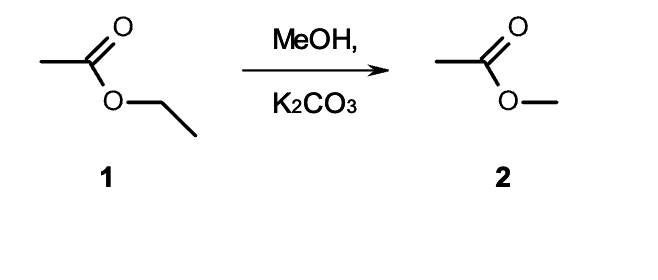
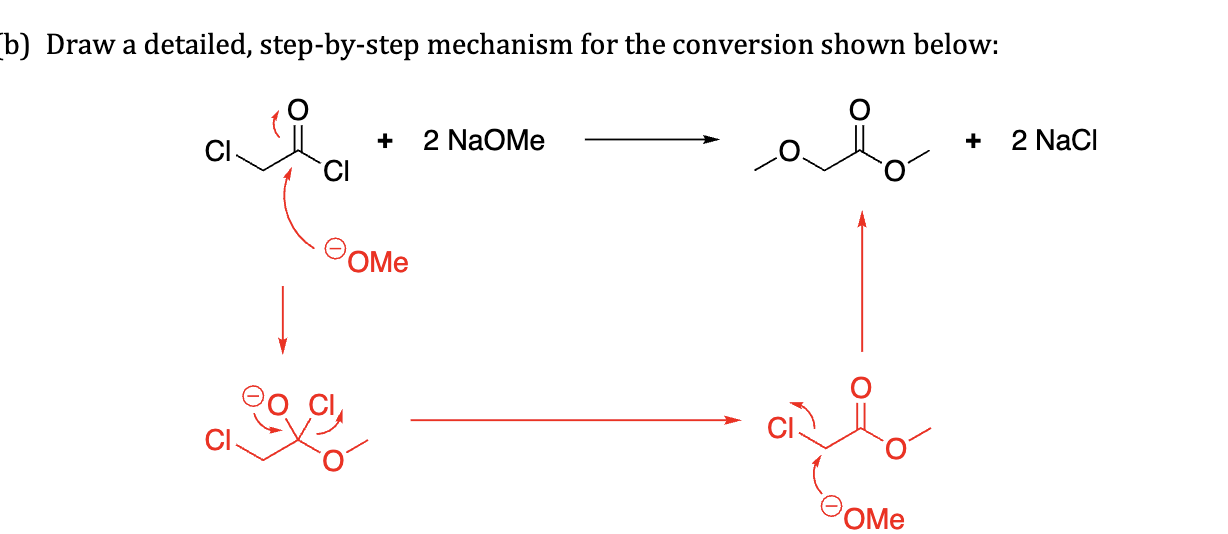
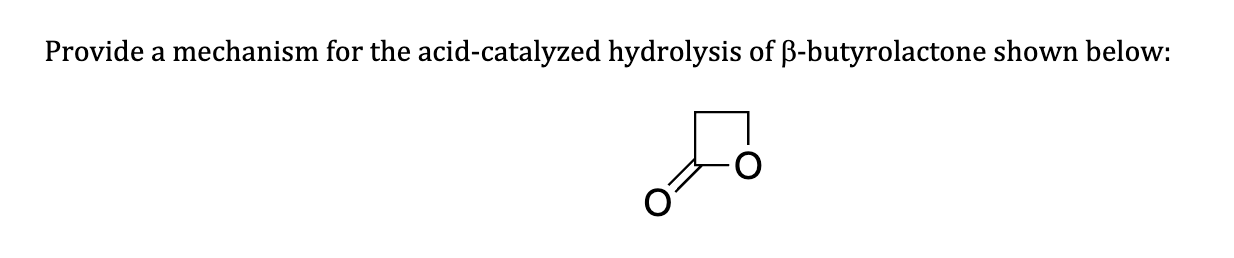
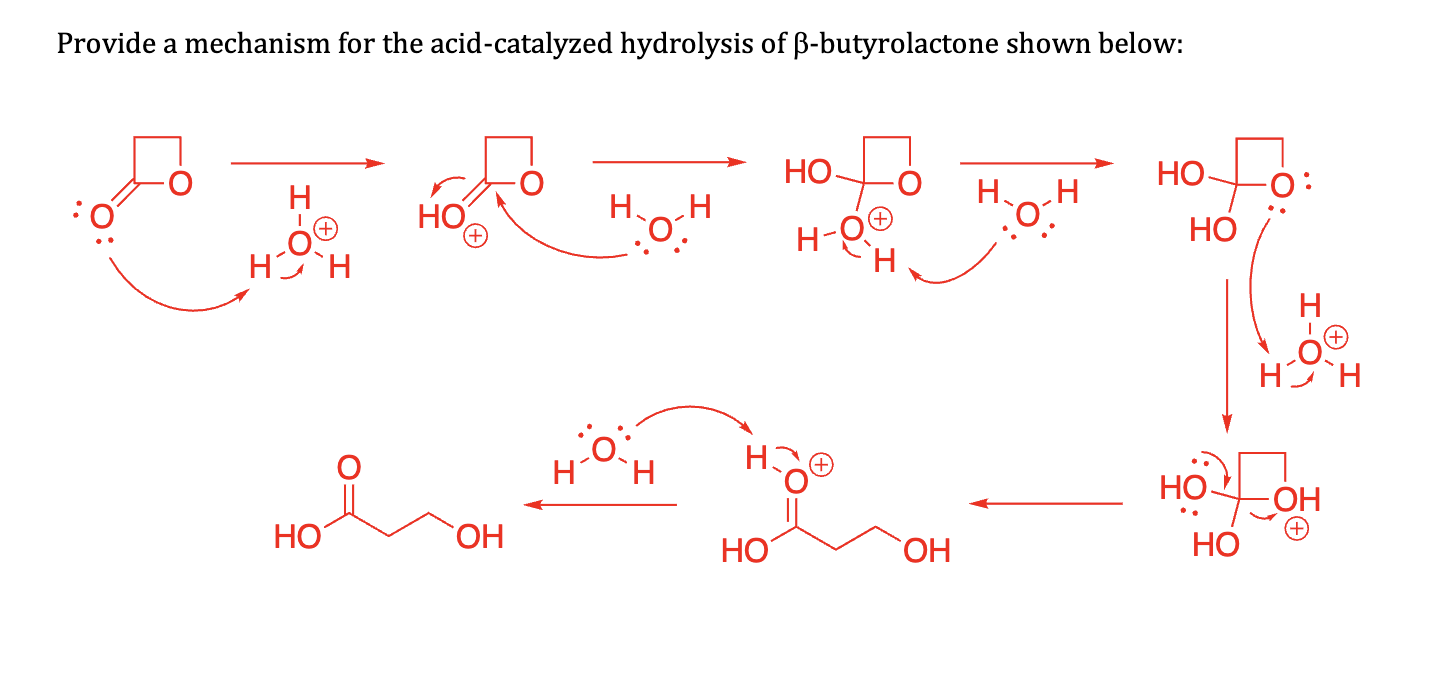
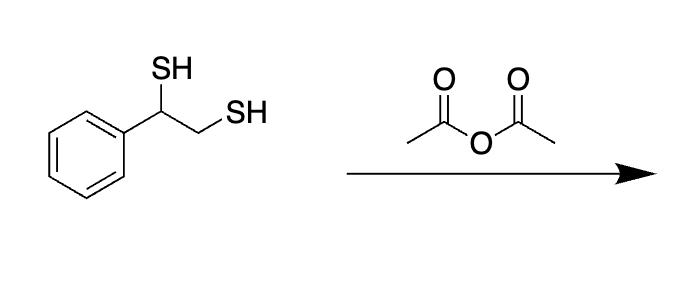
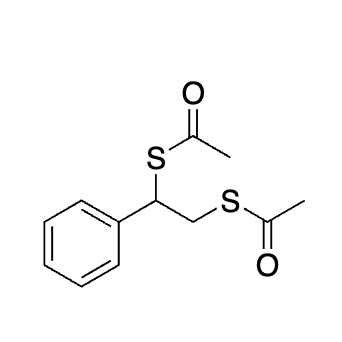
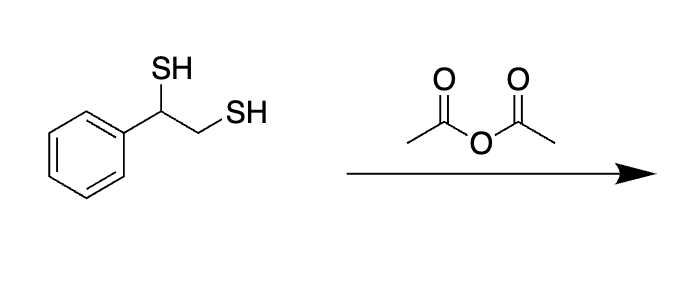
why does this not act like a protecting group
No acid to help catalyze that action
Not aldehyde or ketone
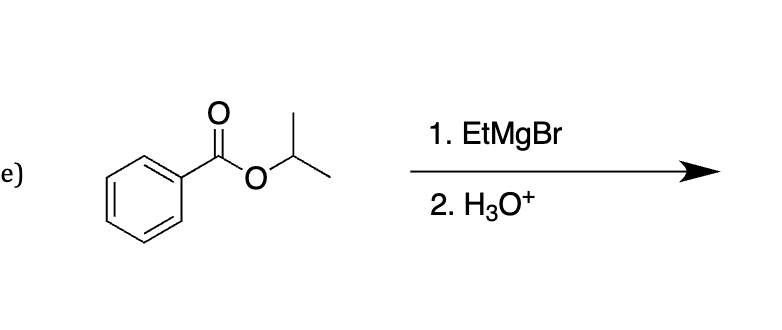
grignard with class two do it twice
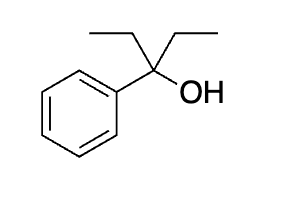
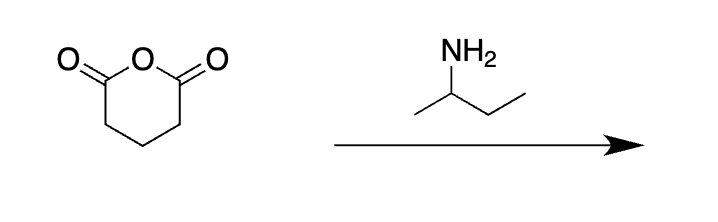
do it once
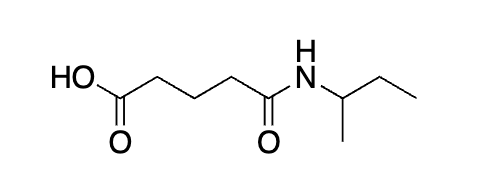
why does it not add on the OH to?
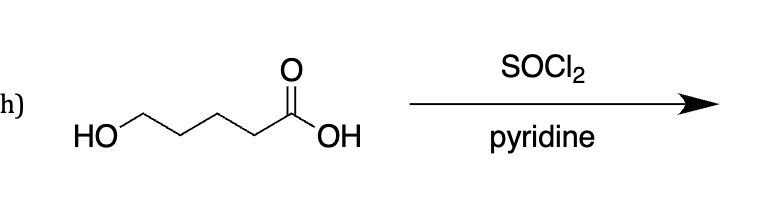
socl2 pyridine
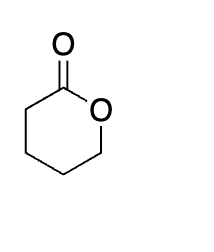
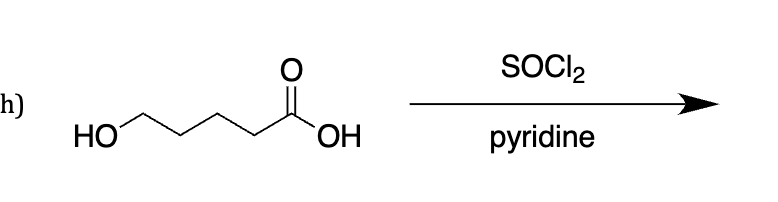
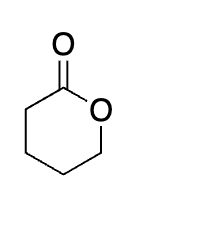
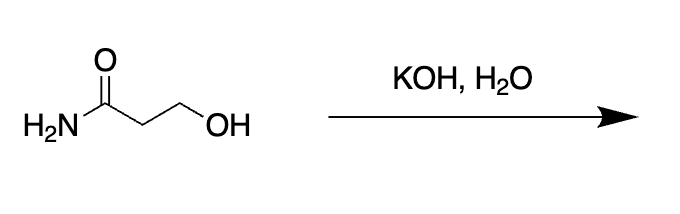
what is this
base hydrolyses gets rid of NH