Chemistry 3.3.1 and 2 - Intro to Organic
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Empirical formula
The simplest whole number ratio of atoms of each element in the compound
Molecular formula
The actual numbers of atoms of each element in the compound
Displayed formula
Compound is drawn with all atoms and all bonds shown
Structural formula
The compound is shown without bonds between atoms
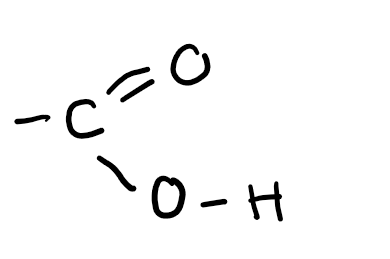
Functional group
Carboxylic acid (-oic acid)
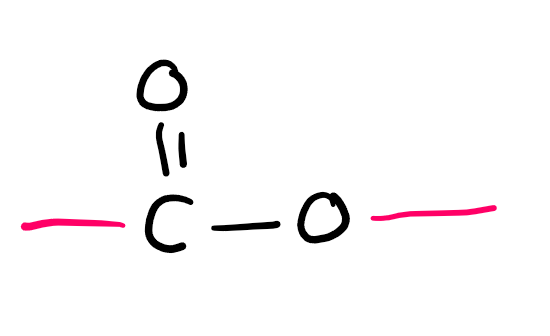
Functional group
Ester (-yl -oate)
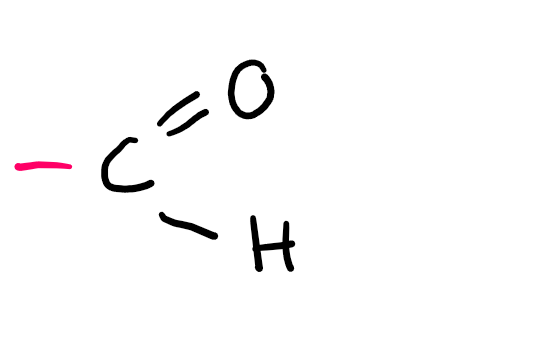
Functional group
Aldehyde (-al; oxo if not PFG)
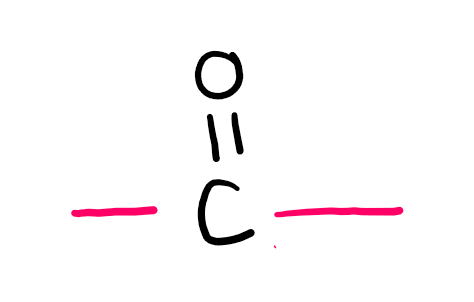
Functional group
Ketone (-one; oxo- if not PFG)
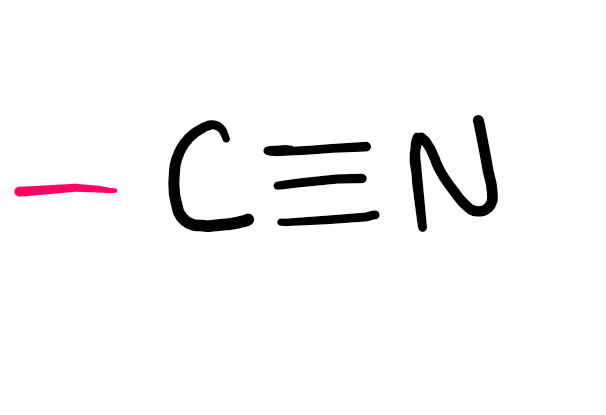
Functional group
Nitrile (-nitrile; cyano- if not PFG)
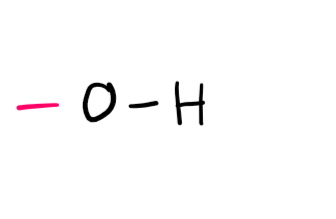
Functional group
Alcohol (-ol; hydroxy- if not PFG)
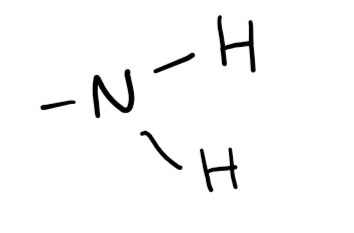
Functional group
Amine (-amine; amino- if not PFG)
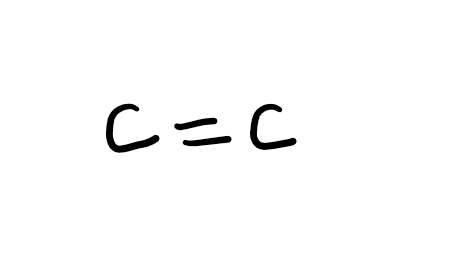
Functional group
Alkene (-ene; -en with higher priority FG)
Functional group order of importance (most to least)
Carboxylic acid, ester, aldehyde, ketone, nitrile, alcohol, amine, alkene, halogen
Structural isomerism
Same molecular formula, but different structural formulae
Chain isomer
Molecules with the same molecular formula but different length carbon chains
Position isomer
Molecules with the same molecular formula but with the functional group in a different position on the chain
Functional group isomer
Molecules with the same molecular formula but different functional groups
Stereoisomerism
When molecules have the same structural formula but different 3D arrangement of atoms in space
Why does EZ isomerism arise? (2)
Restricted rotation around the C=C double bond
2 different groups attached to each C in the C=C double bond
Saturated hydrocarbons: (3)
Have no C=C double bonds
Maximum number of Hs per C atom
Only C and H atoms
Homologous series
A series of compounds with the same general formula
Features of a homologous series: (5)
Same functional group
Similar chemical properties
Trend in physical properties
Same general formula
Each member differs by CH2
Free radical
Species with a high energy unpaired electron
Fractions of crude oil in order (lowest to highest b.p.)
Butane & propane, petrol, kerosene, diesel, fuel oil, bitumen
Use for butane & propane (i.e. natural gas)
Heating, cooking
Use for petrol
Car fuel
Use for kerosene
Jet fuel, lights
Use for diesel
Lorry fuel
Use for fuel oil
Ships, power stations
Use for bitumen
Roofs and roads
Thermal cracking temperature and pressure
1000 - 1200K, 70 atm
Catalytic cracking conditions
720K, 1 - 2 atm, zeolite catalyst
Problems caused by CO
Toxic - binds to haemoglobin in blood and prevents it from carrying enough oxygen; highly flammable
How are oxides of nitrogen formed?
Combustion of nitrogen in air at very high temperatures
Problems caused by oxides of nitrogen
Respiratory problems; acid rain (NO2+H2O → HNO3)
Problems caused by SO2
Respiratory problems; acid rain (SO2+ H2O+O2→H2SO4)
Catalytic converter
NOx → Nx + Ox
CO + O2 → CO2
Hydrocarbons → CO2 + H2O
Removal of SO2 from atmosphere
Can be reacted with CaO to produce CaSO3
Removal of C particulates from atmosphere
Filters in cars catch soot before entering atmosphere
Catalytic converter catalysts
Platinum, palladium, rhodium