energy, enzymes, mitochondria and respiration 4A, 4B, 4C
1/88
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
89 Terms
What is energy in biological systems?
Ability to do work in cells; required for all cellular processes
What are the forms of energy?
Kinetic (movement); thermal (heat); potential (stored); chemical (in bonds)
What is the first law of thermodynamics?
Energy cannot be created or destroyed, only converted between forms
What is the second law of thermodynamics?
Energy transfer increases entropy (disorder) of the universe
What is entropy?
Measure of disorder/randomness in a system
What is metabolism?
All chemical reactions in a cell or organism
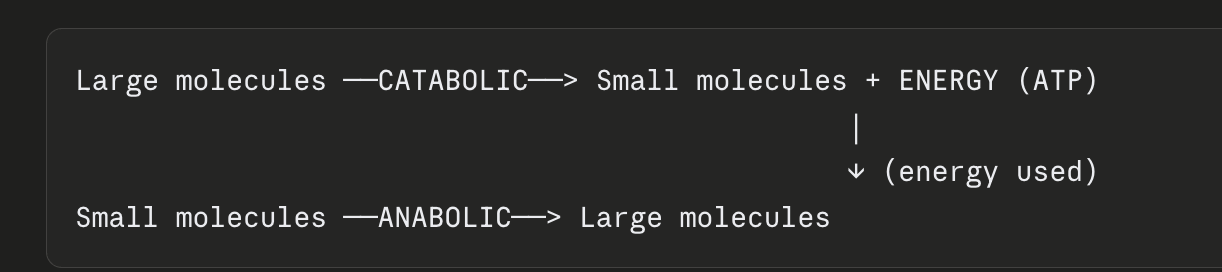
What are catabolic reactions?
Break down molecules; release energy
What are anabolic reactions?
Build molecules; require energy
What is free energy (G)?
Energy available to do work in a system
What is ΔG?
Change in free energy during a reaction
What does ΔG = 0 mean?
System is at equilibrium
What does negative ΔG mean?
Exergonic; releases energy; spontaneous
What does positive ΔG mean?
Endergonic; requires energy input; non-spontaneous
What is an exergonic reaction?
Reaction that releases energy (ΔG < 0)
What is an endergonic reaction?
Reaction that requires energy (ΔG > 0)
What is an example of an exergonic reaction?
Cellular respiration (glucose breakdown)
What is an example of an endergonic reaction?
Photosynthesis
What is activation energy (EA)?
Energy required to start a reaction
Why do reactions need activation energy?
To reach transition state where bonds can break/form
What is ATP?
Adenosine triphosphate; main energy carrier in cells
What are the components of ATP?
Adenine; ribose; three phosphate groups
Where is energy stored in ATP?
Bonds between phosphate groups
How much energy does ATP release?
~30.5 kJ/mol per ATP
What happens during ATP hydrolysis?
ATP → ADP + Pi + energy
What is energy coupling?
Using energy from exergonic reactions to drive endergonic reactions
How does ATP drive endergonic reactions?
Transfers phosphate to substrate making it more reactive and increasing its energy so the reaction can proceed.
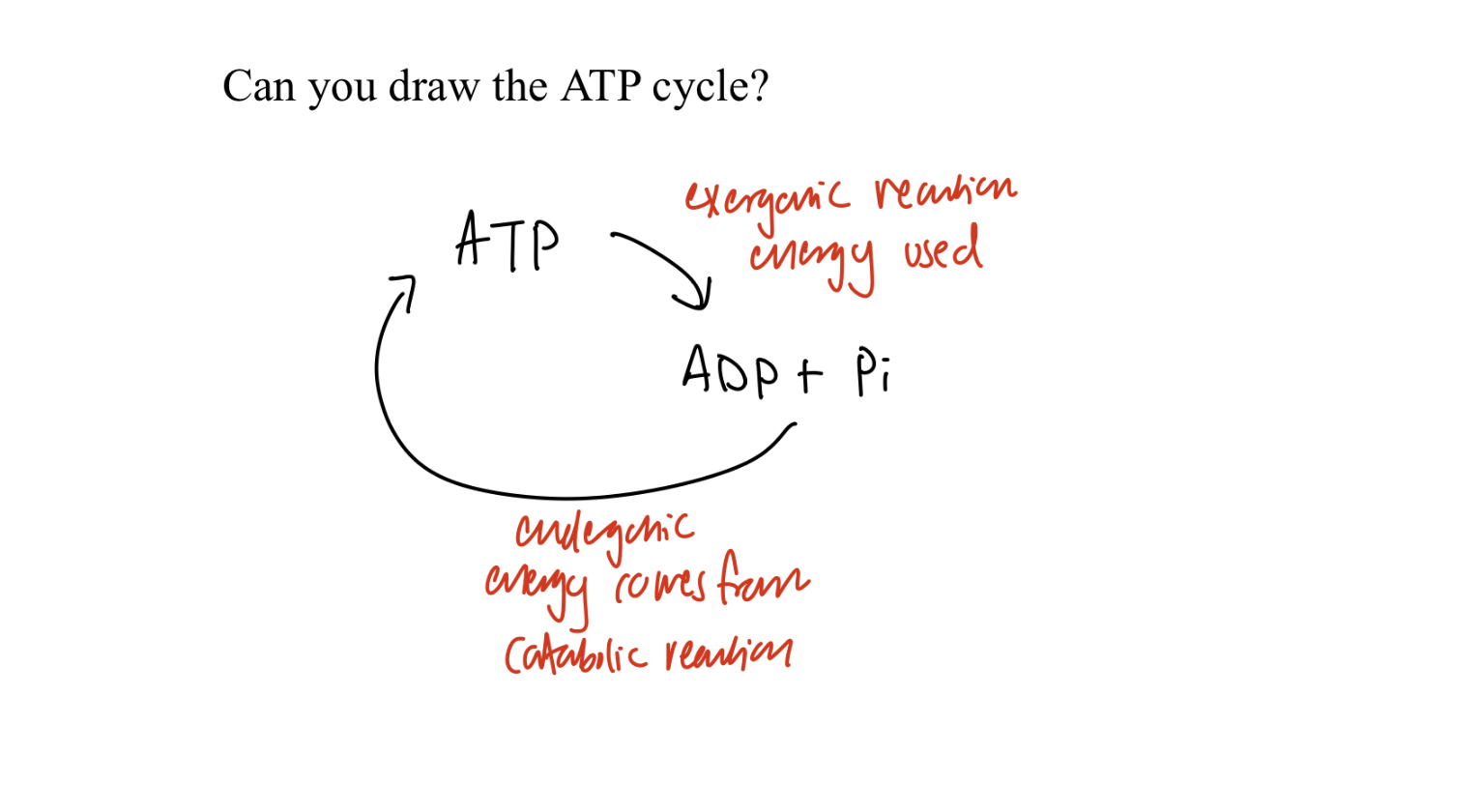
What is the ATP cycle?
ATP ↔ ADP + Pi (continuous regeneration)
What are enzymes?
Protein catalysts that speed up reactions without being used up
What is a substrate?
The molecule an enzyme acts on
What is the active site?
Region where substrate binds on enzyme
What is an enzyme-substrate complex?
Temporary complex formed when enzyme binds substrate
What happens during enzyme action?
Substrate binds → reaction occurs → products released
What is the lock and key model?
Substrate fits exactly into enzyme active site (rigid model)
Why is lock and key model limited?
Enzymes are flexible and can change shape
What is the induced fit model?
Enzyme changes shape to fit substrate better
How do enzymes lower activation energy?
lower activation energy by binding substrates in ways that make reactions easier to occur. They can orient substrates correctly for collision, strain or weaken existing bonds, create a favourable chemical environment, and form temporary interactions with substrates that stabilise the transition state and promote the reaction.
Do enzymes change ΔG?
No, they only lower activation energy
What factors affect enzyme activity?
pH; temperature; environment; cofactors
What are cofactors?
Inorganic ions required for enzyme function
What are coenzymes?
Organic molecules that assist enzymes
What are competitive inhibitors?
Bind active site; compete with substrate
What are non-competitive inhibitors?
Bind elsewhere; change enzyme shape
What is allosteric regulation?
Binding at regulatory site affects enzyme activity
What are allosteric activators?
Stabilise active form of enzyme
What is feedback inhibition?
End product inhibits early enzyme in pathway
What is protease activation?
Enzymes produced inactive and activated when needed
What is cellular respiration?
Process that converts glucose + O2 → CO2 + H2O + ATP
What type of reaction is respiration?
Redox (electron transfer)
What is oxidation?
Loss of electrons
What is reduction?
Gain of electrons
Where does respiration occur?
Cytosol and mitochondria
What is the structure of mitochondria?
Outer membrane; inner membrane; cristae; matrix; intermembrane space
What is the outer membrane?
Smooth outer boundary
What is the inner membrane?
Folded membrane containing ETC
What are cristae?
Folds increasing surface area
What is the matrix?
Inner space where Krebs cycle occurs
What is intermembrane space?
Space between membranes
What are the 3 stages of respiration?
Glycolysis; Krebs cycle; electron transport chain
Where does glycolysis occur?
Cytosol
Where does Krebs cycle occur?
Matrix
Where does ETC occur?
Inner membrane
What happens in glycolysis?
Glucose → 2 pyruvate; 10 enzyme steps
What is energy investment phase?
Uses 2 ATP
What is energy payoff phase?
Produces 4 ATP and 2 NADH
What is net ATP from glycolysis?
2 ATP
What is NADH?
Electron carrier
What happens to pyruvate?
The pyruvate moves into the matrix of the mitochondria and is converted into Acetyl CoA which enters the Citric Acid cycle
CO2 released
What happens in Krebs cycle?
Acetyl-CoA + oxaloacetate → citrate → CO2 + NADH + FADH2 + ATP
What is regenerated in Krebs cycle?
Oxaloacetate
What is produced in Krebs cycle?
6 NADH; 2 FADH2; 2 ATP; 4 CO2
the NADH from glycolysis and the NADH and FADH2 from the Citric Acid cycle enter
an electron transport chain in the inner membrane of the mitochondria.
What happens in electron transport chain?
Electrons passed through protein complexes releasing energy
What is the role of oxygen?
Final electron acceptor
What happens to H+?
Pumped into intermembrane space
What is chemiosmosis?
H+ flows through ATP synthase to generate ATP
What is ATP synthase?
Enzyme that produces ATP
What is oxidative phosphorylation?
ATP production via ETC and proton gradient
How many ATP per glucose?
~36–38 ATP
What is fermentation?
Process used when oxygen is absent
Why is fermentation needed?
Regenerates NAD+ so glycolysis can continue
What are the types of fermentation?
Lactic acid fermentation; alcohol fermentation
What happens in lactic acid fermentation?
Pyruvate → lactate
What happens in alcohol fermentation?
Pyruvate → ethanol + CO2
Metabolism definition
Metabolism is the totality of an organism's chemical reactions — the sum of all chemical processes that occur in a living cell or organism to maintain life. It includes both catabolic (breaking down) and anabolic (building up) pathways.
Catabolic vs anabolic pathways
ATP reaction
ATP + H₂O → ADP + Pᵢ + Energy (~30.5 kJ/mol)
why energy coupling is important
Allows cells to carry out non-spontaneous reactions (building macromolecules, active transport, muscle contraction) that are essential for life
ATP is the universal coupling agent in cells
Makes metabolism efficient — energy from catabolic reactions isn't wasted; it's captured in ATP and reused
atp cycle
whats a kj
A kilojoule (kJ) is a unit used to measure energy
whats a mole
a unit amount of a substance. 1 mole = 6.02 × 10²³ particles