nuclear physics
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
what did the plum pudding model say
an atom was made up of a sphere of positive charge with small areas of negative charge evenly distributed throughout
how did rutherford scattering change what we know about atoms
demonstrated the existence of s nucleus
produced the nuclear model
what was the rutherford experiment
alpha source and gold foil in an evacuated chamber covered in a fluorescent coating (so you could see where the charged alpha particles hit the inside of the chamber)
miscroscope to observe the path of the alpha particles
what would we expect to happen in the rutherford experiment, according to the plum pudding model
the positively charged alpha particles would be deflected by a very small amount when passing through the foil
results and conclusions of the rutherford experiment
most alpha particles passed through the foil with no deflection (an atom is mostly empty space)
a small amount of alpha particles were deflected by a large angle (the centre of an atom is positively charged as positive alpha particles were repelled from the centre and deflected)
very few alpha particles were deflected by more than 90° (the centre of an atom is very dense, as it could deflect fast alpha particles, but also very small as a very small amount of particles were deflected by this amount)
an atom has a small, dense, positively charged nucleus at its centre
define radiation
an unstable nucleus emits energy in the form of EM waves or subatomic particles in order to become more stable
different properties of alpha, beta and gamma radiation
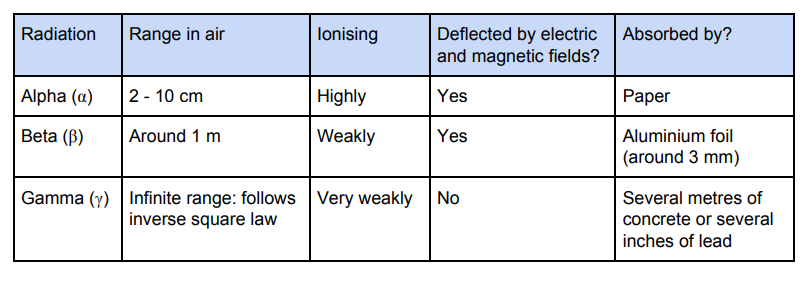
uses of gamma radiation
as a detector - a radioactive source with a short half-life (to reduce exposure), which emits gamma radiation, can be injected into a patient and the gamma radiation can be detected using gamma cameras in order to help diagnose patients
to sterilise surgical equipment - gamma kills any bacteria
radiation therapy - kills cancerous cells
used in medicine as it is weakly ionising (less damage than alpha and beta)
inverse square law
as gamma radiation moves through the air, it spreads out in all directions equally
intensity follows an inverse square law
I = k/ x2
x is distance from source
sources of background radiation
radon gas released from rocks
artificial sources caused by nuclear weapons testing and nuclear meltdowns
cosmic rays that enter the Earth’s atmosphere from space
rocks that contain naturally occurring radioactive isotopes
what does radioactive decay being a random process mean
you can’t predict when the next decay will occur
what does λ represent
the decay constant of a given radioactive nucleus
the probability of the nucleus decaying per unit time
you can only use it when there is a large number of nuclei in the sample
how can radioactive nuclei with long half-lives be used
dating of objects - nuclei with long half-lives (e.g. carbon-14) can be used to date organic objects
measure the current amount of carbon-14 and compare it to the initial amount (the percentage of which is approximately equal in all living things)
how can radioactive nuclei with relatively short half-lives be used
medical diagnosis - used as radioactive tracers in medical diagnosis
e.g Technetium 99m as it is a pure gamma emitter, has a half life of 6 hours and can be easily prepared on site
explain nuclear instability
nuclei are held together by the strong nuclear force
protons experience a force of repulsion due to the electromagnetic force
if the forces are unbalanced the nuclei will become unstable and experience radioactive decay
four reasons a nucleus might become unstable
it has too many neutrons, protons, or nucleons, or too much energy
depending on why it is unstable it will decay in a different way
nuclei may decay through several types of emission before becoming stable
how will an unstable nucleus decay if it has too many neutrons
through beta-minus emission
neutron changes into proton and beta minus particle and antineutrino are released
how will an unstable nucleus decay if it has too many protons
through beta-plus emission or electron capture
in beta-plus decay a proton changes into a neutron and beta plus particle and neutrino are released
in electron capture an orbiting electron is taken in by the nucleus and is combined with a proton, causing the formation of a neutron and a neutrino
how will an unstable nucleus decay if it has too many nucleons
through alpha emission
how will an unstable nucleus decay if it has too much energy
through gamma emission
usually occurs after a different type of decay because the nucleus becomes excited and has excess energy
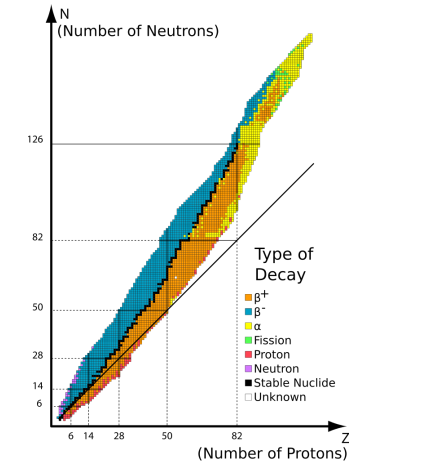
what does this graph show and why
number of neutrons and protons in a stable nucleus doesn’t increase uniformly beyond around 20 of each
the electromagnetic force of repulsion becomes larger than the strong nuclear force beyond this amount
more neutrons are needed to increase the distance between the protons to decrease the magnitude of the electromagnetic force to keep the nucleus stable
what is the distance of closest approach
e.g. an alpha particle is fired at a gold nucleus
the alpha particle has a KE which can be measured
as it moves towards the positively charged nucleus, it experiences an electrostatic force of repulsion
this causes it to slow down, and KE is converted to electric potential energy
the point at which it stops (KE = 0) is its distance of closest approach
put epe = ke and solve for r
gives an estimate (overestimate) for the nuclear radius of an atom
how can you use electron diffraction to find the nuclear radius of an atom
electrons are leptons so don’t interact with nucleons in the nucleus through the strong nuclear force, as an alpha particle would, so more accurate estimate for the radius than with the distance of closest approach
electrons are accelerated to very high speeds and directed at a very thin film of material in front of a screen
they diffract through gaps between nuclei and form a diffraction pattern (concentric circles that are brighest at the centre)
can use the angle of the first minimum in the formula sinθ = 0.61λ/R
λ = De Broglie wavelength
show that nuclear density is constant
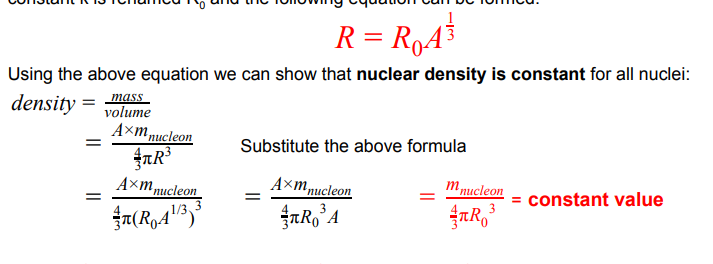
explain mass defect
mass of nucleus is less than the mass of the nucleons that it is made up of
the mass “lost” is converted into energy and released when the nucleons fuse to form a nucleus
define binding energy
the energy required to separate the nucleus into its nucleons
define the atomic mass unit (u)
equal to 1/12 the mass of a carbon-12 atom
describe nuclear fission
the splitting of a large nucleus into two smaller daughter nuclei
occurs in very large nuclei that are unstable (e.g. uranium)
why is energy released in nuclear fission
the daughter nuclei have a higher binding energy per nucleon
describe nuclear fusion
two smaller nuclei join together to form one larger nucleus
only occurs in fairly small nuclei
why is energy released in nuclear fusion
the larger nucleus has a much higher binding energy per nucleon
difference in energy released + required for fission and fusion
fusion releases more energy but only occurs at extremely high temperatures as a lot of energy is needed to overcome the electrostatic force of repulsion between nuclei
element with the highest binding energy per nucleon
iron (nucleon number 56)
binding energy per nucleon vs nucleon number graph
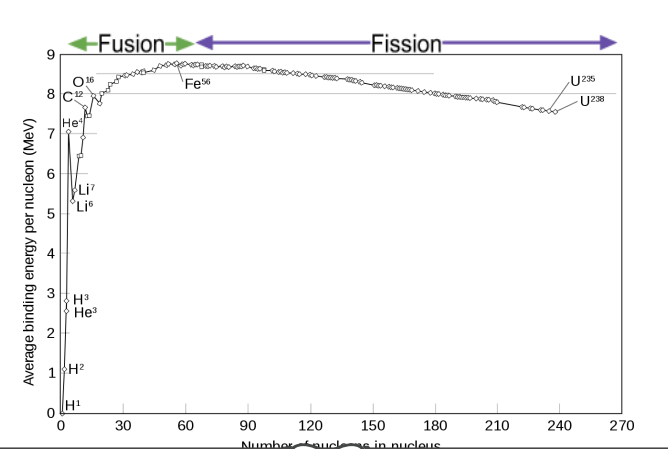
benefits of nuclear reactors
creates electricity without the emission of greenhouse gases
less fuel needed (compared to coal)
what is a thermal neutron
a low-energy, slow-moving free neutron in thermal equilibrium with its surroundings
describe induced fission
Fission can be induced in certain elements such as uranium-235
done by firing a thermal neutron into the uranium nucleus causing it to become extremely unstable
thermal neutrons have a low energy meaning they can induce fission whereas neutrons with a higher energy rebound away from the uranium-235 after a collision and do not cause a fission reaction
The products of fission are two daughter nuclei and at least one neutron.
The neutrons released during fission go on to cause more fission reactions forming a chain reaction, where each fission goes on to cause at least one more fission.
what is the critical mass
minimum mass of fuel required to maintain a steady chain reaction
moderator
slows down the neutrons released in fission reactions to thermal speeds through elastic collisions between the nuclei of the moderator atoms and the fission neutrons.
The closer the moderator atoms are in size to a neutron, the larger the proportion of momentum which is transferred, therefore the lower the number of collisions required to get the neutrons to thermal speeds
water is often used as a moderator as it contains hydrogen, also it’s inexpensive and not very reactive making it a good material for a moderator.
Graphite is also sometimes used
control rods
absorb neutrons in the reactor in order to control chain reactions
The height of the control rods in the nuclear reactor can be controlled to control the rate of fission to control the amount of energy produced
made of materials which absorb neutrons without undergoing fission such as boron and cadmium
coolant
absorbs the heat released during fission reactions in the core of the reactor
This heat is then used to make steam which powers electricity-generating turbines
Sometimes water is both the coolant and moderator as it has a high specific heat capacity meaning it can transfer large amounts of thermal energy.
Other materials such as molten salt or gas (e.g helium) can be used as a coolant
safety aspects of nuclear reactors
fuel rods lowered in remotely to reduce worker’s exposure
very thick concrete shielding, which blocks radiation from escaping from the reactor and affecting the workers in the power station
how is waste from nuclear reactors dealt with
high level waste (spent fuel rods) and low level waste
1. The waste is removed and handled remotely, so that exposure is limited.
2. Any material removed from the reactor will be extremely hot due to fission reactions occurring within the reactor, so they must be placed in cooling ponds for up to a year while they may still be producing heat due to radioactive emissions. Cooling ponds are usually on the same site as the reactor or very close by so that these materials do not have to be transported through long distances, which will increase the risk of exposure.
3. At this point any plutonium or usable uranium is removed from spent fuel rods in order to be recycled.
4. The waste is then vitrified (encased in glass) and placed in thick steel casks and stored in deep caverns in geologically stable locations, so that there is no chance of the waste coming free of its casing. Locations to store radioactive waste are chosen so that they make a minimal impact on the environment and people living in the area are consulted about it beforehand.
exam q: explain why the distance of closest approach provides an estimate for nuclear radius
depends on the energy of alpha particle
recoil of nucleus is ignored
alpha particle has finite size