Unit 1 - Chemistry of Life
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
What defines an element? Essentially, this “thing” determines what element you have (like gold).
The amount of PROTONS you have.
e.g., helium has 2 protons and if helium somehow got another proton, it would become lithium.
What’s the difference between carbon-12 and carbon-13?
Carbon-12 has 6 neutrons (and 6 protons), while carbon-13 has 7 neutrons (and 6 protons)
If you change the amount of protons, you have an entirely new element.
What are the four most common elements common to all living organisms?
Oxygen, carbon, hydorgen, and nitrogen.
What is the mass of a single proton and neutron?
1.67 × 10-24 grams
OR
1 amu (atomic mass unit)
Why is carbon, of all elements, so useful for life? Why are we saying carbon-based lifeforms?
It’s because of its ability to form so many bonds with other structures. Think of it with hydrogen. With its four valence electrons, carbon can create four covalent bonds, creating methane (literally methane), and long carbon-chains.
Additionally, it’s desire to fulfill the octet rule still applies. It needs to find four more electrons to fill its outer shell.
What kind of shape does carbon with four covalent bonds form? Think AP Chemistry!
It is tetrahedral. Imagine a pyramid with a triangular base and at each corner is a hydrogen. The center contains the carbon.
Edit this card to see a picture!
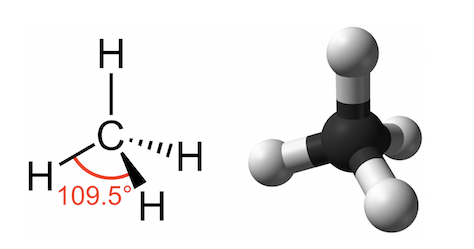
What are hydrocarbons? What is the advantage of them?
These are organic (cuz carbond) molecules that consists of only carbon and hydrogen.
They are useful because they can store a lot of energy, which is released when they are oxidized.
What are functional groups?
These are specific groups of atoms (usually branching off of hydrocarbons) that influence the reactivity/properties of organic compounds.
What is the hydroxyl functional group? Why does it bind?
When it is attached to a carbon-backbond of a hydrocarbon, it turns it into an alcohol.
The hydroxyl group will also make something polar, because of that electronegative bonding between the O and the H.
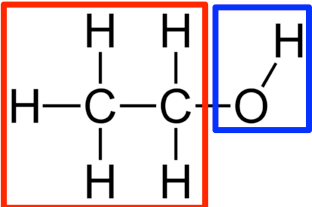
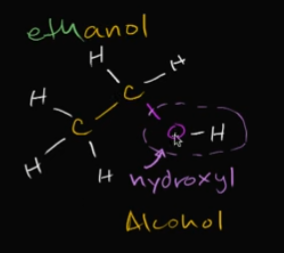
Let’s say we have a two-stringed hydrocarbon (two carbon chains) and one of the backbone carbons binds with a hydroxyl group. What does the molecule become name-wise? Why?
When we have a two-carboned chain, we call that eth
Then, since a hydroxyl group binded to one of those carbon-backbones, it became an alcohol, so… ethanol!
Edit the card to see the image!
What is a sulfhydryl group? How does this affect polarity?
It is very similar to a hydroxyl group, however, sulfur is LESS electronegative than oxygen, meaning that it’s less polar.
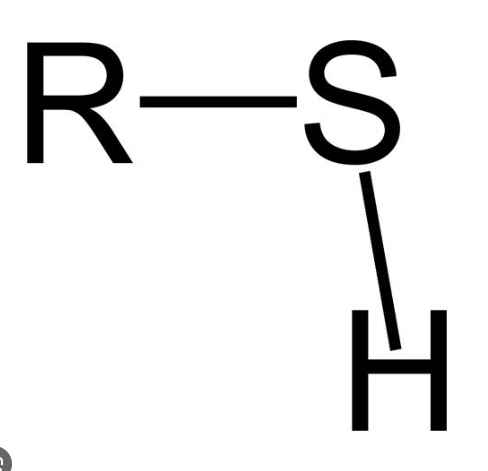
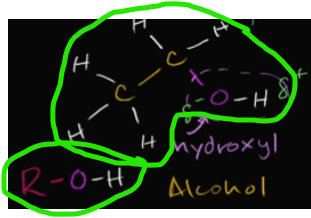
These two symbols are the exact same. What does R represent?
It simply represents the hydrocarbon chain with an OH (hydroxyl) group binding to one of the carbon atoms.
What is a carbonyl group?
This is when a single oxygen atom bonds to one of the carbons
What happens when there’s multiple groups on a single carbon, say a carbonyl group AND a hydroxyl group? Is it basic or acidic?
It becomes a carboxyl group, making the molecule acidic BECAUSE with both oxygen there, hogging the electrons, it kind of gets rid of that extra hydrogen from the hydroxyl group.
What is an amino group? Is it basic or acidic?
This is whenever NITROGEN is involved in a hydrocarbon. The nitrogen is usually has a lone pair (two valence electrons) to pick up more hydrogen (this means it’s BASIC)
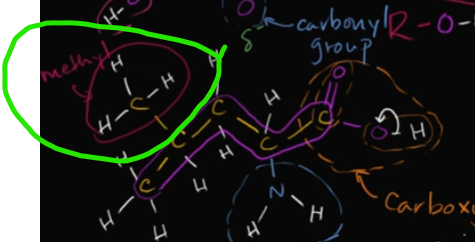
What is a methyl group?
It’s exactly what it sounds like! You have another hydrocarbon (this time methane which is one C bonded by 4 H) attaching to a backbone carbon.
Edit this card to view the image!
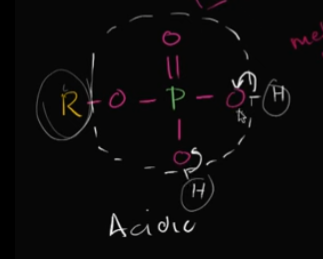
What is a phosphate group? Is it basic or acidic?
This is when you have phosphorus bonded to 4 oxygen and one of those oxygen is bonded with the hydrocarbon (carbon backbone).
It’s considered acidic because it can easily dump off hydrogen ions (if it has any bonded to the O’s).
Edit this card to view the image!
What makes a functional group able to dissolve in water? Hint: AP chemistry reference!
It’s whether or not it’s polar. If it’s polar, then it will dissolve!
think: like dissolves like!
What elements do Nucleic Acids, Proteins, Carbohydrates, and Lipids contain? (besides hydrogen and carbon)
Nucleic Acids: Oxygen, Nitrogen, Phosphorus
Proteins: Oxygen, Nitrogen, Sulfur
Carbohydrates: Oxygen
Lipids: Oxygen
What is an electron transfer?
This is when one atom LOSES an electron and another one GAINS an electron.
EX: Sodium and Chlorine!
The reason ionic bonds are important is because they can create electrolytes. Discuss some examples of electrolytes AND why they’re important for life.
Some popular electrolytes include sodium, potassium, and calcium. They’re important because they help nerve impulse conduction, muscle contractions, and water balance.
What is a polar covalent bond?
This is when electrons in a covalent bond spend more time with the more electronegative atom, creating a partially positive and negative end (hence polarity).
Think of water (oxygen pulls the electrons from the H to make it more negative).
As another justification of polarity, we know the bonds between water are polar, what about its MOLECULAR SHAPE?
It’s bent, meaning that you can clearly see a positive and negative end forming.
In terms of the four large biological macromolecules, tell me (in broad terms) what each one does…
Proteins
structural support or specific jobs, like catalyzing metabolic reactions or receiving/ transmitting signals.
Lipids
Long term energy, insulate, and form cell membranes
Carbohydrates
Quick energy and structure (like the cell wall for plants)
Nucleic Acids
Stores and transfers hereditary information, like how to MAKE proteins.
What actually makes up a macromolecule? Specifically, why can we call carbohydrates, proteins, nucleic acids, (not really lipids; I’ll explain in the card) macromolecules?
It is because they’re all polymers—long chains made up of repeating molecular units (monomers).
Lipids are not usually considered a macromolecule because they generally DON’T form polymers.
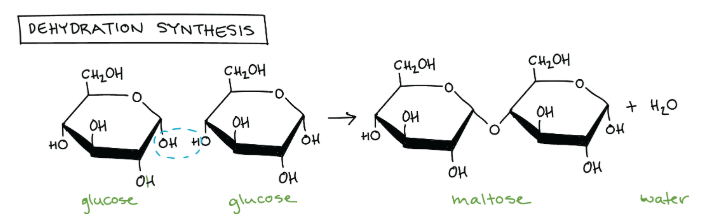
How do you build polymers from monomers?
Where does the bond typically occur?
This usually happens through dehydration synthesis, when one monomer forms a covalent bond to another monomer, releasing a water molecule.
The bond typically occurs from the hydroxyl group of one monomer with a hydrogen from another monomer.
Edit this card to see the image! The blue circle is everything that is lost.
Is it possible for a polymer to contain a ton of different monomers? Give some examples!
Absolutely. This is seen all throughout carbohydrates, proteins, and nucleic acids.
EX: there are four types of nucleotide monomers in DNA
How do polymers turn back into monomers? First, explain why this would ever happen in the first place.
The reason this happens is to recycle one molecule to build another one.
The reason polymers turn back is because of hydrolysis reactions, which involves breaking a bond by an additional, outside water molecule.
What is the end result of a hydrolysis reaction (hint: don’t just say that you have multiple monomers C:)?
At the end, you are left with one monomer with an additional hydroxyl (-OH) group and another one with an additional hydrogen atom.
Edit this card to see the image!
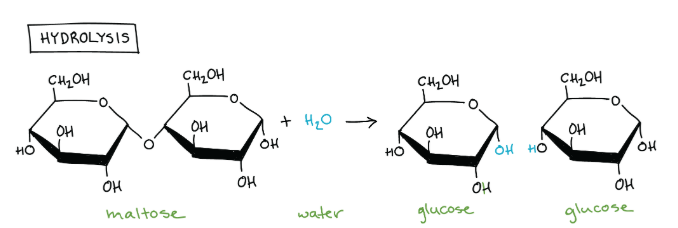
Dehydration synthesis (require/don’t require) energy, while hydrolysis reactions (require/don’t require) energy.
require; don’t require
Dehydration synthesis requires energy to build up molecules. Hydrolysis reactions break down big molecules, releasing energy.
How might enzymes be useful in both dehydration synthesis and hydrolysis?
In both cases, they catalyse (speed up) the reaction.
What is normally true about enzymes that end in -ase? What enzyme breaks down lipids? Proteins?
They usually break bones. For example, maltase enzyme breaks down maltose. Lipase breaks down lipids. Peptidase breaks down proteins .
peptidase is also called polypeptide!
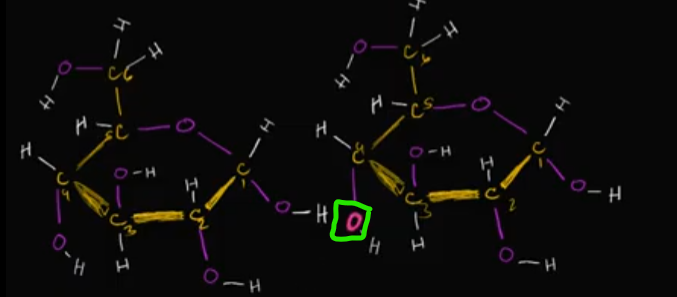
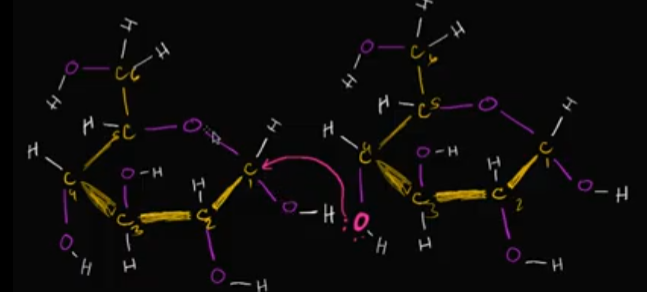
If we’re looking at these two glucose molecules, specifically, that oxygen circled in with neon green, where might it bond with that second glucose molecule? Why?
ALLLLLLLLLLLSO, what’s the end result of this besides just a bigger macromolecule?
It’ll bond with the carbon that already has two oxygens bonded to it. This is because of the electronegative-ness that entire bond has, making it easy to attract one of the TWO lone pairs on that neon green oxygen!
Edit this card to see the image of where it could bond!
The END RESULT is a water molecule floating around!
What is a monosaccharide?
It is a single sugar (the simplest form of a carbohydrate)! Think of a single water molecule—same thing but for glucose!
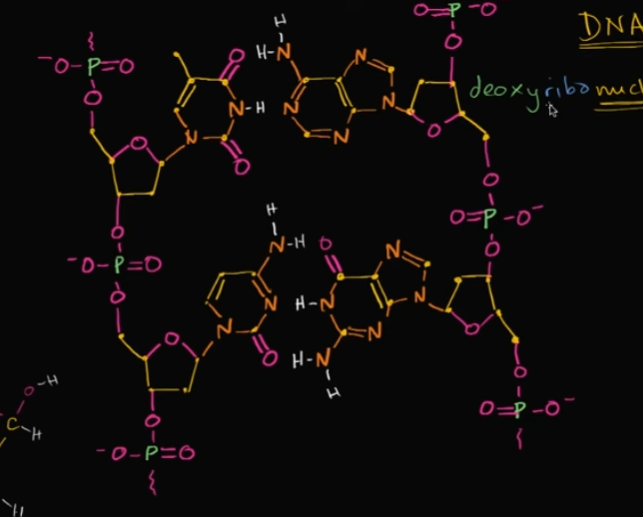
In this image, how many nucleotides are pictured?
Four! Look at the image!
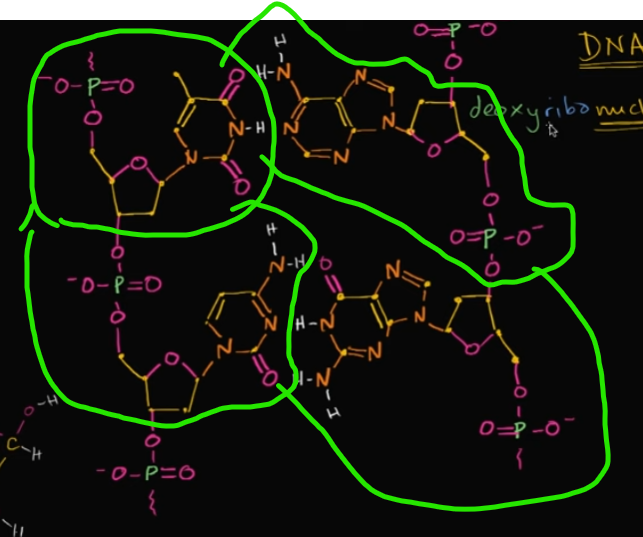
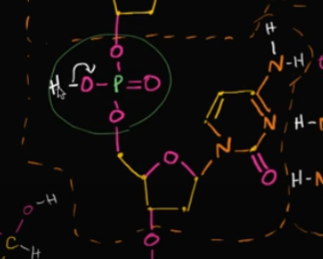
What actually makes DNA (deoxyribonucleic ACID) an acid?
Well, it’s because of the phosphate groups that are each in a nucleotide. In each phosphate group, there’s bonded 4 oxygen, which are VERY electronegative.
When it’s in a solution, those oxygen want to gain all of those electrons, making it LOSE a proton (an H) to become acidic.
Edit this card to view an image! In an original setting, there’s an H (without a solution!)
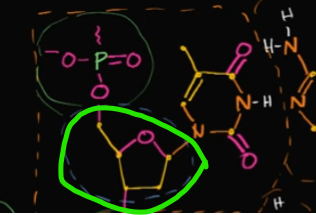
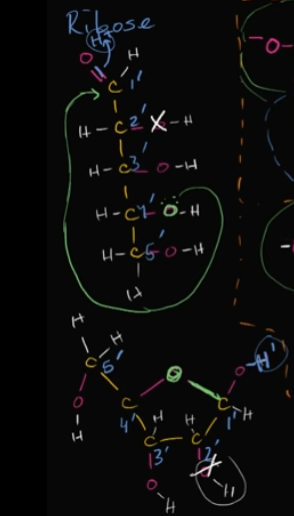
What is this called?
Explain how it’s formed like this in DNA
This is ribose, a sugar.
It takes this form in DNA when the oxygen in the fourth carbon (think of it as a five-carbon chain) bonds with the first-prime carbon that’s attached to a double bond with an oxygen.
When this bond forms, it allows the original, double-bonded oxygen, to form a bond with a hydrogen, and thus forms that shape.
Edit this card to see what I’m talking about 🙂
What are the four nitrogenous bases in DNA called? Tell me which one bonds to what 🙂
Also, discuss HOW they bond!
Adenine, Thymine, Cytosine, and Guanine.
A with T
C to G
They’re bonded very simply—through hydrogen bonds! If you look at the nitrogen bonded with two hydrogen, it forms hydrogen bonds with that double-bonded oxygen.
Edit this card to see what I’m talking about C:
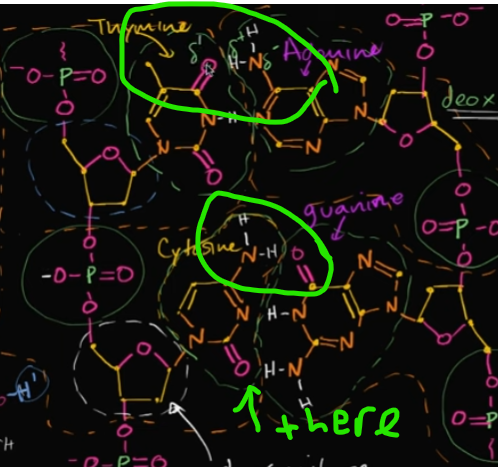
How would you actually describe the direction of a DNA sequence? We know there’s two strings, but they’re flipped compared to each other.
What kind of structure is this called?
Well, we would base it on the deoxy-ribose sugar we saw. Essentially, we’re going from 5th-prime carbon to 3rd-prime carbon (or vice versa). Look at the image:
The left strand goes from 5 to 3.
this is called an ANTI-parallel structure!
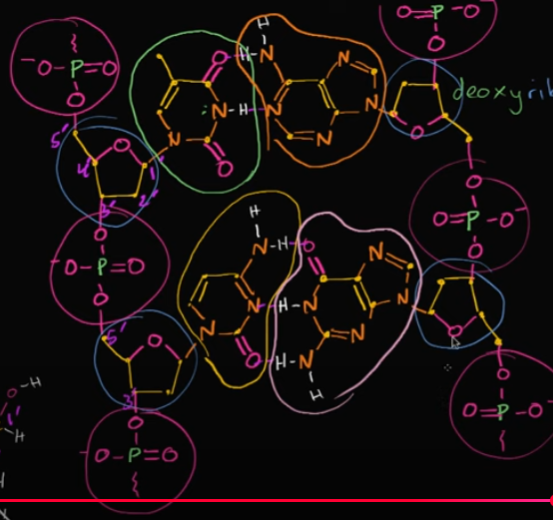
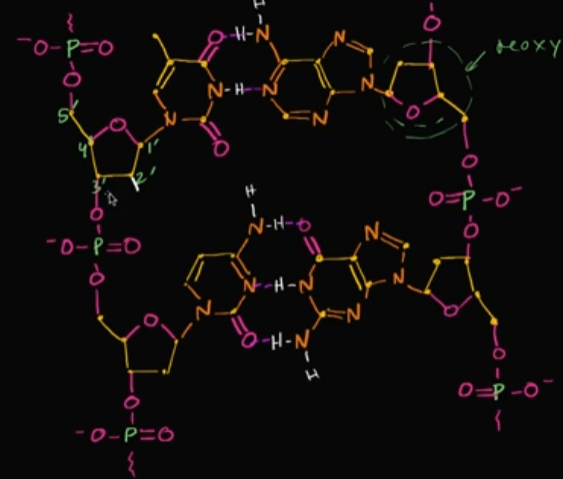
Let’s say we wanted this DNA to become RNA (or at least one part of it, since RNA is single-stranded). How would we even do that?
Well, all you would need to do is turn the deoxyribose into ribose by adding a hydroxyl group to the 2nd-prime carbon chain. You would want to do this on all the sugars on the backbone of one chain.
What are the nitrogenous groups in RNA?
Adeine, Guanine, Cytosine, and URACIL! (not thymine!)
What is the difference, in terms of the structure, between uracil and thymine?
Uracil just doesn’t have the methyl group that thymine once had.
Look at the image! That shows uracil. If there was a line (methyl group) where that cursor was, then it’d be thymine!
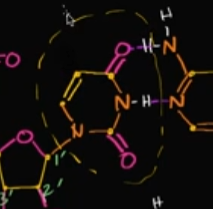
What is mRNA?
This is messenger RNA. It essentially carries genetic information from DNA to the ribosome, where proteins are synthesized.
What is tRNA?
This is transfer RNA. It essentially transfers amino acids to the ribosome during protein synthesis.
What is rRNA?
This is within the ribosomes and helps to assemble the amino acids into proteins.
What is the really basic pathway for protein creation?
DNA is created, then transcripted into mRNA, then translated where tRNA and amino acids create the protein.
In many proteins, which are created from amino acids, you have a commonality among each amino acid. What is it?
Additionally, what actually makes it an acid?
In every amino acid, you have an amino group, which is a nitrogen bonded with two hydrogen that’s bonded with a carbon-base. You also have a carboxyl group!
Edit this card to see what this looks like.
The thing that makes it an acid comes from the carboxyl group, aka where the most oxygen are because of their electronegativity. It likes to donate the proton on one of those oxygen.
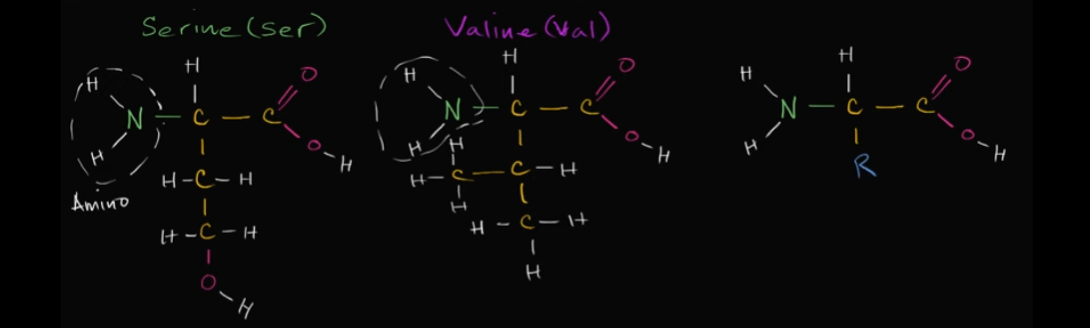
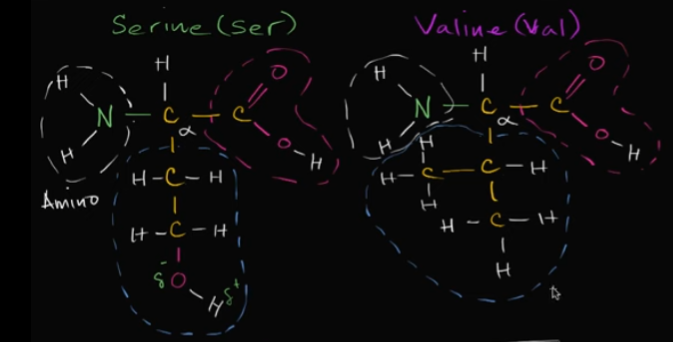
Based on these two amino acids, which one would be hydrophilic?
That’s easy! It would be serine because of that alcohol group at the bottom. It has an oxygen and a hydrogen, so it’s polar! It’s going to dissolve in water (it’s also polar!).
How do amino acids connect to each other? One amino acid doesn’t make up an entire protein 🙂
They end up bonding to each other via peptide bonds.
Multiple peptide bonds becomes a polypeptide!
What is the first structure of a protein called? What does it do?
The first structure is called the primary structure. Essentially, it is the sequence of the amino acids (or the order) that DNA put it in.
What is the second structure of a protein called? What does it do?
The second structure is called the secondary structure (haha). This is what allows a protein to be bent into its shape from all the interactions between the amino acids, like hydrogen bonding.
What is the third structure of a protein called? What does it do?
It is the tertiary structure! This is what allows the protein to gain its 3-D shape given the intearcatoins among the side chains (those R groups) in each amino acid.
What is the fourth structure of a protein called? What does it do?
It’s the quaternary structure. This is what actually arranges the protein based on the different peptide chains.
In hemoglobin for example, there were four peptide chains. Look at the image:

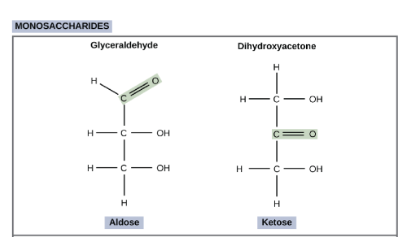
How do we categorize sugars (carbs)? (this is just the two groups!)
We can do this by looking at the different groups—aldehyde group and ketone group.
An aldehyde group (aldose) is present if the carbonyl C (C double bonded to O) is the last one in the chain.
A ketone group (ketose) is present if the carbonyl C is surrounded by other carbons on both sides of it.
Look at the image.
How else can we categorize sugars, besides those two major groups 👀
Look at the number of carbons! Some of the most common include: trioses (three carbons), pentoses, and hexoses.
Look at this card!
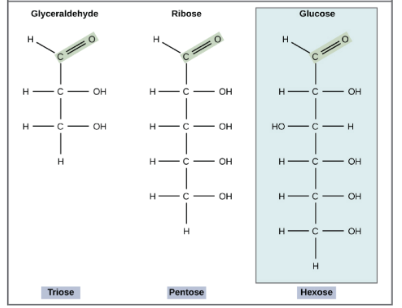
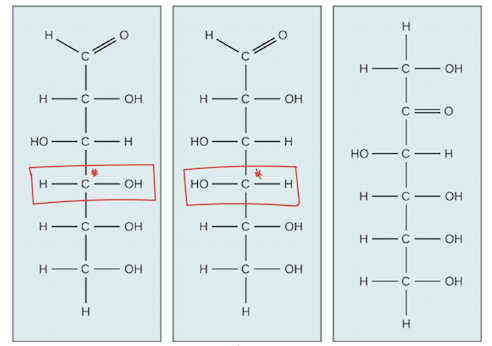
From this image, identify each isomer…
Leftmost: glucose
Middle: galactose
Rightmost: fructose
Notice glucose and galactose. Galactose will always have a H in the same direction as the double bonded oxygen aldose group.
When two sugars combine (aka two monomers) via a _________ reaction, what type of bond forms?
dehydration; a glycosidic bond forms!
Why is glycerol often referred to as a sugar alcohol?
It’s because of the three hydroxyl (-OH) groups it has! aka, this is what makes it an alcohol!
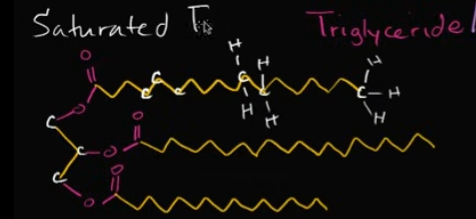
What does a saturated fat look like chemically?
This is when you see only single bonds between the carbons and for the rest, you have hydrogens. You’re essentially fitting on as many hydrogens as possible, aka saturating it!
Look at the image!
What does an unsaturated fat look like? What about one with multiple double bonds (here is your strong hint)?
This is when you have a triglyceride with at least one double bond between the carbons. This makes the fat kind of kink-up or change its shape.
A poly-unsaturated fat contains MULTIPLE double bonds with the carbons, and forms an even more kinked shape.
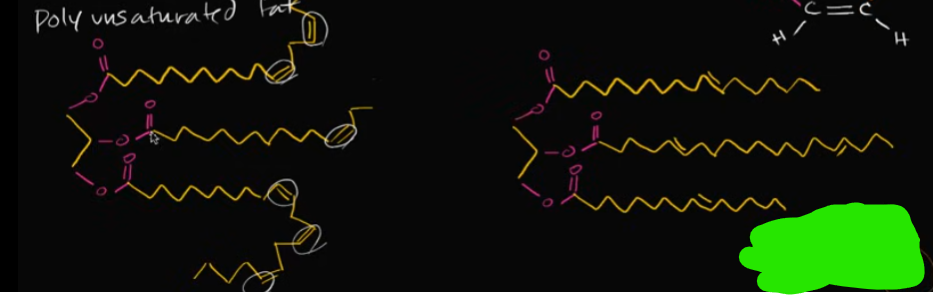
Why is it that these two structures are different in their shape, but one of them is still linear and NOT an unsaturated fat?
This all comes down to the structure of a double bonded carbon. A cis-bonded carbon will actually produce those kinks, while a trans-bonded carbon won’t.
The carbon chains in that right image are flipped after a double-bonded carbon. This is how we get TRANS-FATS!!!!
When you have joining amino acids to create a polypeptide chain, where do incoming amino acids attach to? Essentially, what’s the starting point of an amino acid where a dehydration reaction can occur?
It will always start at the -NH2 (amino group). It will end at the carboxyl group (-COOH)
in the image, the carboxyl group will attach to the amino group!
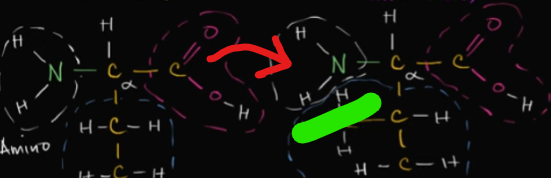
What are chaperonins?
These are special proteins that often help fold a polypeptide into its 3D-shape during the tertiary stage.
The phosphate group in nucleotides is always attached to the ____ carbon in the sugar, and the ____ carbon always has a hydroxyl group which new nucleotides may be added.
5-prime; 3-prime