GCSE Physics: Atomic Structure, Radiation, and Nuclear Fission & Fusion
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
What is the structure of an atom?
An atom consists of a positively charged nucleus containing protons and neutrons, surrounded by negatively charged electrons.
What is the relative mass and charge of a proton?
Relative mass: 1, Relative charge: +1
What is the relative mass and charge of a neutron?
Relative mass: 1, Relative charge: 0
What is the relative mass and charge of an electron?
Relative mass: 0 (0.0005), Relative charge: -1
What is the typical radius of an atom?
1 × 10−10 metres
What is the radius of the nucleus compared to the atom?
The nucleus is 10,000 times smaller than the atom.
What are isotopes?
Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
Give an example of isotopes of carbon.
Carbon-12, Carbon-13, and Carbon-14.
What happens when electrons absorb EM radiation?
Electrons move to a higher orbit, further from the nucleus.
What happens when electrons emit EM radiation?
Electrons fall to a lower orbit, closer to the nucleus.
What did Dalton propose about atoms in 1800?
Dalton proposed that everything was made of tiny spheres (atoms) that could not be divided.
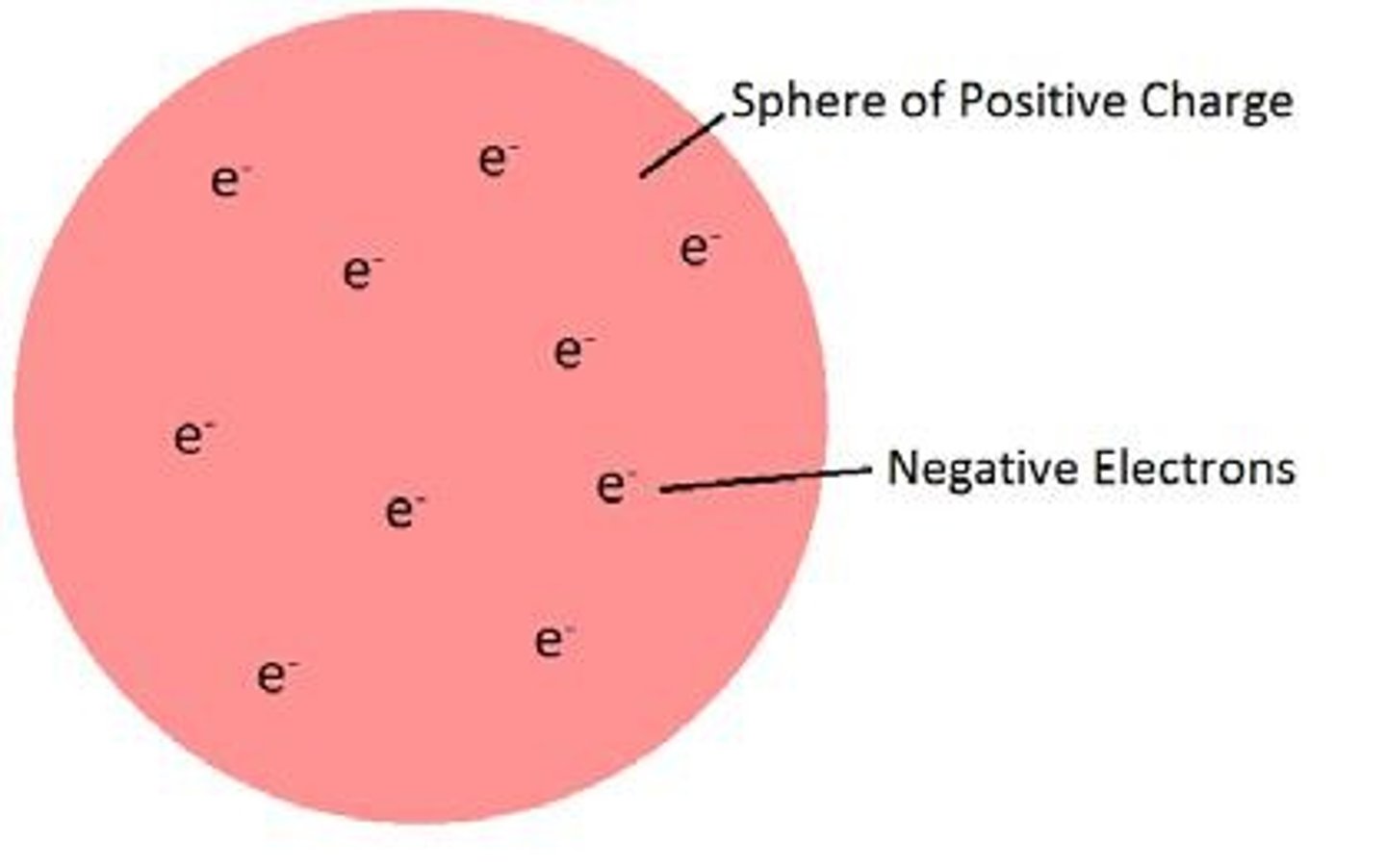
What was the Plum Pudding Model?
The model proposed by JJ Thomson where negative electrons were dispersed through a positive 'pudding' to cancel out the charges.
What did Rutherford discover about the atom?
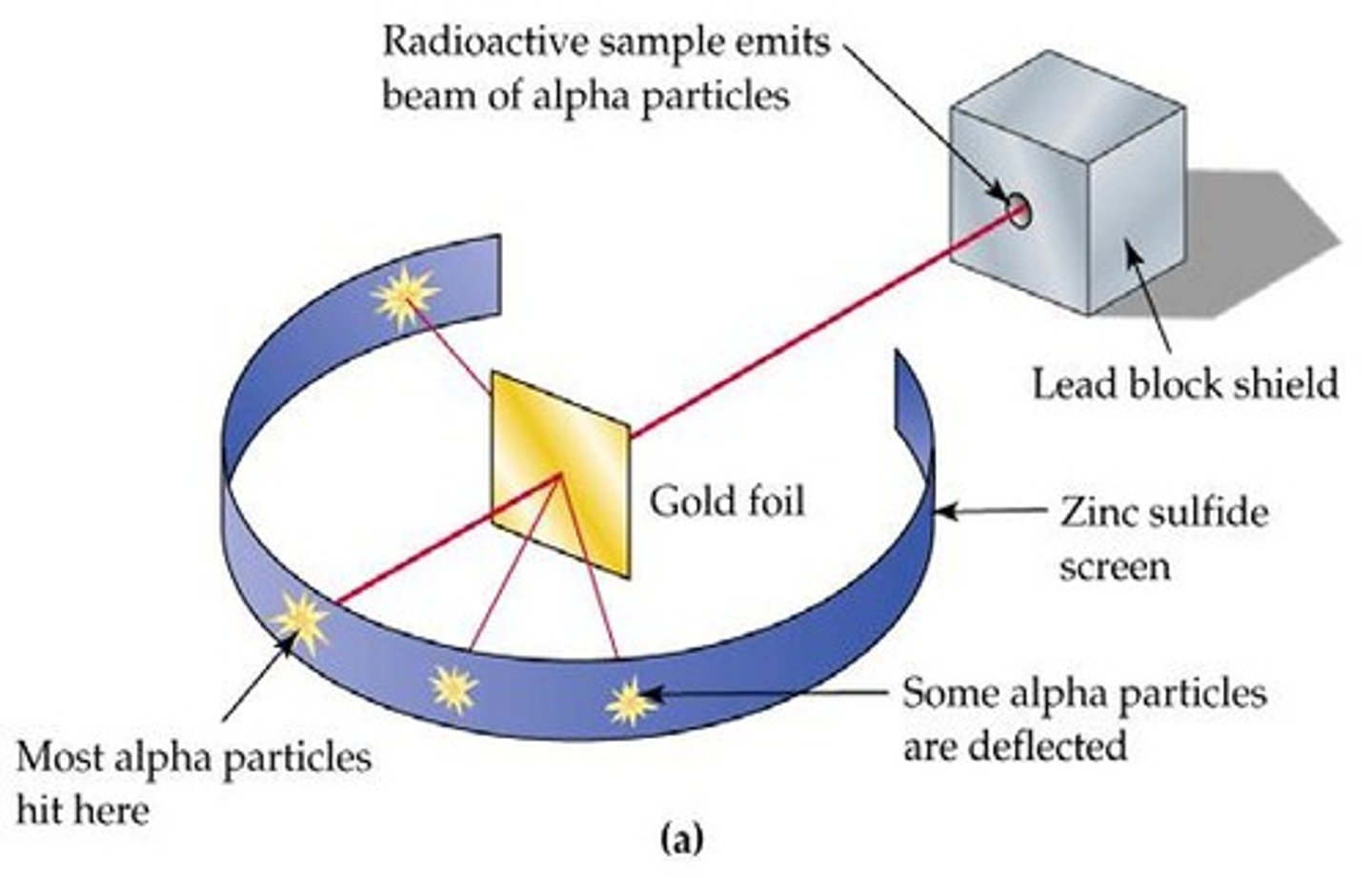
Rutherford discovered that most of the atom is empty space and that the nucleus is charged.
What is the Gold Foil Experiment?
An experiment by Rutherford showing that most alpha particles passed through gold foil, indicating empty space in the atom.
What is the significance of the Bohr model?
Bohr proposed that electrons exist in fixed 'orbitals' around the nucleus, preventing the atom from collapsing.
What is radioactive decay?
A random process where unstable atomic nuclei emit radiation to become more stable.
What is activity in the context of radioactive decay?
Activity is the rate at which a source of unstable nuclei decays, measured in Becquerel (Bq).
What are the three types of nuclear radiation?
Alpha (α), Beta (β), and Gamma (γ) radiation.
What is alpha decay?
Alpha decay causes both the mass and charge of the nucleus to decrease.
What is beta decay?
Beta decay does not change the mass of the nucleus but increases its charge.
What is gamma decay?
Gamma decay does not cause any change in mass or charge of the nucleus.
What is half-life?
The time taken for half the nuclei in a sample to decay or for the activity to decay by half.
What is the half-life of the radioactive source mentioned in the notes?
5 minutes.
What is a characteristic of a short half-life radioactive source?
It presents less long-term risk as it quickly becomes less radioactive.
What is a characteristic of a long half-life radioactive source?
It remains weakly radioactive for a long period of time.
What is Americium used for?
It is used in smoke alarms and has a half-life of 432 years.
How does Americium function in smoke alarms?
It emits alpha particles, which decrease in number when smoke is present, triggering the alarm.
What is radioactive contamination?
The unwanted presence of radioactive atoms on other materials, posing a hazard due to decay.
What is irradiation?
The exposure of an object to nuclear radiation without making it radioactive.
What is background radiation?
Weak radiation detectable from natural or external sources, such as cosmic rays and nuclear fallout.
What unit is used to measure radiation dose?
Sieverts (Sv).
What is Technetium used for in medicine?
As a medical tracer with a half-life of 6 hours, decaying into a safe isotope.
How does chemotherapy utilize radiation?
Gamma emitters target cancerous cells, but may also affect surrounding healthy cells.
What is nuclear fission?
The splitting of a large and unstable nucleus, such as uranium or plutonium, releasing energy.
What initiates nuclear fission?
The absorption of a neutron by an unstable nucleus.
What is a chain reaction in nuclear fission?
A process where one fission event causes further fission events, releasing more energy.
What is nuclear fusion?
The process of two small nuclei fusing to form a heavier nucleus, releasing a large amount of energy.
Why is fusion considered more efficient than fission?
It produces more energy and has less long-term waste, but is not yet feasible for practical energy production on Earth.
What happens to the mass during nuclear fusion?
Some mass is converted into energy, which is released as radiation.
What is the role of peer review in scientific reports on radiation effects?
It ensures accuracy in measurements to prevent unsafe safety levels based on flawed studies.
What is the effect of background radiation on radiation dose?
It can vary based on occupation and location.
What is the significance of the decay of radioactive atoms in contamination?
It releases radiation, which poses health hazards.
What is the difference between contamination and irradiation?
Contamination involves radioactive atoms on materials, while irradiation involves exposure to radiation without making the object radioactive.
What is the impact of a controlled chain reaction?
It allows for the safe release of energy, unlike an uncontrolled reaction which can lead to an explosion.
What is the significance of the neutron in nuclear fission?
It provides the necessary energy to initiate the splitting of the nucleus.
How does the half-life of a radioactive source affect its use in medical applications?
A short half-life minimizes harm while allowing sufficient time for detection in the body.