Raman Spectroscopy
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
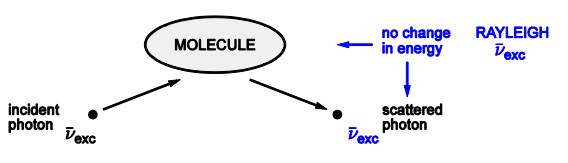
Describe Rayleigh light scattering
A collision between a photon and a molecule can result in scattering which is elastic
Being elastic means it has no change in energy of the photon or the molecule
Weak
Provide a diagram detailing rayleigh light scattering
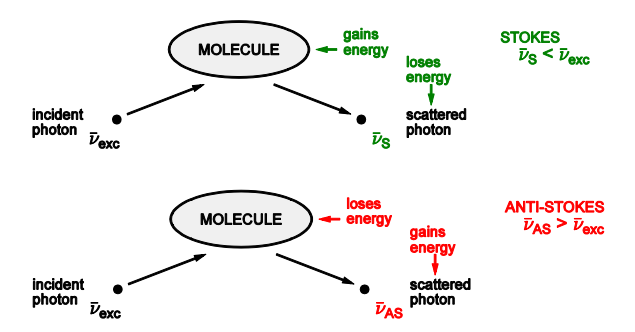
Describe Raman light scattering
A collision between a photon and a molecule can result in scattering which is inelastic
Energy is exchanged between the photon and the molecule
Very weak
Draw diagrams for the two types of raman light scattering naming each
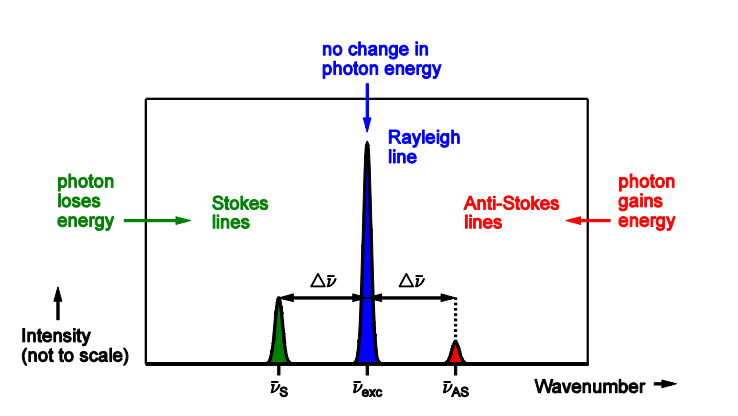
draw a graph of intensity against wavenumber
Give equations for stokes and anti-stokes

What is scattering intensity proportional too

Give the gross selection rule for Raman scattering
Raman scattering must have a change in polarizability on :
Rotational Raman
Vibrational Raman
What is polarizability
Polarizability is the ease with which the electrons within a molecule can be moved by an applied electric field.
draw the polarizability ellipsoid and describe what it entails

what is an anisotropic ellipsoid
Anisotropic ellipsoid : different in different directions
Changes as the molecule rotates
Rotational Raman transition is allowed
What is an isotropic ellipsoid
Isotropic ellipsoid : same in all directions
Applies only to spherical rotors
Doesn’t change when rotating
Describe rotational raman spectroscopy
Molecules change their rotational energy by inelastic light scattering
Rotational energy levels are the same as described prior and depend only on the molecule
Describe specific selection rule for rotational raman spectroscopy
Specific selection rule
Delta J = 0, +- 2 (in diatomic molecules
Different from absorption and emission
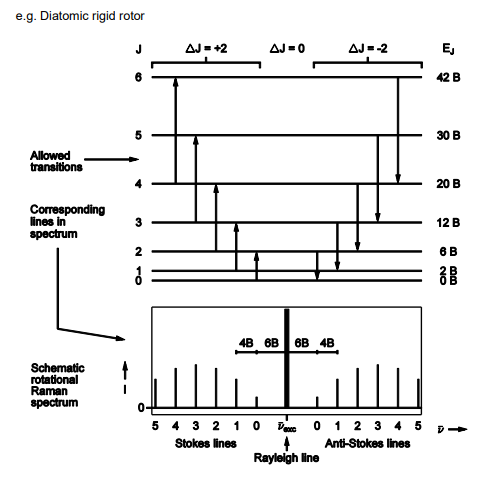
Describe and draw the rotational raman spectroscopy graph
Range of J levels populated and as such spectrum has several lines
Spacing between the line are different from what they were in microwaves
On the stokes lines molecule gain energy on the Anti-stokes they lose energy.
Describe Vibrational Raman Spectroscopy
A molecule changes its vibrational energy by inelastic light scattering
Vibrational energy levels
Describe Specific selection rule for Vibrational Raman Spectroscopy
Specific selection rule
Delta v = +- 1 (Harmonic oscillator)
Same as for absorption and emission
Describe Vibrational Raman Spectrum
Spectrum
Most molecules in v = 0
One stokes band in spectrum
One anti-stokes band (v = 1-> 0) generally not observed
generally reported and plotted as a change in wavenumber
Δ v ~ = v ~ exc – v ~ stokes (Sometimes called the Raman shift)
showing only the Stokes region (energy gained by molecule) to resemble an IR spectrum
What is the mutual exclusion rule
Molecule has a centre of inversion, then it is Raman active and IR inactive.
Vice versa
What happens if a molecule has no center of inversion
If a molecule does not have a center of inversion then some or all modes may be both Raman and IR active
What are band positions determined by for raman vibrations
Bands Positions are determined by the molecule via its energy levels
What are band strengths determined by for raman vibrations
Band strengths are determined by the spectroscopic technique via its selection rules