Chemistry Exam 3
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Boyles law is pressure and volume is inversely proportional, True or False
True
Boyles law is also that volume and temperature are not inversely proportional, True or False
False, in Boyles Law volume and temperature are directly proportional
In Avogadro’s Law volume and number of moles are
Directly proportional
In Gay-Lussac Law pressure and temperature are
Directly proportional
What is the ideal gas law? Are the other gas laws direvied from the ideal gas law?
Yes, the ideal gas law is Gas molecules do not attract or repel each other, The particles themselves are considered infinitely small, taking up zero volume compared to the space they inhabit( no volume or intermolecular forces)
PV= nRT
P= pressure ( atm)
V= volume (L)
N= mol
R= universal gas constant
R = 0.08206> atm / 62.36> torr
Charles Law is temp and volume are
Directly proportional
What is the kinetic molecular theory?
Constant Motion: Particles move rapidly and randomly.
Negligible Volume: Gas particles are small, with vast empty space between them.
No Intermolecular Forces: Particles do not attract or repel.
Elastic Collisions: Collisions conserve kinetic energy and momentum.
Temperature & Energy: kinetic energy is directly proportional to Kelvin/temperature
Explaining Gas Laws with Kinetic molecular theory
Boyles law: less space= more frequent collusions
Charles law: Increasing temperature increases particle kinetic energy (they move faster). To keep pressure constant, the particles must spread out, creating a larger volume so that the faster particles do not hit the walls more frequently.
Gay-Lussc: high temp = Faster/Harder collisions
Avagrodo: Adding more particles (moles) to a container means more particles are hitting the walls. To keep the pressure the same, the volume must increase to reduce the collision frequency.
Something you need to know when it comes to Kinetic molecular theory….
The Velocity of gas particles are inversely proportional to square root of molecular weight.So, Heavier gas molecules move slower!
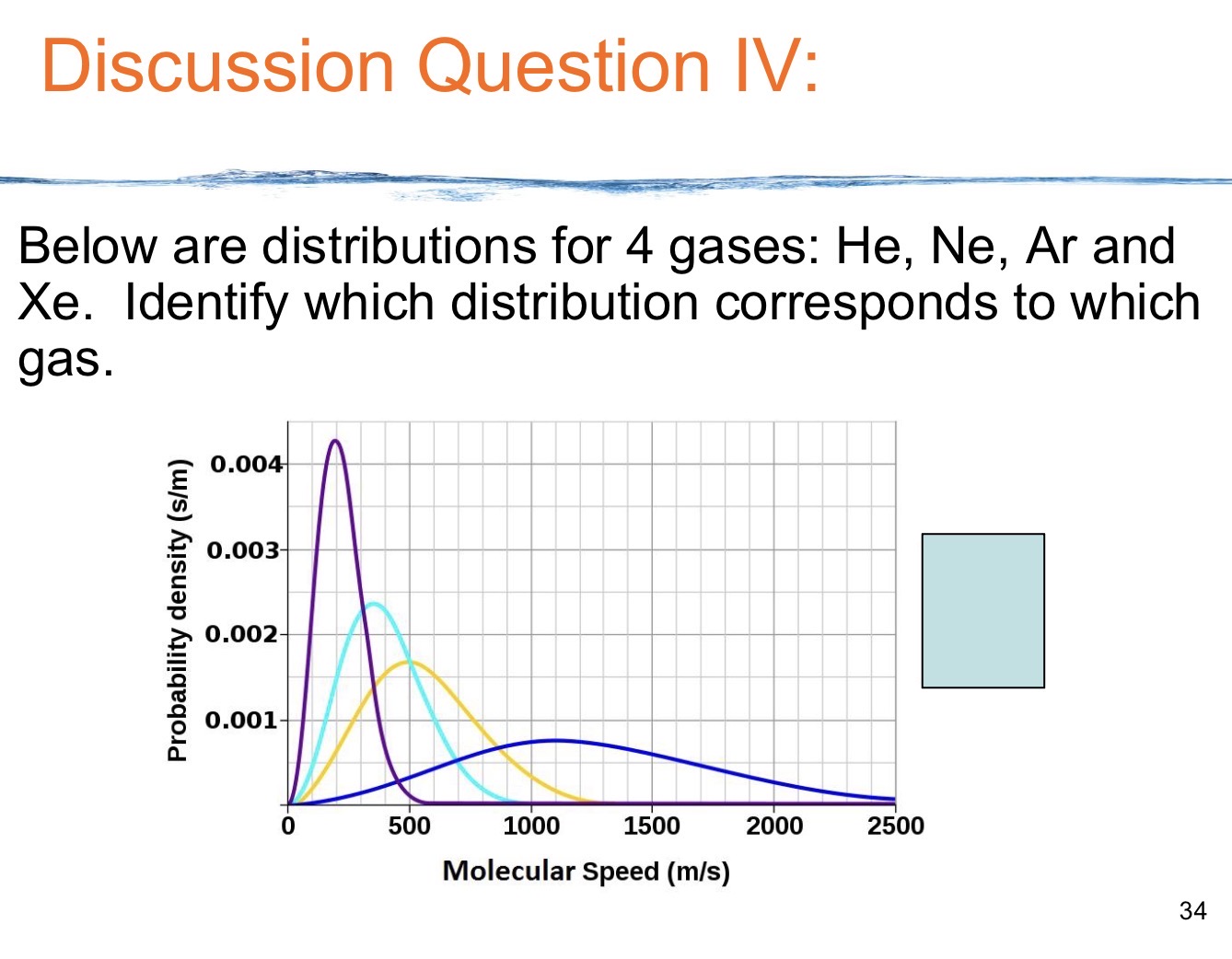
Explain velocity of gas particles distribution.( The Maxwell Boltzmann Curve)
gas molecules in a container do not all move at the same speed; instead, they have a wide range of speeds, with most particles moving near an average speed, while some move very fast and others very slow.
This distribution is usually visualized as an asymmetric "bell curve" where the x-axis is molecular speed and the y-axis is the number of molecules.
temp and mass dependent
Temp: As temperature increases, the average speed increases.
Mass: Lighter molecules (like Helium) move faster and have a broader distribution of speeds, while heavier molecules (like Xenon) move slower and have a narrower distribution, even at the same temperature.
Explain how temperature effects kinetic energy(the velocity of particles)
Higher temp makes gas particles move faster
Diffusion definition
The process where gas molecules spread out, moving from high to low concentration areas
Effusion definition
The process in which gas molecules escape from a container through a tiny hole into a evacuated space
What to know about the Rate of effusion/difussion and velocity
The rate of effusion and diffusion depend on molecular velocity, which is directly related to the gas's temperature and inversely related to its molar mass( temp and gas velocity directly proportional) lighter gas have a high rms velocities, lighter gases move faster
Graham’s Law:
The rate of effusion/diffusion is inversely proportional to the square root of the molar mass of gas
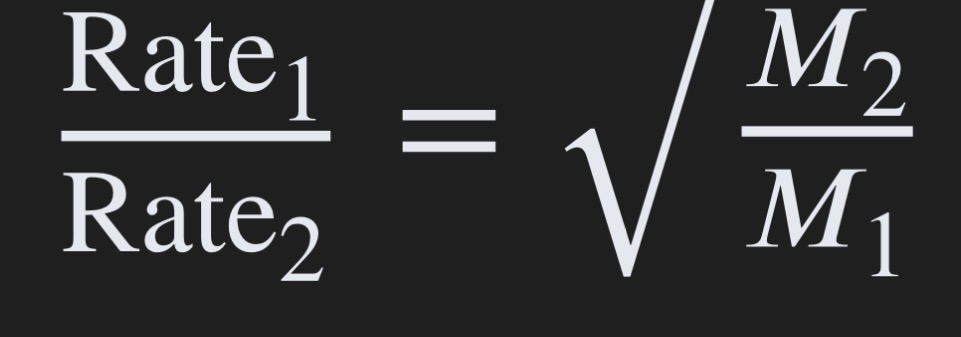
Memorize the Van der Waal equation
(P + n² a/ V² ) (V—nb) =nRT
Constants: a and b
A: intermolecular attraction
B: volume of gas molecules
At large volumes a and b are relatively small and van der Waal’s equation reduces to ideal gas law at high temp and low pressures
Conditions that cause deviations from ideality(real gases)
when these assumptions are not true: ideal gas particles are assumed to have no volume
Ideal particles are assumed to have no interactions with each other
At low temp and high pressures b/c molecules are forced closer together, making their finite volume significant and allowing intermolecular attractions to become influentia
Arrhenius definition of acid-base reactions
acid: H+ ions in aqueous solution
Base: OH- ions in aqueous solutions
Boyle’s Law equation
P1V1=P2V2
Charles Law equation
V1 /T1 =V2 /T2
Gay Lusaac’s Law equation
P1 / T1 = P2 / T2
Avogadro’s Law
V1 / n1 = V2/n 2
Combined Gas Law
P1V1 / T1 = P2V1/T2
T is kelvin
Vander Waal’s equation
P+ a(n/V)² x (V-nb)=nRT
Bronstead-Lowry definition
Acid: H+ donor
Base: H+ acceptor
Lewis definition of acid-base reactions
Acid: e- pair acceptor
Base: e- pair donor
In determine which elements is oxidated and reduced reactions H2O is
0 charge
Sigma vs. pi bonds
Sigma overlap on line
Pi don’t overlap over line
Same phase ( + + / - - ) bonding
Not different phases (+ - / - +) anti bonding