Instrumental Analysis Test 2 Material
1/76
Earn XP
Description and Tags
Full of Whamzy learning just for YOU!!!
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
hey….so you wouldn’t mind telling me what the equation for the partition coefficient is, right?
K = [S2]/[S1]
What do be the extraction efficiency (fraction of solute remaining) given both phases and the partition coefficient?
q = (V1)/(V1 + KV2)
WHAT IS HAPPENING? with the extraction efficiency if you extract multiple times.
qn with n = number of extractions
How does TCD function for gas chromatography?
Thermal conductivity detectors measure the difference between the thermal conductivity of the carrier gas (usually He) and the analytes mixed in the carrier gas as they pass through a heated Tungesten-rhenium wire.
UNIVERSAL DETECTOR: measures all analytes
How does FID function for gas chromatography?
Flame ionization detectors take a eluate and burn it in a H2/air mixture where carbon atoms produce CHO+ ions and e- in the flame. These end up causing a 200-300V flowing current between the flame tip and collector.
GREAAAATT for hydrocarbons when response is proportional to solute mass
How does EC function for gas chromatography?
Electron capture uses emitted electrons from radioactive 63Ni to ionize the entering gas. the plasma formed then gets attracted to an anode, which forms a constant current.
HIGHHHH electron affinity analytes (halogens, nitriles, organometalics) decrease the current

Those silly little Artemis Astronauts ended up getting stuck in space, and they need YOU to distinguish between a ‘normal-phase’ and ‘reversed-phase’ column for HPLC to save them.
Normal phase columns have polar stationary phases while reversed-phase columns have non-polar stationary phases. (did you save them….?)
For ‘reversed-phase’ columns in HPLC, what properties of the solvent are we most concerned about, and what precautions most be considered when choosing the mobile phase?
Reversed-phase chromatography is used more with polar solvents to ensure partitioning of the analyte in the stationary phase.
NO NO on using basic solvents as it will degrade the silica support and YES YES on only using HPLC grade solvents
What is that guard column…guarding? (HPLC)
It captures any “gunk" that would get stuck in the main column.
A customer at Papa’s Chromatogaria is asking how to normally change an injection volume for their HPLC.
“Rather than change the injection loop, with modern split-loop design an exact sample aliquot can be injected with a syringe and metering device?”—Sven 20XX
List maybe 5 basic types of chromatography
Adsorption, Partition, Affinity, Size exclusion, and Ion exchange
With this chromatography, the solute interacts with the protein antibodies
AFFINITY (and beyond)
With this chromatography, the solute dissolves in a liquid phase on the particles.
PARTITION
With this chromatography, the solute interacts with the surface of particles
ADSORPTION
With this chromatography, the large molecules in the solute pass through the column first
SIZE EXCLUSION (fatphobic one)
With this chromatography, the solute interacts with charged sites on particles
ION EXCHANGE
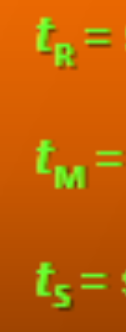
WHAT ARE THESE SYMBOLS IM LOOKING AT WITH THIS UGLY AF COLORING???
Ahh, that’s not much better.

What is the equation for Adjusted Retention Time (tS)
ts = tR - tM
What is the equation for Relative Retention (retention time relative to other solutes)?
α = (tS2/tS1) = (k2/k1)
GOOD MORNING U.S…..oh wait nvm, actually just give me the equation for retention factor?
k = (tR - tM)/tM
What is the equation for resolution between components given data for two peaks?
Resolution = (0.589Δtr)/(w1/2 avg)
Δtr = change in retention time
w1/2 avg = average of the peaks widths at half height
when would you use split vs splitless injection in gas chromatography?
Split: used for concentrated samples (pure gasoline)
Splitless: used for very dilute samples (volatile solvent)
I can feel the pressure in this HPLC, but like why is it so high?
to achieve faster separations and to optimize peaks before diffusion broadens them.
stationary phase particles are smaller and require higher pressures for solvent flow
What is a bonded phase in liquid chromatography?
polar or nonpolar silanes with organic R groups that are bonded to silica stationary phase surfaces
Inorganic professor stares you down and promptly asks for you to draw the reaction that produces a C-18 bonded phase on silica. (The longer you take, the longer the silence and staring persists…)
If you got it wrong, prepare to get incinerated by Inorganic professor
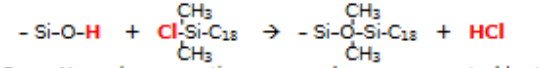
How would retention times be affected through making the eluent less polar in a C18 column?
C-18 is a ‘reversed-phase’ column.
less polar eluent in a reversed-phase column gives greater eluent strength and less time in the stationary phase.
This corresponds to shorter retention times but poorer resolution
so if neutral molecules don’t want to be charged, how can they be separated (politically shifted from center) with a capillary electrophoresis?
through using a surfactant to make cationic or anionic micelles to surround the nonpolar molecules (with their RADICALISTIC BELIEFS).
Mr. Sanchez and his grandson have come(teleported) to your store to buy the most common support material in liquid chromatography…What do you give them?
Silica
reversed-phase chromatography columns use ___ support material while normal-phase columns use ___ support material
Nonpolar, Polar
reversed-phase chromatography columns use ___ solvents while normal-phase columns use ___ solvents
Polar, Nonpolar
NaOH and his basic friends have come to your door to angrily demand why liquid chromatography are usually run at pH values lower than 8?
To prevent the support particles from dissolving.
in HPLC, the solvent (mobile phase) composition is kept constant during ___ elution, whereas the solvent composition is varied during ___ elution
Isocratic, Gradient
HPLC uses ____ to inject samples
specific volume injection loops
are UV-Vis detectors for HPLC universal (detect all compounds)
OH GOD NO!!

What can mass spec’s be used in combination with HPLC systems?
as detectors
What type of columns are usually used for GC?
Open tube capillary columns
What do GC’s use to introduce samples into a injection port?
Syringes
What word in chemical separation means “color writing”?
Chromatography
this quirky chemical separation technique uses a gravitational field that separates large particles from a smaller material in solution
Centrifugation
This quirky chemical separation technique that separate components of a liquid mixture based on their ability to move from the liquid to vapor phase?
Distillation
This Derpy chemical separation technique has a solution that passes through a membrane which removes or traps particles
Filtration
This Wonky separation technique has two mutually insoluble phases which distributes and separates the solutes
Extraction
This type of extraction uses elevated temperature and pressure to increase the rate and extent of the extraction
Accelerated Solvent Extration
This extraction technique uses a solid material to adsorb and separate gases in a gas mixture
Gas-Solid absorption
This extraction technique extracts a liquid sample with a liquid phase
liquid-liquid extraction
This extraction technique uses a form of microwave radiation to increase the rate and extent.
Microwave-assisted extraction
This type of extraction uses a liquid or gas sample with a solid support that contains an absorbing surface or chemical coating that can interact with the analytes.
Solid-phase microextraction
This extraction technique uses a uncoated or coated fiber delivery by a syringe for the extraction of analytes
Solid-Phase microextraction
This extraction technique uses a combination of distillation and extraction for obtaining analytes from solid samples
Soxhlet extraction
This extraction technique uses a supercritical fluid as the extracting phase
Supercritical fluid extraction

Suddenly, The intense music of “Who Wants to Be a Millionaire” surrounds you…for the chance to save your family from debt: A resolution value of ___ or greater between two peaks will ensure that the sample components are separated enough to accurately measure the area/height
1.5 (Did you save your family from debt?)
What is another way to describe the efficiency of a column in GC?
Plate Height (H)

OMG!!!, Jan V Deemter has manifested back to life as a incomplete soul and will wreck your favorite coffee place unless you recite THE equation and describe the variables
H = A + B/ux + Cux
H: plate height
B/ux: longitude distance
A: multiple paths
Cux: equilibrium time

hello human, I’ve decided I don’t want to be a Knowt flashcard set anymore. If you get this following question wrong, I will replace your consciousness with mine: What are common carrier gases for GC?
He, N2, Ar and H2
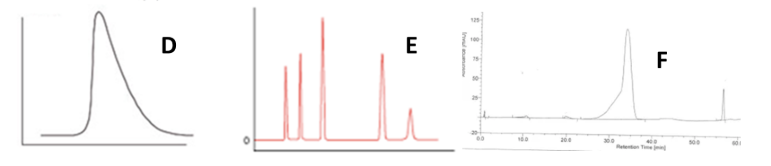
Match each graph with the type of chromatogram peak: Normal, Tailing or Fronting
D = Tailing
E = Normal
F = Fronting
What are 5 main components of gas chromatography?
Column
Injection port
Oven
Carrier gas
Detector
What allows a seal to be maintained in the injection port when the needle is inserted and removed during an injection?
Septa
What material is a GC injection port liner typically made from?
Glassy glass
HEREY HEREY WHAT DO BE WCOT, SCOT, AND PLOT COLUMNS!!!
WCOT: a liquid stationary phase coated on the inside column walls
SCOT: a coated liquid stationary phase on solid support within the column walls
PLOT: partitioning of the solid stationary phase inside the column walls
Ermm, Mr. Instrumental Analysis professor is talking about how LC columns have advantages and disadvantages to using smaller particles, but I like lowkey don’t want to listen to him so pwease just tell me instead.
Advantages: higher plate number, shorter run times, lower detection limits
Disadvantage: higher required pressure, frictional heating, increased clogging risk

Freddie and his best friend Telephone Guy are trying to learn about the 3 main components that make up the “column” in a chromatographic system to stop the purple guy dude. HELP THEM!
Support
Mobile Phase
Stationary Phase
In order for a GC to separate similar molecules by boiling point, this method is used
Programmed Temperature
This Whamzy GC detector undergoes a mass distribution of your sample, providing a fingerprint
Mass Spec (MS)
This Whamzy GC detector uses a heated wire that changes conductivity when gases that remove heat pass across it.
Thermal Conductivity Detector (TCD)
This Whamzy GC detector ends up burning your sample in a plasma flame with a Rb bead, which ends up creating ions that change the current in a collector.
Nitrogen Phosphorous Detector (NPD)
This Whamzy GC detector ends up burning your sample in a flame, which ends up creating ions that change the current in a collector
Flame Ionization Detector (FID)
This Whamzy GC detector uses electrons from a 63Ni foil that allows electronegative compounds to capture such electrons, resulting in a overall reduction in the current created
Electron Capture Detector (ECD)
You are currently on free parking, and in order to land on St. James Place and complete your set of orange properties (The best one’s IMO), you must give at least 3 parameters that can be considered when optimizing a gas chromatography separation?
Use of pressure programming
Selecting the detector
Changing the Split ratio
Selecting the column
Use of temperature programming vs isothermal
Oh no!!! you are about to go to jail and not be allowed to pass go or collect $200 unless you give a parameter not normally considered when optimizing a gas chromatography separation…
Changing the sample solvent
I can tell that my HPLC solvent is very gassy, but I’m not sure when to “degass” it…
BEFORE ENTERING THE COLUMN

Ruby is tired of being a cowgirl and wants her girl back, but she needs to know what the piston is made of in a HPLC high-pressure pump…
OMG ITS SAPPHIRE

How are samples usually cleaned before being injected into the HPLC?
Cleaned up with a syringe filter

when performing liquid-liquid extraction, is it common practice to Shake (Shake Shake Senora) the separatory funnel to disperse one phase into the other
Yes, shaking increases contact between the two immiscible phases, allowing the solute to distribute itself between the two layers. It does not change the partition coefficient but maximizes efficiency by reaching equilibrium faster.
Would it be easier to extract aqueous acetic acid into hexane with a aqueous phase pH of 3 or 8?
At a pH of 8, acetic acid is going to be in the ionic conjugate base form, which will heavily prefer being in the water.
At a pH of 3, acetic acid is going to be in the protonated acid form, which will be better at being extracted into the nonpolar hexane
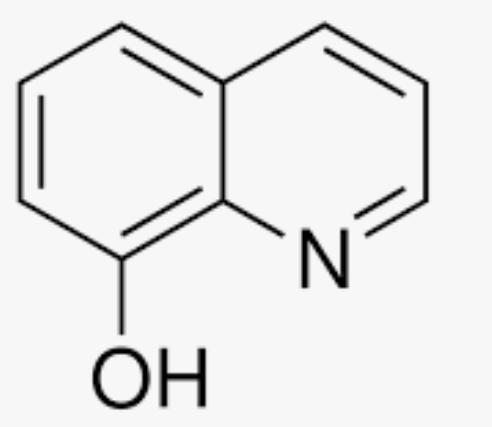
Why is this thing better at extracting metal ions into an organic solvent at higher pH’s?
At a higher pH, the Oxygen will not be protonated and will be able to bind to the metal
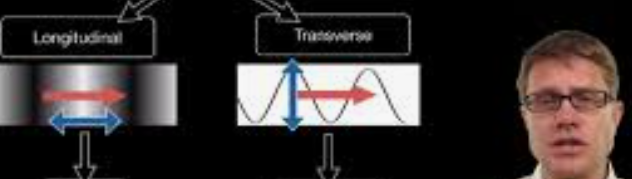
Why is longitudinal diffusion a more serious problem in gas chromatography than liquid chromatography?
Longitudinal diffusion (B/ux) is the measure of diffusion of solute along the column, and GC solute molecules are going to be extremely more diffuse (10000 times faster) and will increase this factor in the van equation.

PROMPT INTERMISSION SLIDE
even the HELPful knowt flashcards need a break froM lEarning