Enzymes and Metabolism: Thermodynamics, ATP, and Regulation in Biology
1/82
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
83 Terms
Metabolism
All the chemical processes that occur within living organisms.
Catabolic pathways
Breaking down of complex molecules that release energy.
Anabolic pathways
Building of larger and complex molecules from smaller and simple molecules that consume energy.
Thermodynamics
The study of energy transformations in a collection of matter.
Isolated system
A system unable to exchange energy or matter with its surroundings.
Open system
A system where energy and matter can be transferred between the system and its surroundings.
Kinetic energy
Energy associated with motion.
Thermal energy
The kinetic energy associated with random movement of atoms or molecules.
Chemical energy
Potential energy available for release in a chemical reaction.
Potential energy
Energy that matter possesses because of its location or structure.
1st Law of Thermodynamics
Energy can be transferred or transformed but cannot be created or destroyed.
2nd Law of Thermodynamics
Every energy transfer or transformation increases entropy.
Entropy
A measure of molecular disorder or randomness.
Spontaneous Process
Any process that leads to an increase in entropy that can proceed without requiring an input of energy.
Non-spontaneous process
Any process that leads to a decrease in entropy.
Non spontaneous process
Any process that leads to decrease in entropy is said non spontaneous process. This process will occur only if energy is supplied.
Free-energy change
The free-energy change of a reaction tells us whether or not the reaction occurs spontaneously.
Energy and entropy changes
Biologists follow the energy and entropy changes during chemical reactions to determine whether they require an input of energy or occur spontaneously.
Spontaneous reaction inquiry
Will this particular reaction happen or will we need to pay into it with energy?
Gibbs free energy (G)
The portion of a system's energy that can do work when temperature and pressure are uniform throughout the system, as in a living cell.
Change in free energy (ΔG)
Used to determine whether a process is spontaneous or not.
Spontaneous processes
Processes that decrease the system's free energy and have a negative ΔG.
Nonspontaneous processes
Processes that have a zero or positive ΔG.
ΔG formula
ΔG = Gfinal state - Ginitial state.
Negative ΔG
Indicates that the system loses free energy and becomes more stable.
ΔG and temperature
Change in free energy during a reaction is related to temperature and changes in enthalpy and entropy.
ΔG formula with variables
ΔG = ΔH - TΔS, where ΔH is change in enthalpy, ΔS is change in entropy, and T is temperature in Kelvin.
Kelvin scale
Same as Celsius, but shifted down so that 0°K = -273°C.
Exergonic reaction
A reaction that proceeds with a net release of free energy to the surroundings.
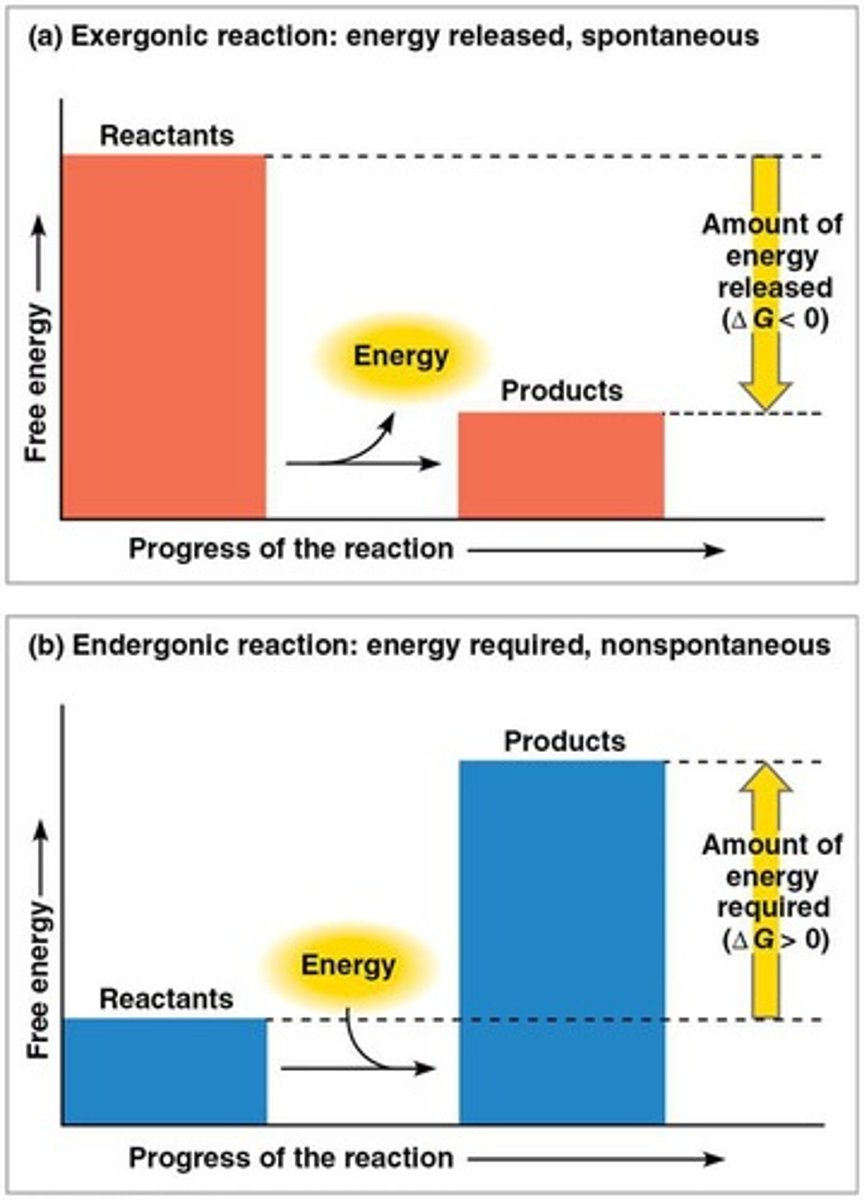
Endergonic reaction
A reaction that absorbs free energy from the surroundings.
ATP (adenosine triphosphate)
Composed of ribose, adenine, and three phosphate groups.
ATP hydrolysis
Energy is released from ATP when the terminal phosphate bond is broken by hydrolysis.
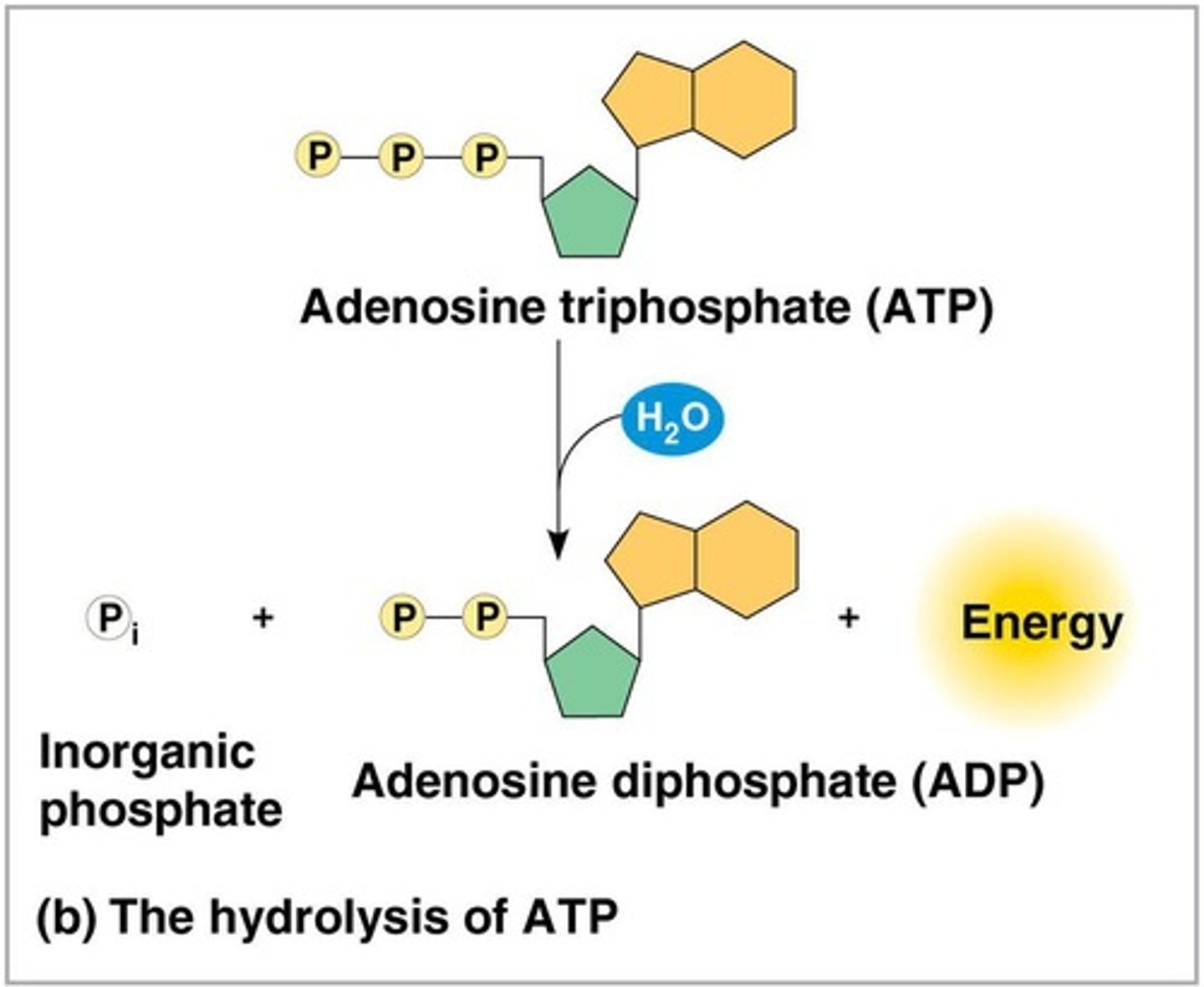
Energy from ATP
Does not come directly from the phosphate bonds, but from the chemical change to a state of lower free energy in the products.
Phosphorylation
Transfer of a phosphate group from ATP to another molecule, typically used to power endergonic reactions.
Phosphorylated intermediate
The recipient molecule that is more reactive and has more free energy than the original molecule.
Regeneration of ATP
ATP is regenerated by addition of a phosphate group to adenosine diphosphate (ADP).
ATP cycle
The shuttling of inorganic phosphate and energy that couples energy-yielding processes to energy-consuming ones.
Enzymes
Speed up metabolic reactions by lowering energy barriers.
Spontaneous reactions
Do not need added energy, but can be very slow.
Example of slow spontaneous reaction
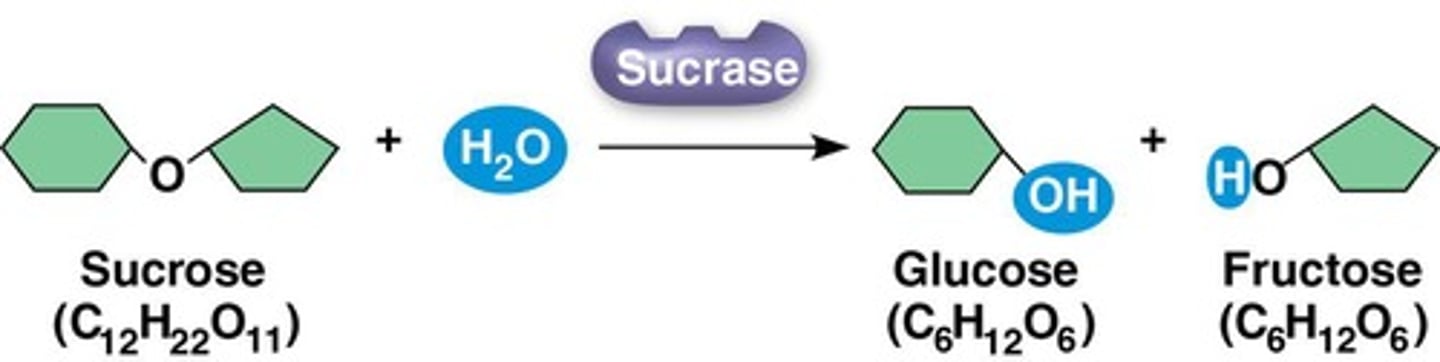
The hydrolysis of sucrose to glucose and fructose can take years at room temperature without appreciable hydrolysis.
Free-energy change (ΔG)
The change in free energy for a process can be used to determine whether it is spontaneous or not.
Zero or positive ΔG
Indicates that a process is non-spontaneous.
ΔG equation
ΔG = Gfinal state - Ginitial state.
Stable system
A system with negative ΔG loses free energy and becomes more stable.
Example of spontaneous reaction
The hydrolysis of sucrose to glucose and fructose is spontaneous but can take years at room temperature.
Catalysts
Chemical agents that speed up a reaction without being consumed in the reaction.
Enzyme
A macromolecule (typically protein) that acts as a catalyst to speed up a specific reaction.
Sucrase
An enzyme that catalyzes the complete hydrolysis of sucrose within SECONDS when added to a sucrose solution at room temperature.
Activation Energy (EA)
The initial energy needed to break the bonds of the reactants.
Transition State
A highly unstable state that a molecule must reach for bonds to break and start a reaction.
Catalysis
The process by which a catalyst selectively speeds up a reaction without itself being consumed.
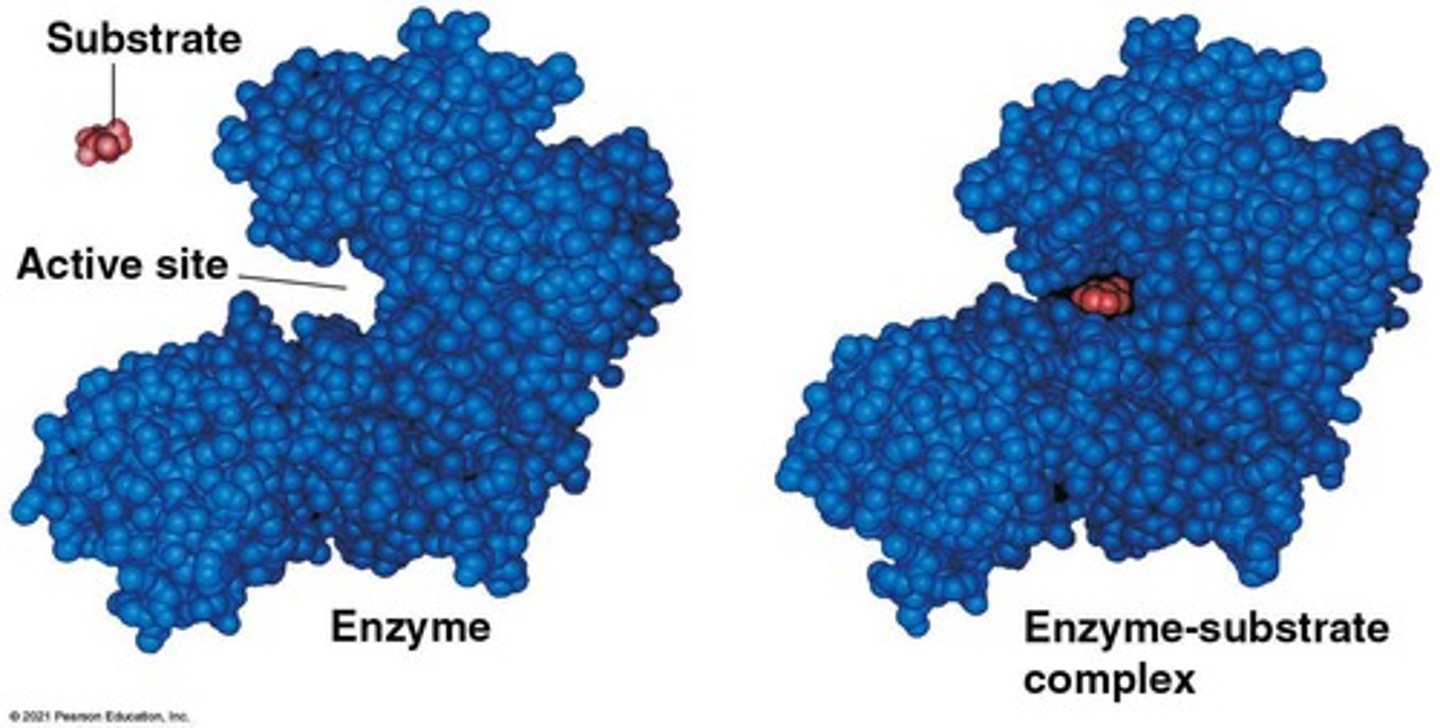
Substrate Specificity
The complementary fit between the shape of the active site of an enzyme and the shape of the substrate.
Active Site
The region on the enzyme, often a pocket or groove, that binds to the substrate.
ΔG
The change in free energy; enzymes cannot change ΔG.
Denature
The process by which proteins lose their structure and function due to heat or other factors.
Energy Profit
The excess energy released in an exergonic reaction after new bonds are formed.
High Activation Energy
Most reactions that require additional energy (usually heat) to reach the transition state.
Moderate Temperatures
The temperature range at which enzymes lower the activation energy barrier enough for reactions to occur.
Chemical Reaction
A process involving the breaking and forming of bonds between molecules.
Molecule Stability
Molecules become unstable when enough energy is absorbed to break bonds.
Heat in Reactions
Adding heat is impractical in cells as it speeds up all reactions, not just those needed.
Enzyme Function
Enzymes speed up reactions that would eventually occur anyway.
Bonds Breaking
The process that must occur for a chemical reaction to start.
Bonds Forming
The process that occurs after bonds break, leading to the release of energy.
Substrates
Molecules that enzymes act upon, which may be oriented or stretched to facilitate reactions.
Optimal Temperature
The temperature at which an enzyme catalyzes its reaction at the maximum possible rate.
Denaturation
The process by which enzymes lose their functional shape due to extreme temperature or pH.
Optimal Temperature for Human Enzymes
Approximately 37°C.
Optimal Temperature for Thermophilic Bacteria
About 75°C.
Optimal pH
The pH level at which an enzyme is most active.
Optimal pH for Pepsin
2, which is suitable for the human stomach.
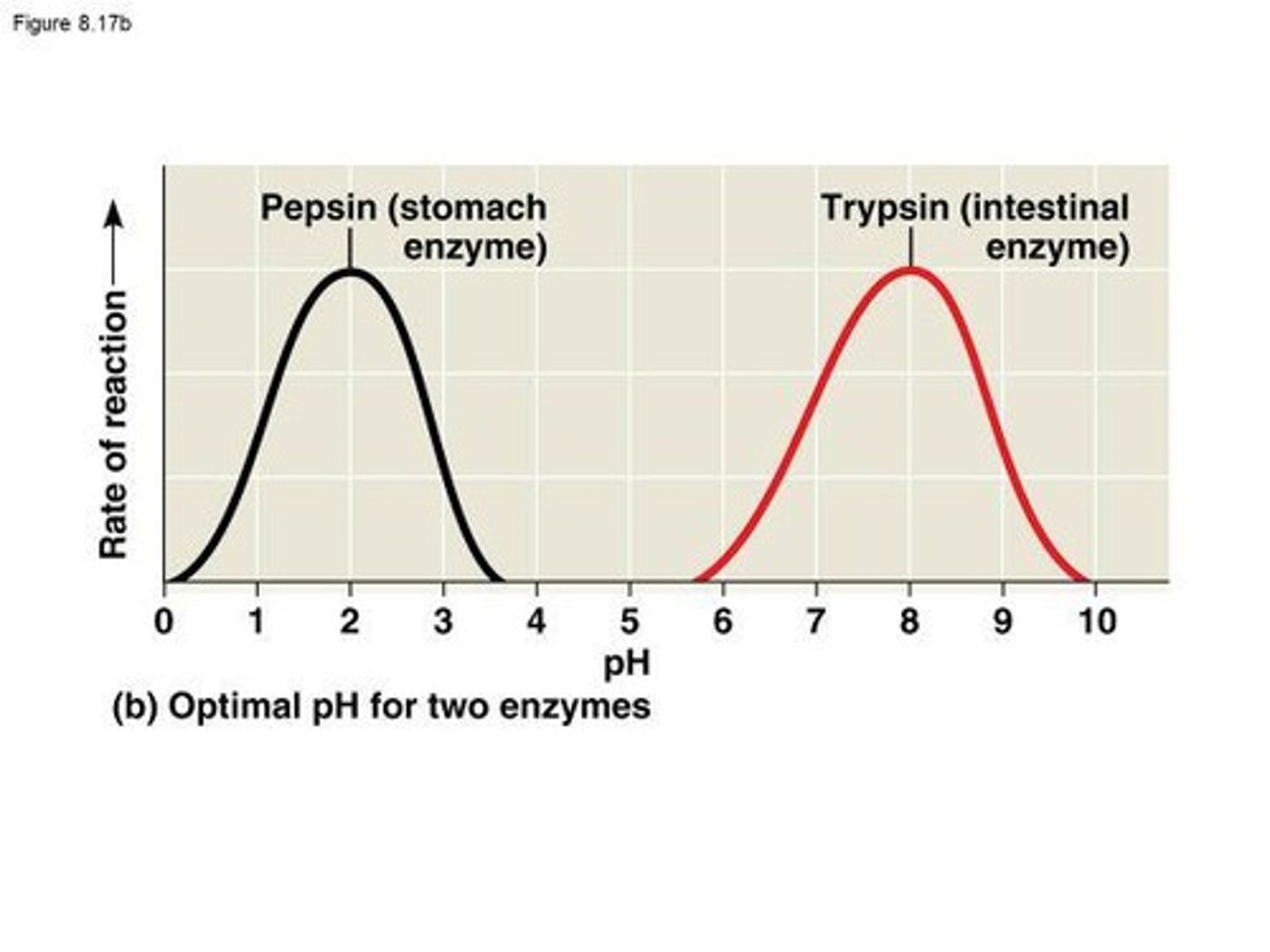
Optimal pH for Trypsin
8, which is suitable for the intestine.
Cofactors
Non-protein helpers that assist enzymes, which can be either inorganic or organic.
Inorganic Cofactors
Metal atoms such as zinc, iron, and copper in ionic form.
Coenzymes
Organic cofactors that assist enzymes, often derived from vitamins.
Enzyme Inhibitors
Chemicals that selectively inhibit the action of specific enzymes.
Irreversible Inhibition
When an inhibitor forms covalent bonds with an enzyme, permanently disabling it.
Reversible Inhibition
When inhibitors bind to enzymes through weak interactions, allowing for recovery of enzyme function.
Competitive Inhibitors
Inhibitors that resemble substrates and bind to the enzyme's active site, blocking substrate access.
Noncompetitive Inhibitors
Inhibitors that bind to an enzyme at a site other than the active site, altering enzyme shape and function.
Metabolic Pathway
A series of chemical reactions in a cell, where a specific molecule is altered to produce a final product.
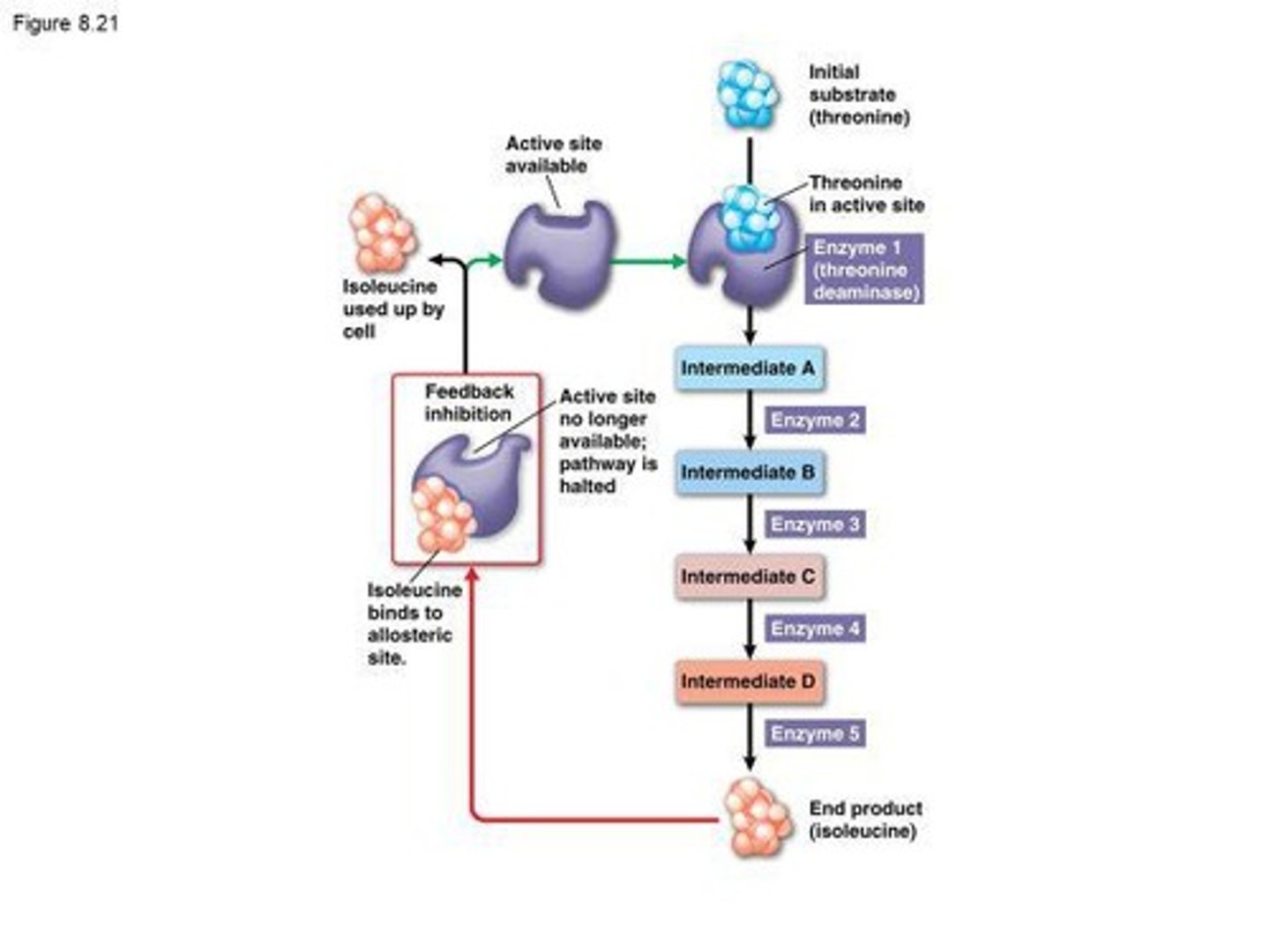
Allosteric Regulation
Regulation of enzyme activity through the binding of a regulatory molecule at a site other than the active site.
Feedback Inhibition
A regulatory mechanism where the end product of a metabolic pathway inhibits an earlier step in the pathway.