Cytotoxic chemotherapy
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Rationale for cytotoxic use
•Cancer chemotherapy aims to induce apoptosis or senescence → either kill it or get it to not divide anymore.
•Often targeted at proteins overexpressed in the cancer
• Affects cell division by targeting:
DNA replication
DNA transcription
DNA repair
Cytotoxic classes
1.Topoisomerase inhibitors
2.Alkylating agents
3.Antimetabolites
4.Antibiotics
5.Microtubule inhibitors
•
Note: Structures do not need to be learnt but you do need to be able to explain how the drugs work.
Targets for cytotoxics – DNA replication and transcription
•Human DNA is supercoiled.
•Relaxation of this supercoiling is required for the replication of DNA or its transcription
•It is achieved by the action of an enzyme called topoisomerase
•There are TWO main topoisomerases:
•DNA topoisomerase I cleaves one strand of DNA which it later reseals.
•DNA topoisomerase II first cleaves both strands of DNA before it reseals.
Targeting topoisomerase prevents DNA replication
•Topoisomerases are over-expressed in many cancers (e.g. cervical, colon liver, pancreatic, non-small cell lung carcinoma, myelomas, leukaemia and multi-drug resistant cancers).
Mechanism of Action of Topoisomerase Inhibitors
4 possible mechanisms of Action:
(1)Stabilization / inhibition of enzyme/DNA complex,
(2)Binding of Drug to DNA totally preventing enzyme action,
(3)Binding of Drug cleaves DNA in the presence of enzyme but resealing by the enzyme is prevented → means you have a break in the DNA, cells cannot repair them all and then dies
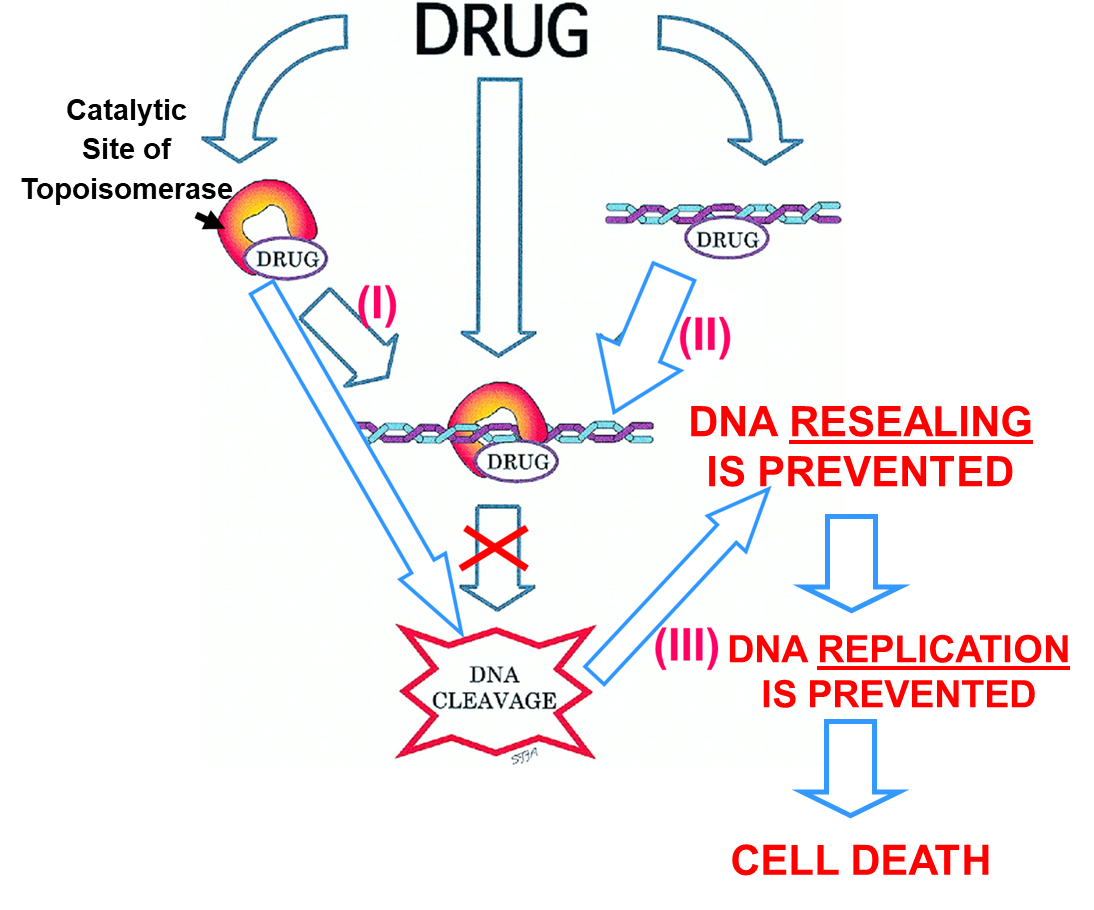
4)Intercalation: The planar ring structure of anthracyclines intercalates between DNA strands, causing structural distortion of the DNA and preventing polymerase progression therefore inhibiting both DNA and RNA synthesis. In other words it causes distortion of the helix preventing DNA replication and transcription.
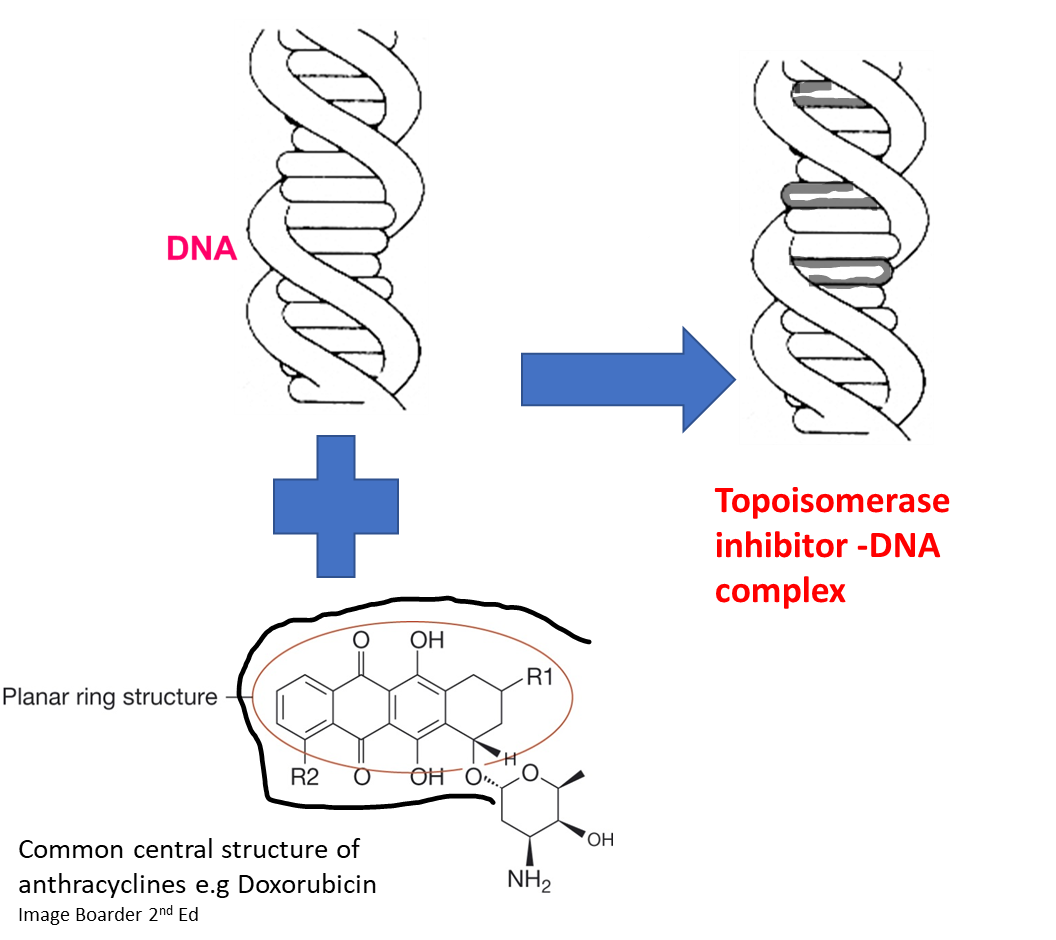
This figure is showing how some topoisomerase inhibitors have a planar structure that sits in the DNA helix
Types of Topoisomerase Inhibitors:
1.Camptothecins (Topo I) (S phase)
•e.g. Camptothecin, Topotecan, Irinotecan
•acts via mechanism 1 → binding to DNA and stopping topoisomerase from getting into it.
2.Podophyllotoxins (Topo II) (S phase)
•e.g. Etoposide
•Acts via mechanism 1 or 3
•Anthracyclines (Topo II) (S and M phase)
•e.g. Doxorubicin
•Acts via mechanism 1, 2, 3 or 4
Metabolism of Irinotecan
1. Effect of genotype
•Irinotecan is a prodrug
•SN-38 is the active metabolite
•SN-38 is inactivated by glucuronidation, the addition of glucuronic acid by uridine diphosphoglucyronyl transferase (UGT1A1)
•Genotype:
•UGT1A1 is a phase II enzyme with >60 alleles
•Some alleles, eg UGT1A1*28 allele (TA)7 repeat in TATA box of its promoter has a low activity phenotype (70% lower gene expression) leading to adverse reactions eg neutropenia in those treated with a normal dose.
•Could be addressed by genotyping or monitoring WBC.
•This genotype does not have an impact on the efficacy of irinotecan
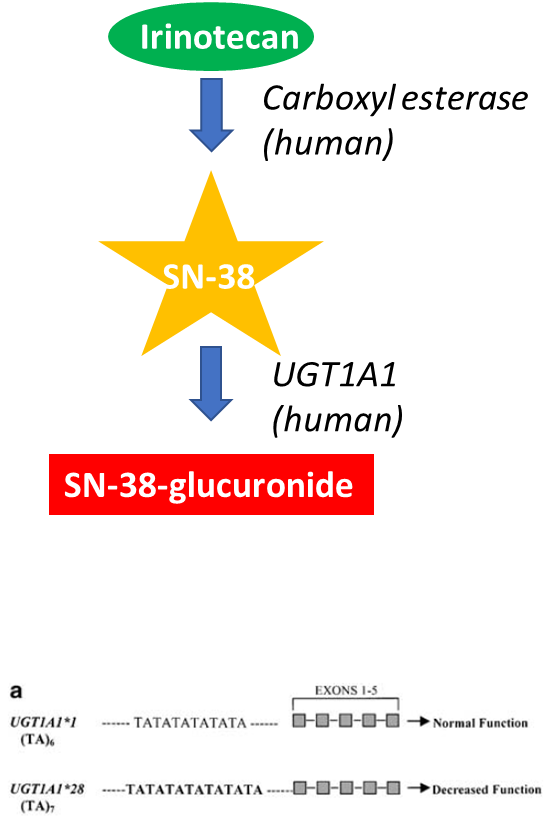
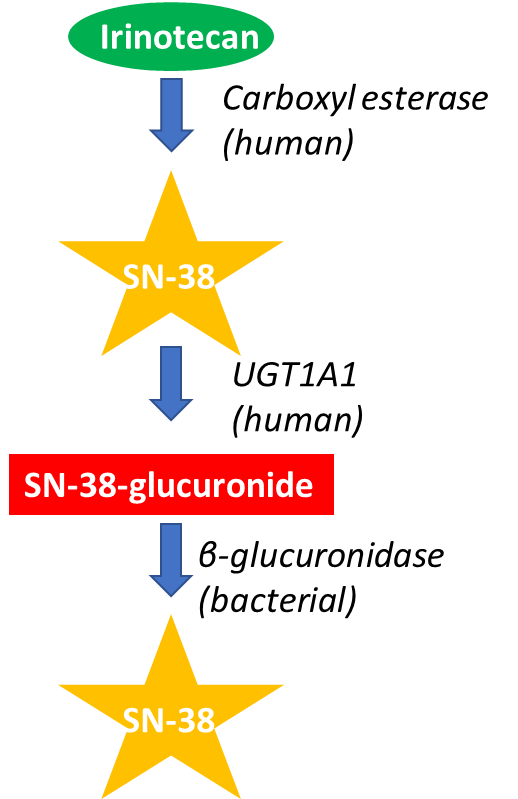
Metabolism of Irinotecan
2. Effect of the environment
•Environment:
•After inactivation SN-38-glucuronide is secreted via bile into the intestines
•There it can be re-activated through hydrolysis by an enzyme produced by gut microflora causing severe diarrhoea.
•Could be addressed by inhibiting the bacterial enzyme to prevent the intestinal metabolism of irinotecan
•
Monitoring the phenotype to take into account these genetic and environmental factors could reduced adverse effects of the drug, potentially allowing dose escalation that will enhance the drug's efficacy.
Alkylating agents
•E.g. Platinum compounds and Nitrogen Mustards
•Common features
•form covalent bonds with DNA via the production of a reactive species.
•bivalent therefore bonds are formed across or within stands of DNA
•They are effective cytotoxics because they crosslink DNA causing
•DNA strand denaturing or breakage
•DNA Repair
•Replication Inhibition
•Transcription Inhibition as polymerase can’t act on the DNA.
•Cell Cycle Arrest as cell won’t go through cell cycle unless cycle is okay.
•Cellular Stress
•Cell Death
Platinum Compounds
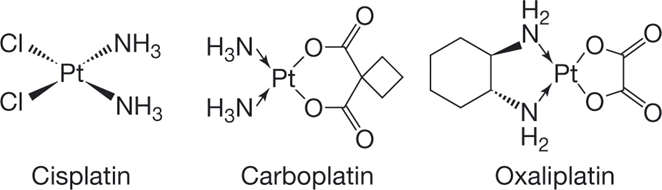
•Platinum compounds form reactive intermediates that bind to the 7’ nitrogen of adenine or guanine crosslinking DNA
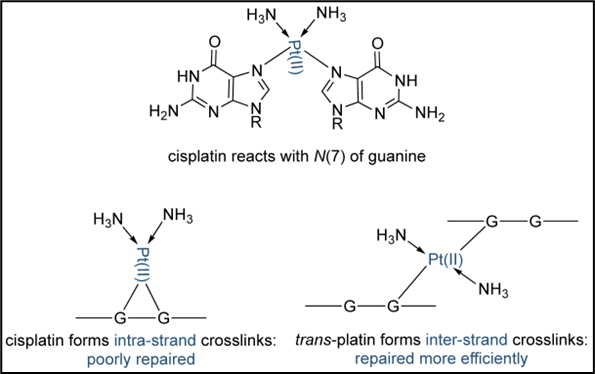
•This prevents unwinding of the DNA helix i.e. blocks DNA replication and transcription,
•Crosslinking can result in strand breakage, DNA repair is attempted but is not possible due to the crosslinking leading to cell stress and apoptosis.
Nitrogen Mustards
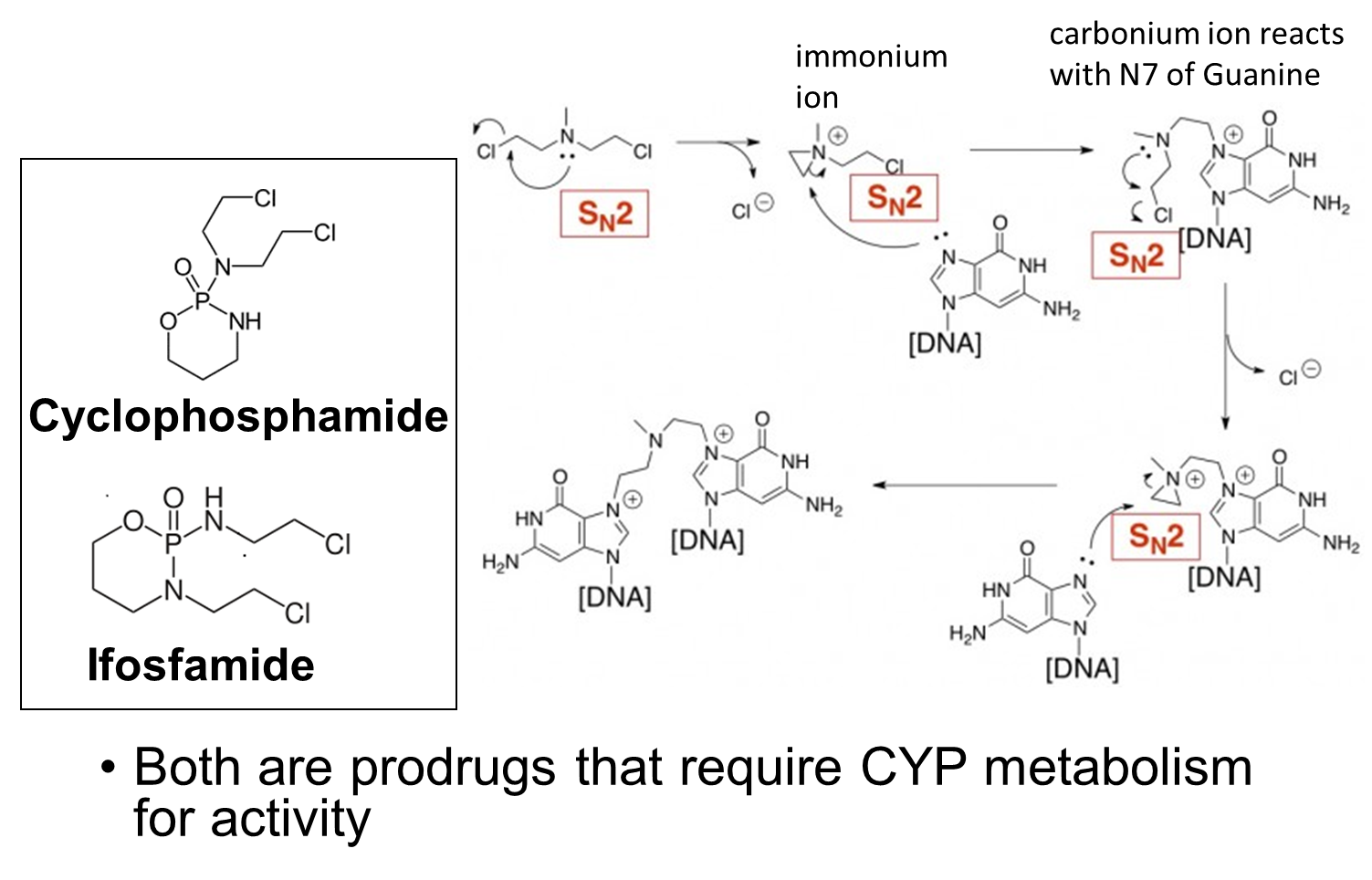
Nitrogen mustards have a reactive species formed. It requires CYP450 activity for this reaction to occur
Cyclophosphamide conversion to the active Phosphoramide mustard
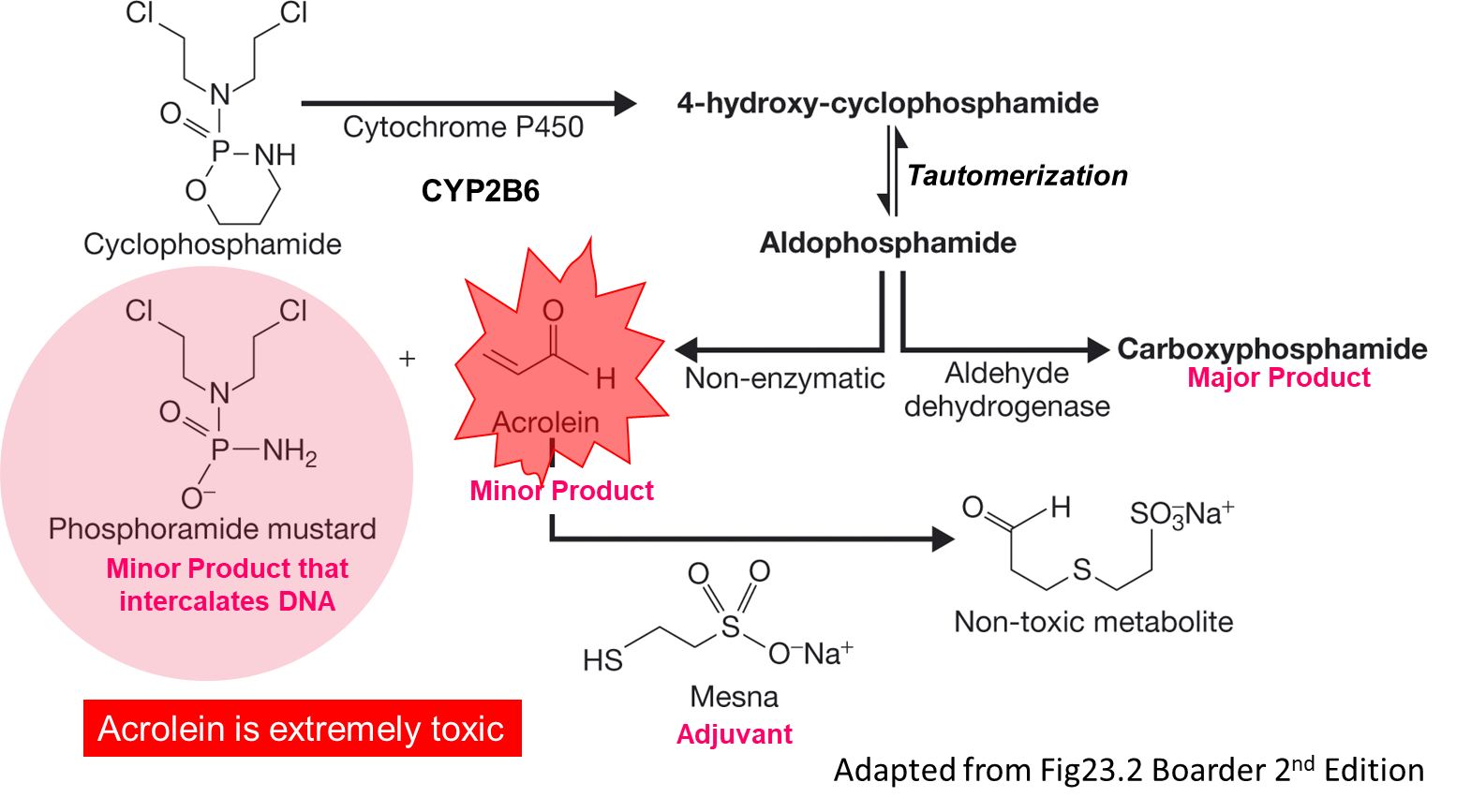
A very small portion of the drug that is given is actually active. Oxidative metabolism of the drug forms an intermediate which is acted on by aldehyde dehydrogenase which produces the major product that is totally inactive. There is also a non enzymatic reaction that happens that causes the mustard/minor product to be formed which intercalates the DNA. Also forms acrylamide so is often given meth mesna which leads to an inactive form from this. High concentrates of acrylamide in the bladder gives really big problems.
Antimetabolites
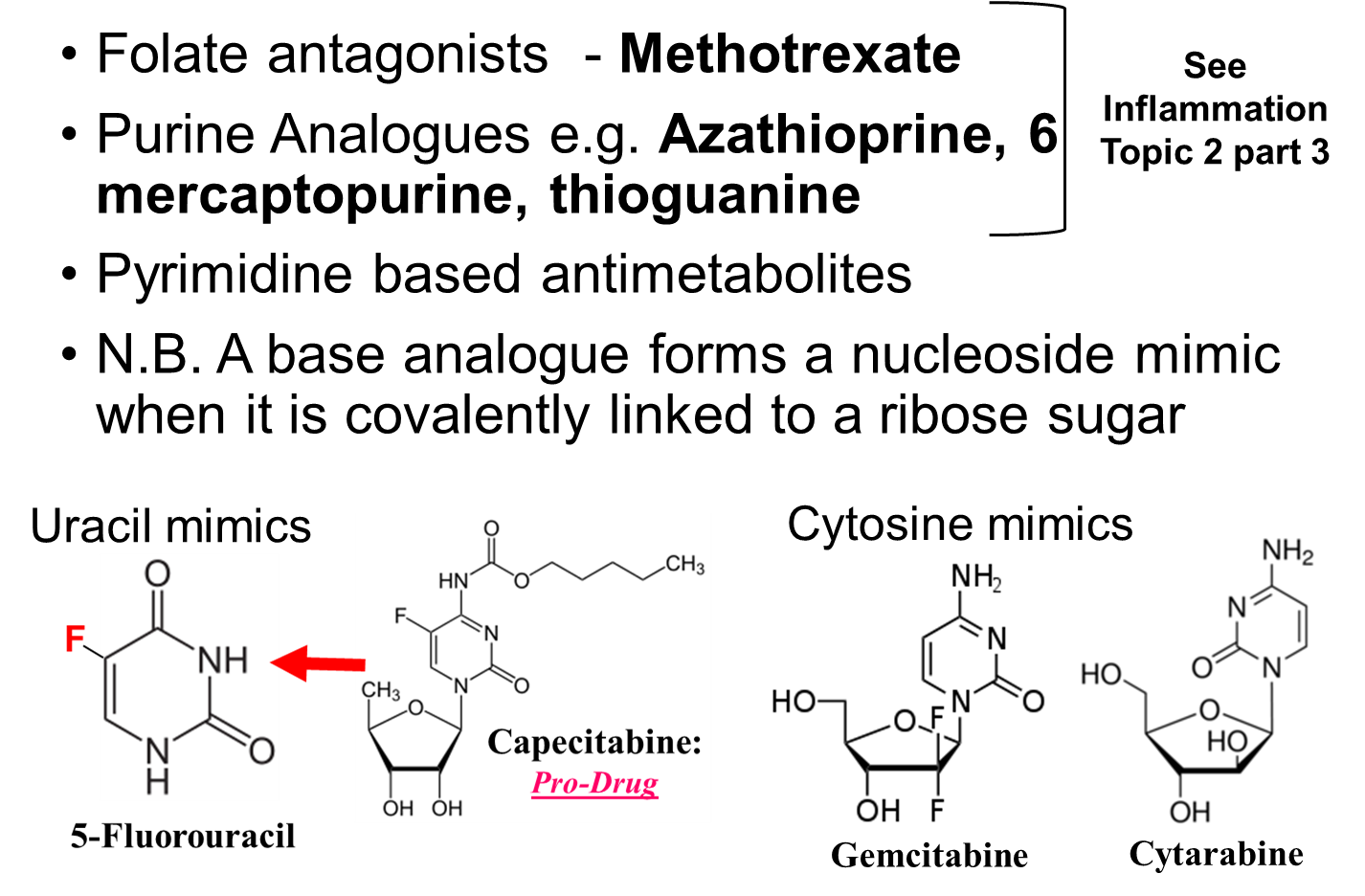
All these antimetabolites haves a stricture that is similar to that of a nucleotide and will interfere with the production of nucleotides or will be incorporated into the DNA forming a steric block on any further replication or transcription.
Pyrimidine based antimetabolites are based on uracil or cytosine.
Uracil mimics - Capecitabine and
5’-flurouracil
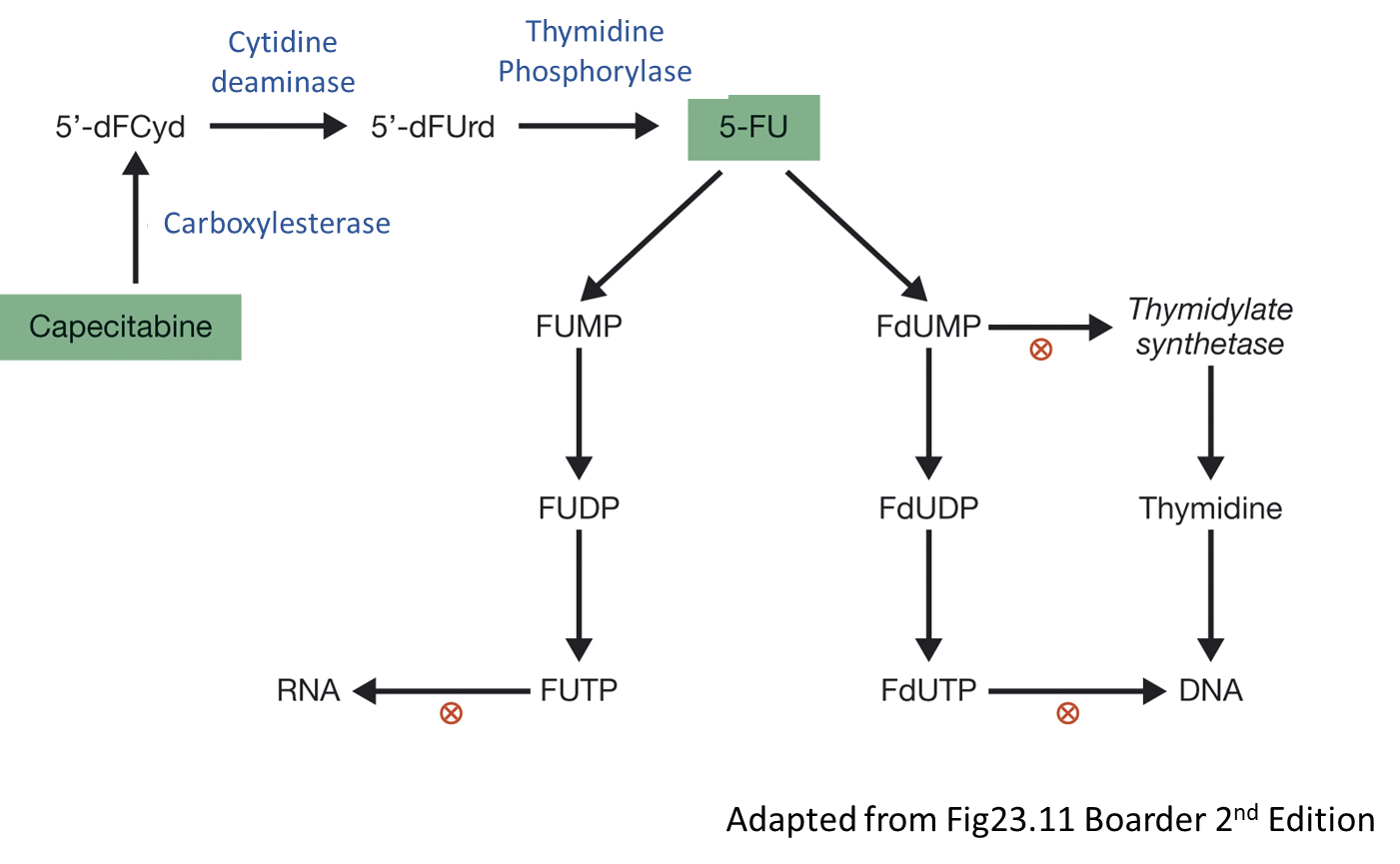
Uracil mimics need conversion into active form and then we get incorporation of DNA/RNA
Cytosine mimics – Gemcitabine & Cytarabine
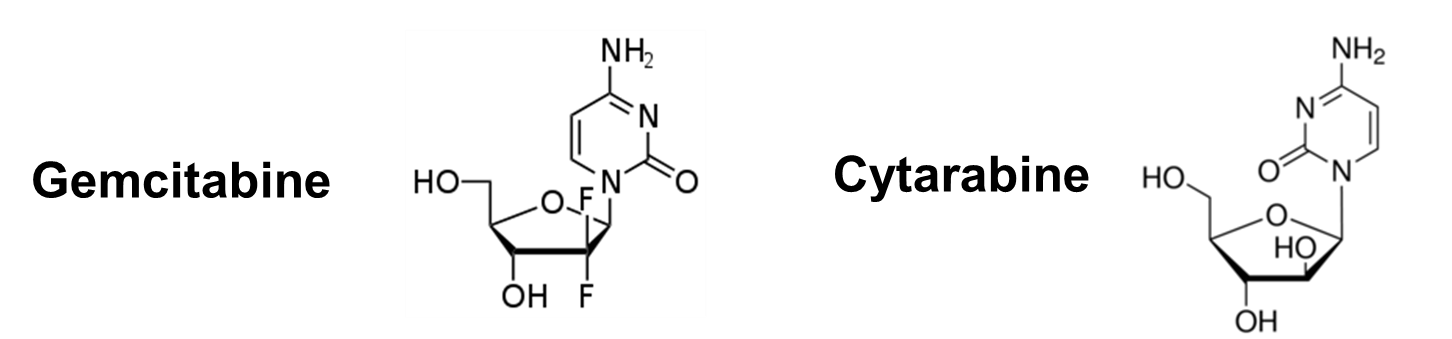
Gemcitabane.
•Phosphorylated to Gemcitabine triphosphate
•Inhibits DNA polymerase
•Inhibits Ribonucleotide reductase
•Can be incorporated into DNA
•Also known as Ara C
•Phosphorylated to AraCTP
•inhibits DNA polymerase (minor effect
Can be incorporation into DNA (major effect
N.B Both are deoxy-ribonucleosides
DNA replication in the presence of a nucleoside mimic
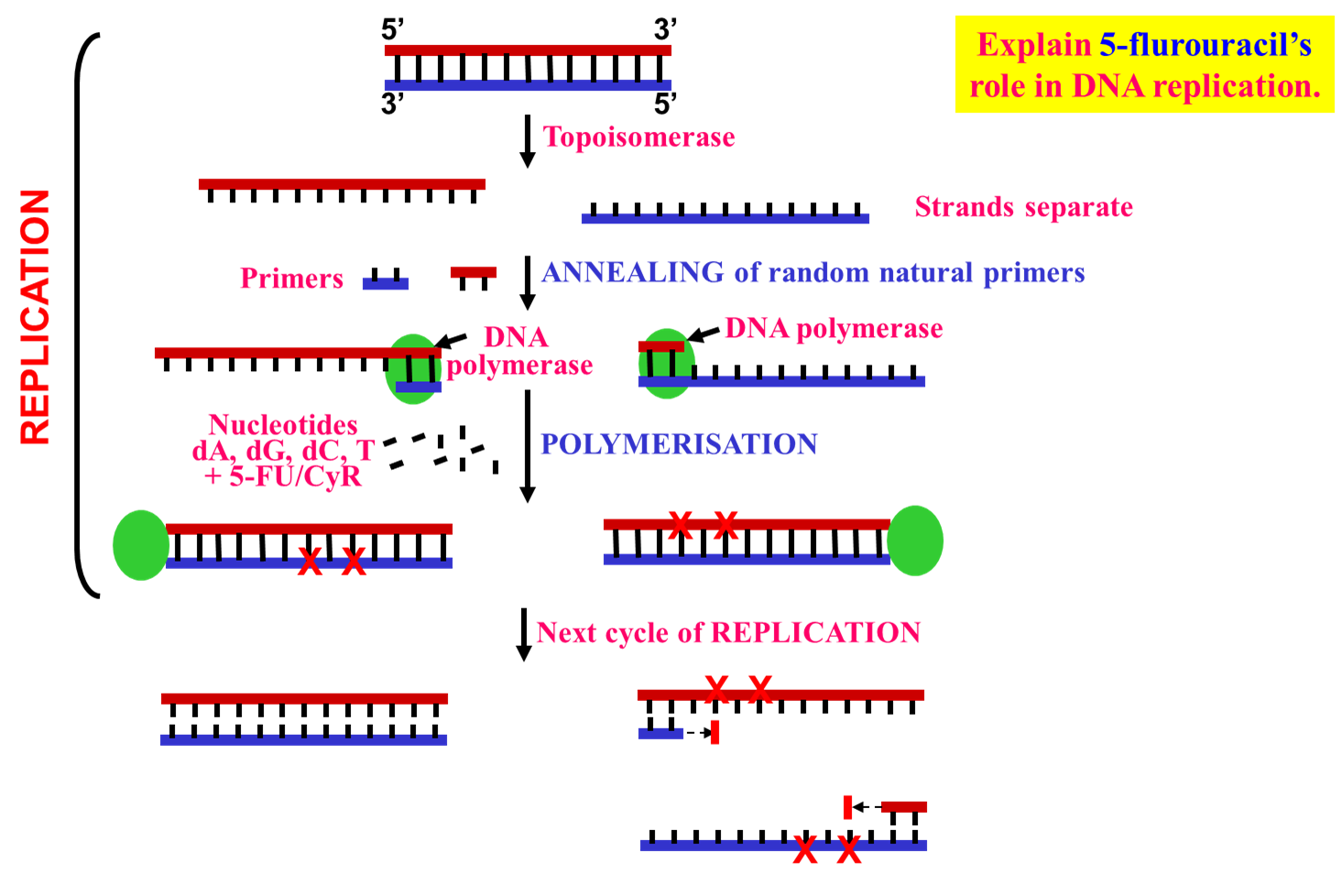
Here we have double stranded DNA. We require action of topoisomerase to release the supercoiling. Nucleoside mimics will prevent the next round round of replication and therefore takes a while to exert its effect because only one daughter cell will have the damaged DNA. As you move along the parental cells can no longer be replicated and that can cause stress and expression of p53 pushing cell towards apoptosis.
Antibiotics as cytotoxics - Bleomycin
•Glycopeptide antibiotic produced by the bacterium Streptomyces verticillus.
•Bleomycin refers to a family of structurally related compounds that bind iron in the presence of oxygen to produce a reactive peroxide
•DNA undergoes an oxidation reaction in its presence to induce single or double strand breaks
•Disrupts the cell cycle in stages G2 and M
Targets for cytotoxics – Mitosis
•Microtubule is a major filament of the cytoskeleton.
•It is composed of alpha and beta-tubulins protofilament rings, tubulins give microtubules polarity.
•Its polymerization and de-polymerization regulate microtubular dynamics and play an essential role in the formation of the mitotic spindle and cell cycle progression
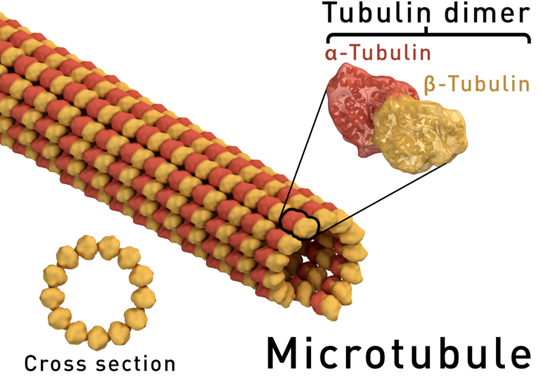
•The protofilaments bundle parallel to one another, so, in a microtubule, there is one end, the (+) end, with only β-subunits exposed, while the other end, the (−) end, has only α-subunits exposed.
•Elongation of microtubules typically only occurs from the (+) end.
•Polarity is important for microtubular biological function
Microtubule inhibitors
•Vinca alkaloids
e.g. Vinblastine, Vincristine
Bind to β-tubulin to inhibit microtubule polymerisation
•Taxanes
e.g. Paclitaxel and Docetaxel
Bind to β-tubulin to stabilise the microtubule to prevent polymerisation and depolymerisation
Both prevent formation of spindle fibres and therefore prevent sepration of DNA.
Adverse effects of cytotoxics
•Bone marrow suppression
•Gastrointestinal effects
Nausea and vomiting
Diarrhoea
•Mucositis
•Alopecia
•Infertility