topic 10-11 chem equlibrium, calculating Kc and Kp
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
define homogenous reaction
reactants and products are in the same state (liquid / gas / solid)
define heterogeneous reaction
reactants and products are in different states (liquid / gas / solid)
give 2 features of a reaction in a dynamic equilibrium
- the rates of the forward and backward reactions are the same
- concentrations of the reactants and products remain constant
2NO2(g) [brown gas] ⇌ N2O4(g) [colourless gas]
when an equilibrium is set up in a gas syringe, the mixture is pale brown. When the mixture is compressed the colour becomes:
A darker
B lighter
C darker and then lighter
D lighter and then darker
C
mixture initially becomes darker, then turns lighter due to more N2O4 being produced as the equilibrium shifts to the side with less gas moles
what is omitted in a heterogenous reaction (for Kc and Kp)
- all solids are constant
- reactants that feature a liquid phase (l)
- reactants that feature aqueous phase (aq)
calculating the units for Kc / Kp
products / reactacts
* same for Kp, except use atm instead of mol dm-3
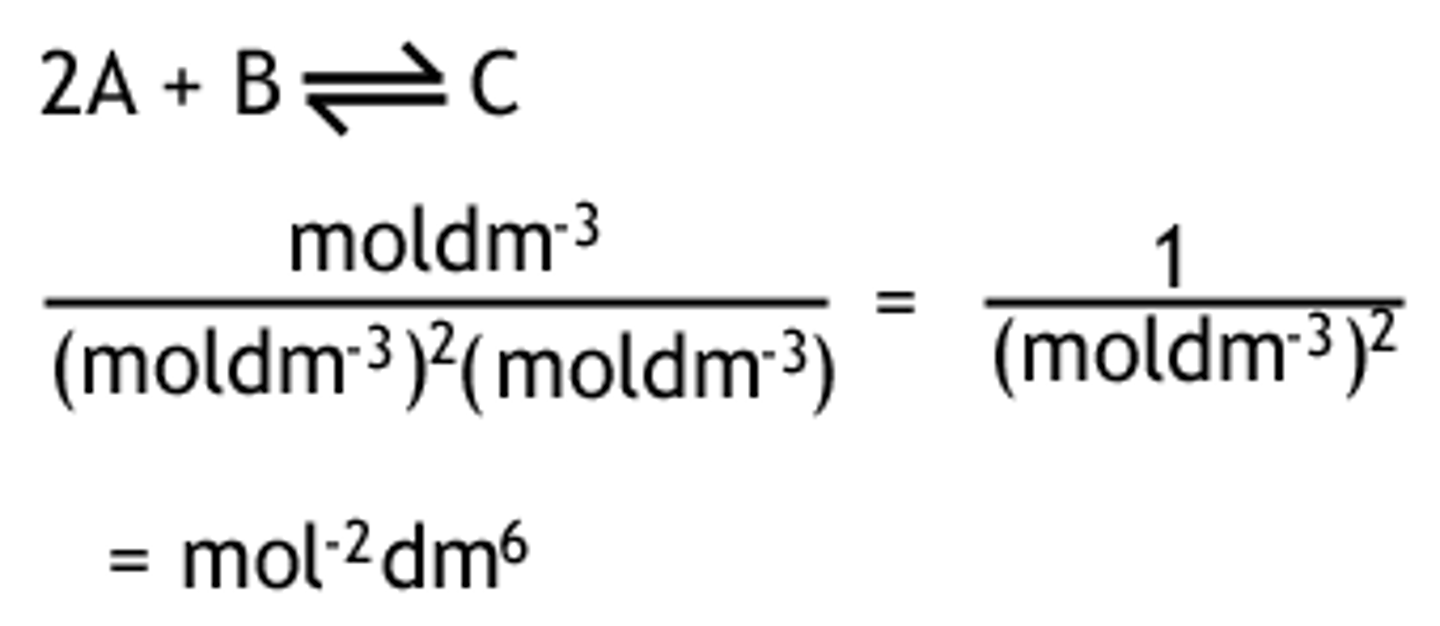
name the only factor that changes the value of Kc or Kp
temperature
walkthrough the process of calculating Kc
1. calculate equilibrium moles using ICE method
★ take ratios into account for 'change in moles'
2. calculate equilibrium concentration: mol / volume (volume is given in question)
3. calculate Kc: [products] / [reactants]
4. calculate unit for Kc (mol dm-3)
![<p>1. calculate equilibrium moles using ICE method</p><p>★ take ratios into account for 'change in moles'</p><p>2. calculate equilibrium concentration: mol / volume (volume is given in question)</p><p>3. calculate Kc: [products] / [reactants]</p><p>4. calculate unit for Kc (mol dm-3)</p>](https://knowt-user-attachments.s3.amazonaws.com/9d8f0deb-90a4-4f0a-affa-b5c2e3cab908.jpg)
what does Kc = 1 mean?
the concentrations of the products must be equal to the concentrations of the reactions at equilibrium
CO (g) + H2O ⇌ CO2 (g) + H2 (g)
at 1100K, Kc = 1.00
in an experiment, 1 mol of carbon monoxide was mixed with 1 mol of steam, 2 mol of carbon dioxide and 2 mol of hydrogen.
deduce, with reasons, the direction in which the reaction will shift to reach equilibrium.
- concentration of products must = concentration of reactants at equilibrium since Kc = 1
- so the concentration of CO2 and H2 need to decrease, while the concentration of CO and H2O need to increase
- the reaction will shift left
what does the value of K in Kc tell us?
- large Kc value suggests position of equilibrium shifted to forward direction
- small Kc value suggests position of equilibrium is shifted to backwards direction
CH3COOH (l) + CH3CH2CH2OH (l) => CH3COOCH2CH2CH3 (l) + H2O (l)
explain why it is possible to calculate Kc using equilibrium amounts in moles, rather than equilibrium concentrations in this case.
- same number of moles on both sides
- so volume cancels in Kc expression
identify a safety issue associated with hydrogen gas
hydrogen is flammable / explosive
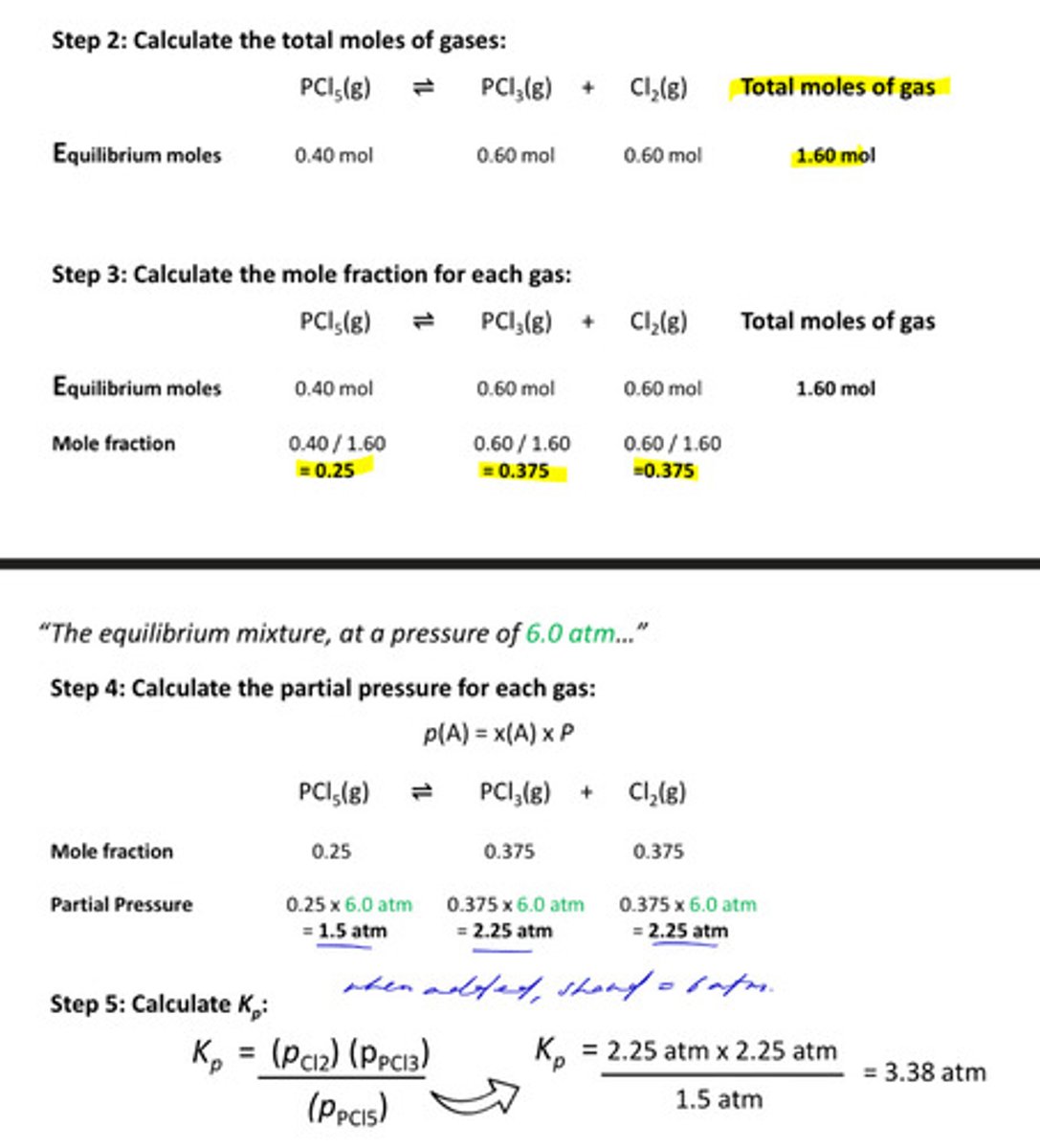
walkthrough the process of calculating Kp
1. calculate equilibrium moles using ICE method
★ take ratios into account for 'change in moles'
2. calculate total moles of gas (t) by adding everything up in equilibrium moles
3. calculate the mole fraction of each gas (mol / t)
4. calculate the partial pressure for each gas: mole fraction x atm (given in question)
5. calculate Kp: p(products) / p(reactants)
6. calculate unit for Kp (atm)
what would you expect to happen to the value of Kp as temperature increases? Give your reasons.
(CH3COOH)2 (g) ⇌ 2CH3COOH (g)
- dimer is formed by HYDROGEN BONDING between monomer molecules
- this will be disrupted when temperature increases, hence at higher temperature more monomers will be present
- Kc increases

explain what you could do to confirm that 1 week is sufficient time for the mixture to reach equilibrium
- repeat the experiment and leave it out for longer
- ensure Kc value remains unchanged
2SO2 (g) + O2 (g) ⇌ 2SO3 (g)
deduce, by referring to Kp, how the number of sulfur dioxide molecules will change if more oxygen is added to the equilibrium mixture
only temperature changes Kp, so SO2 has to decrease in order for O2 to increase
Mg(OH)2 (s) ⇌ Mg2+ (aq) + 2OH- (aq)
predict the effect of adding MAGNESIUM SULFATE SOLUTION to a saturated solution of magnesium hydroxide in contact with solid magnesium hydroxide. justify your answer in terms of the effect on equilibrium.
- equilibrium position shifts to the left
- due to increased concentration of Mg ions
Mg(OH)2 (s) ⇌ Mg2+ (aq) + 2OH- (aq)
predict the effect of adding DILUTE HYDROCHLORIC ACID to a saturated solution of magnesium hydroxide in contact with solid magnesium hydroxide. justify your answer in terms of the effect on equilibrium
- equilibrium shifts to the right
- as the H+ from HCl neutralises the OH-
what is the best piece of apparatus to measure out the volumes of liquids from a sealed test tube?
syringe
suggest why a reason why test tubes are sealed
to prevent vapour from escaping
state the role of concentrated HCl on the equilibrium reaction
provides H+ as a catalyst
explain why, in an industrial process, a catalyst is used
- increases rate of reaction
- lower activation energy
- allow milder conditions to be used, lowering costs
give 3 reasons why the product in a equilibrium reaction may be removed before its maximum concentration is achieved
- equilibrium shifts right = yield
- removal of products decrease rate of backwards reaction
- time to attain equilibrium is too long
explain the effect of an increase in pressure on the rate of reaction
- rate of reaction increases
- more molecules per unit volume
- increased frequency of collisions
the platinum-rhodium catalyst used in the reaction is heterogeneous. state what that means
- catalyst is in a different state than reactants
ΔH = - 178 kJ mol-1
chlorine is reacted with ethene at a temperature between 50C and 80C
give one reason why a temperature below 50C and another reason, apart from costs, why a temperature above 80C would not be used for this process
- below: reaction is too slow
- above: yield is lower due to exothermic forward reaction
give 2 characteristics of all reactions at equilibrium
- concentration of all components are constant
- rate of backwards reaction equals the forward reaction
C4H10 (g) ⇌ C2H6 (g) + C2H4 (g)
give the expression for the equilibrium constant, Kp, for this reaction
p(C2H6)p(C2H4) / p(C4H10)
Which statement(s) is/are true for a mixture of ice and water at equilibrium?
I. The rates of melting and freezing are equal.
II. The amounts of ice and water are equal.
III. The same position of equilibrium can be reached by cooling water and heating ice.
A. I only
B. I and III only
C. II only
D. III only
B
H2O (l) ⇌ H2O (s)
← heating ice
→ cooling water