all m3
1/91
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
92 Terms

(6 marks)

Chlorine is used in water treatment. State one benefit and one risk of using chlorine in water treatment. (1 mark)
benefit: kills bacteria
AND
risk: forms toxic compounds
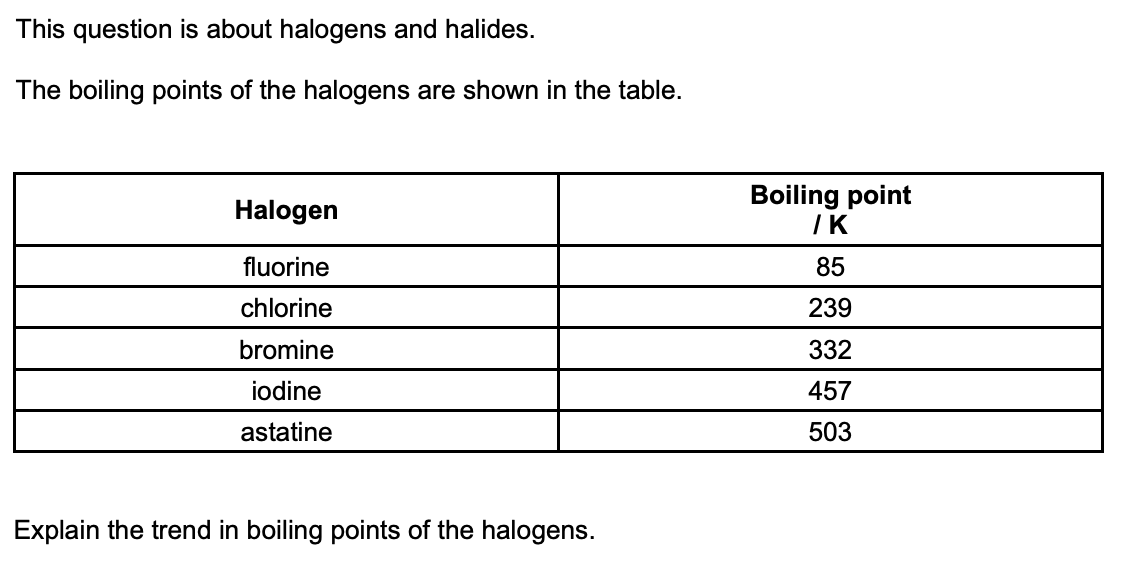
(3 marks)
as you go down the group, number of electrons increases
strength of london forces increases
more energy required to overcome intermolecular forces
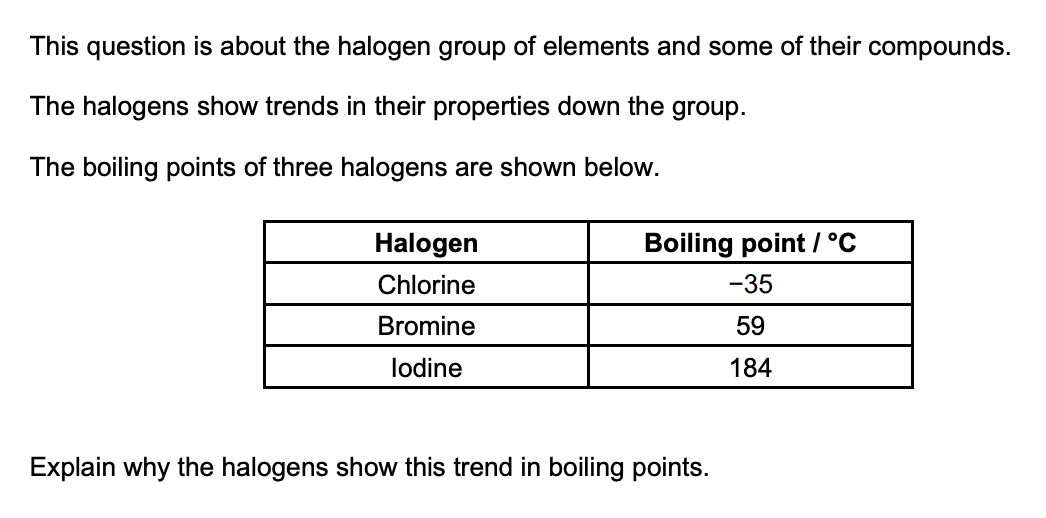
(3 marks)
as you go down the group, strength of london forces increase
number of electrons increases
more energy required to break intermolecular forces


Chlorine is used to purify water on a large scale. State one disadvantage of using chlorine for the purification of drinking water. (1 mark)
forms toxic compounds
Explain why iodine is less reactive than bromine (3 marks)
iodine has a larger atomic radius
iodine has greater shielding/ more shells
iodine has weaker nuclear attraction
Bromine disproportionates when it reacts with potassium hydroxide solution. Suggest an equation for this reaction. (1 mark)
Br2 + 2KOH → KBr + KBrO + H2O


Write the electron configuration of a bromide ion, in terms of sub‐shells. (1 mark)
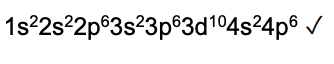
Bromine can be extracted by bubbling chlorine gas through concentrated solutions containing bromide ions. Write an ionic equation for this reaction and state why this reaction takes place in terms of reactivity of the halogens. (2 marks)


(5 marks)

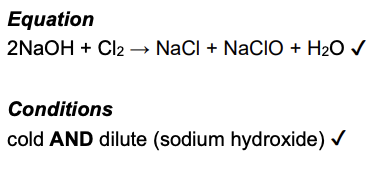
Chlorine reacts with aqueous sodium hydroxide to form bleach. Write the equation and state the conditions for this reaction. (2 marks)


Outline a simple practical test that would confirm the presence of chloride ions in the lower layer, and give the expected result. (2 marks)
test: add a few drops of aqueous silver nitrate
result: white precipitate
A student is provided with samples of three nickel compounds. One sample is nickel(II) bromide, another is nickel(II) sulfate and the third is nickel(II) carbonate. The student doesn’t know which sample is which. Describe the tests that the student could carry out to identify the anion (negative ion) in each sample, and write equations for any reactions (6 marks)
Tests for anions:
Carbonate test: Add HNO3(aq)/HCl(aq)/H2SO4(aq)/H+(aq) fizzing/ forms CO2(g) → Carbonate identified
Sulfate test: Add Ba(NO3)2(aq) OR BaCl2(aq) White precipitate → Sulfate identified
Bromide test Add AgNO3(aq) Cream precipitate → Bromide identified
Equations:
Carbonate: 2HNO3 + NiCO3 → Ni(NO3)2 + H2O + CO2
Sulfate: Ba(NO3)2 + NiSO4 → BaSO4 + Ni(NO3)2
Bromide: 2AgNO3 + NiBr2 → 2AgBr + Ni(NO3)2
carbonate → sulfate → bromide
Describe simple tests that would identify the cation and anion present in ammonium bromide. Include reagents, expected observations and relevant equations (5 marks)
test for anion (Br-)
Silver nitrate/AgNO3 AND cream (precipitate)
AgNO3 + NH4Br → AgBr + NH4NO3
test for cation (NH4+)
(Heat with) NaOH/ hydroxide
paper turns blue
NH4Br + NaOH → NaBr + NH3 + H2O
Compare the electrical conductivities of solid and aqueous barium chloride. Explain your answer in terms of the particles involved. (2 marks)
barium chloride doesn’t conduct electricity when solid AND because it has ions which are fixed in position/ in lattice
barium chloride conducts electricity when aqueous AND because it has mobile ions
Describe the use of aqueous barium chloride in qualitative analysis. (2 marks)
to test for sulfate
white precipitate forms (when barium chloride is mixed with a solution containing sulfate ions)
How would the appearance of the precipitates allow you to distinguish between chloride, bromide and iodide ions? (1 mark)
chloride: white ppt
bromide: cream ppt
iodide: yellow ppt
Complete the electron configuration of a bromide ion. (1 mark)
(1s2) 2s2 2p6 3s2 3p6 3d10 4s2 4p6
A student adds a small volume of aqueous silver nitrate to an aqueous solution of bromide ions in a test-tube. The student then adds a similar volume of dilute aqueous ammonia to the same test-tube.
i) Describe what the student would see in the test-tube after the addition of aqueous ammonia. (1 mark)
ii) Write an ionic equation for any precipitation reaction which occurs in the student's tests. Include state symbols. (1 mark)
i) cream AND precipitate
ii) Ag+(aq) + Br-(aq) → AgBr(s)
The reaction between aqueous sodium chlorate(I) and dilute hydrochloric acid produces aqueous sodium chloride as well as chlorine. Suggest an equation for this reaction. (2 marks)
NaClO + 2HCl → NaCl + Cl2 + H2O
State the conditions of temperature and pressure used for standard enthalpy measurements. (1 mark)
25C AND 100kPa
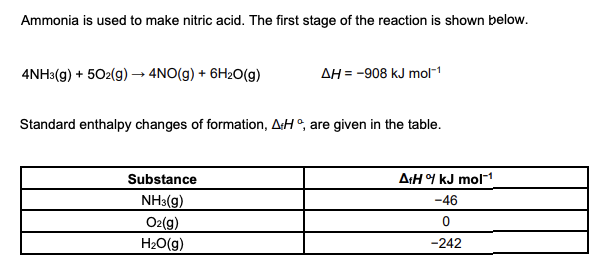
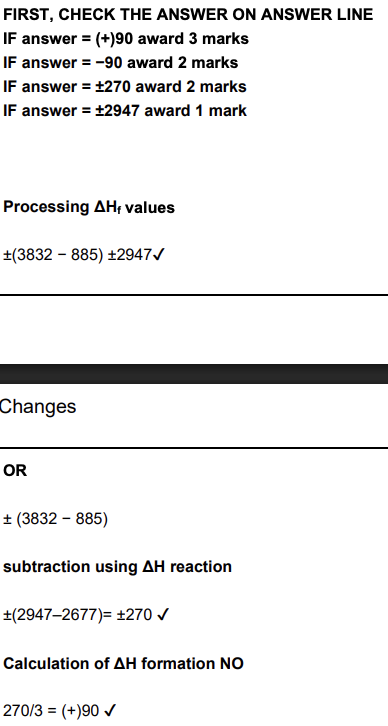
Calculate the standard enthalpy change of formation for NO(g). Give your answer to a whole number. (3 marks)




At the end of the experiment, the student adds a few drops of aqueous sodium chloride to the reaction mixture in the polystyrene cup to test whether all the aqueous silver nitrate has reacted. Explain how the results would show whether all the aqueous silver nitrate has reacted. Include an equation with state symbols in your answer. (2 marks)



Enthalpy change of vaporisation’ is the enthalpy change when one mole of a substance changes from a liquid to a gas at its boiling point. i. Write an equation, including state symbols, to represent the enthalpy change of vaporisation of bromine. (1 mark)
Br2(l) → Br2(g) ✓
‘Enthalpy change of vaporisation’ is the enthalpy change when one mole of a substance changes from a liquid to a gas at its boiling point. Suggest whether the enthalpy change of vaporisation of bromine is exothermic or endothermic. Explain your answer. (1 mark)
endothermic AND energy required to overcome london forces

5.10 tonnes of NH3 are converted into NO. Calculate the energy released, in kJ, for this conversion. Give your answer in standard form and to an appropriate number of significant figures. (4 marks)

Average bond enthalpy is the enthalpy change for the breaking of 1 mole of bonds in gaseous molecules. Why do Br2 and I2 not exist in the gaseous state under standard conditions? (1 mark)
because energy is needed to break london forces between molecules

Calculate the enthalpy change of formation, △fH, for IBr. (2 marks)
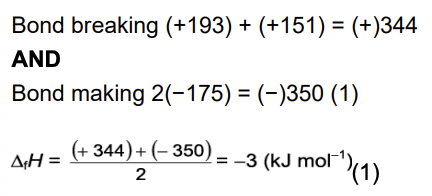
Explain, in terms of bond breaking and bond forming, why a reaction can be exothermic (1 mark)
more energy is released by forming bonds than energy required when breaking bonds
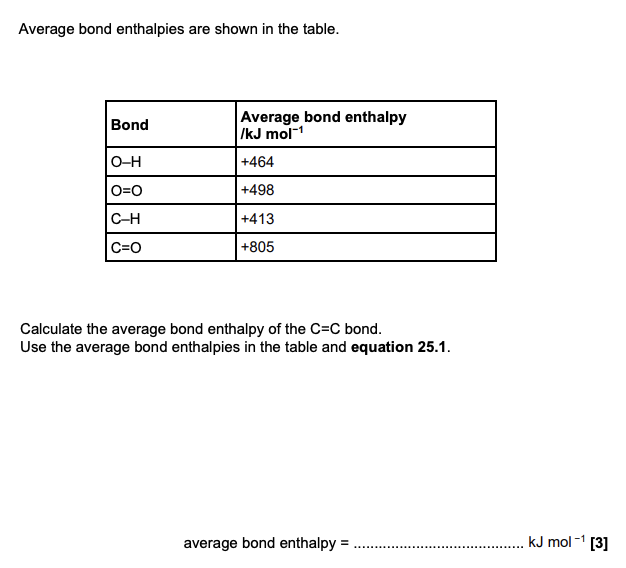
C2H4(g) + 3O2(g) → 2CO2(g) + 2H2O(g) ΔH = −1318 kJ mol −1 equation 25.1



The calculated value of ΔcH for hexane from this experiment is different from the data book value. Suggest two reasons for this difference. (2 marks)
heat released to the surroundings
incomplete combustion
non-standard conditions




ii) minimum mass: 0.0125 × 24.3 × 1.25 = 0.38g

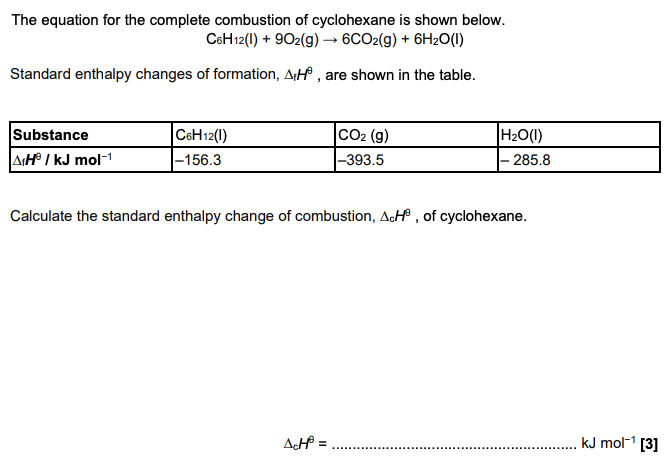
Define standard enthalpy change of combustion. Include the standard conditions that are used. (3 marks)
enthalpy change that occurs when one mole of a substance
completely combusts
25C AND 100kPa

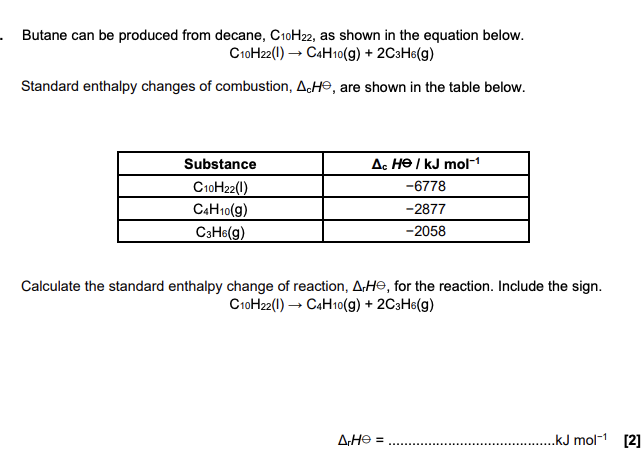
The equation that represents the enthalpy change of formation, ΔHf, of nonane is shown below. Calculate the standard enthalpy change of formation of nonane. 9C(s) + 10H2(g) → C9H20(l) (2 marks)

What is meant by the term average bond enthalpy? (2 marks)
average enthalpy change when one mole of bonds
of gaseous covalent bonds is broken

Calculate the bond enthalpy for the bond in carbon monoxide. Show your working. (3 marks)





Explain, in terms of bond breaking and bond making, why the enthalpy change of formation of NO is endothermic. (1 mark)
more energy is required for bond breaking than is released by bond making

The calculated value of ΔHc from this experiment is different from the value obtained from data books. Apart from heat loss, suggest two reasons for the difference. Assume that the calculation has been carried out correctly (2 marks)
incomplete combustion
non-standard conditions
evaporation of alcohol/ water




What is meant by the term standard enthalpy change of formation? Give the standard conditions. (3 marks)
enthalpy change when one mole of a compound
is formed from its elements
25C AND 100kPa
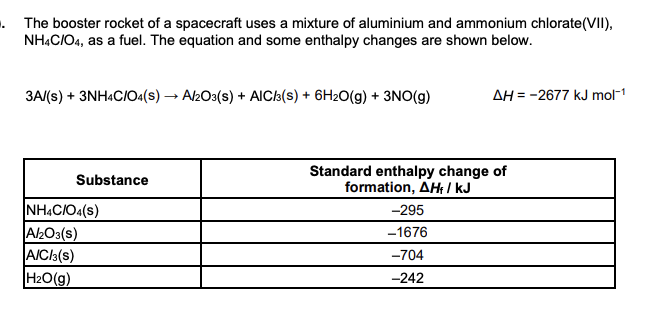
Write the equation, including state symbols, for the reaction that represents the standard enthalpy change of formation of NH4ClO4(s). (2 marks)
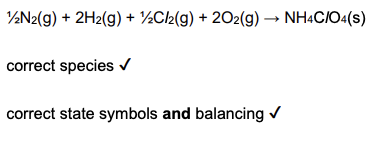
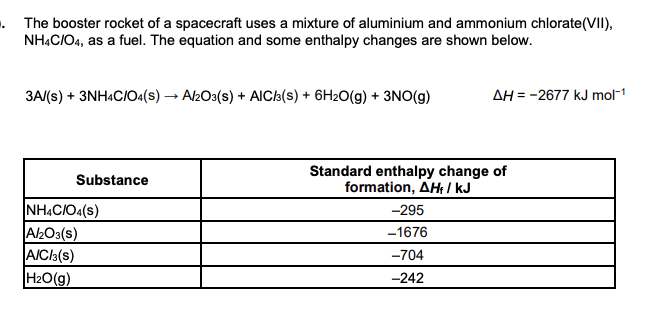
Calculate the enthalpy change of formation of NO(g) using the data above. (3 marks)

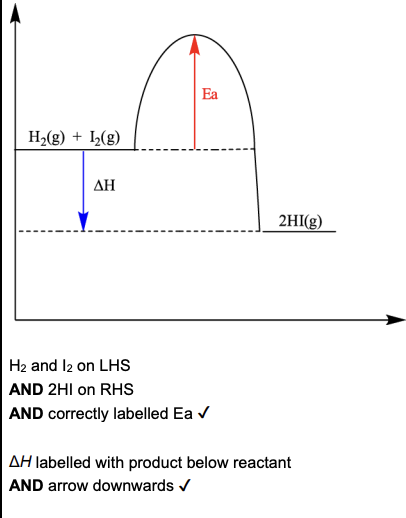
Calculate the activation energy, Ea, for the reverse reaction. (1 mark)
+182kJmol-1

When the reverse reaction takes place hydrogen iodide, HI, decomposes to form iodine and hydrogen. Calculate the enthalpy change when 336 dm3 of hydrogen iodide, measured at room temperature and pressure, decomposes. Include the sign for enthalpy change in your answer (2 marks)
number of moles of HI: 14 moles
enthalpy change: +63kJ

calculate the bond enthalpy for the H-I bond given that the bond enthalpy of H-H is 436 and I-I is 151 (2 marks)


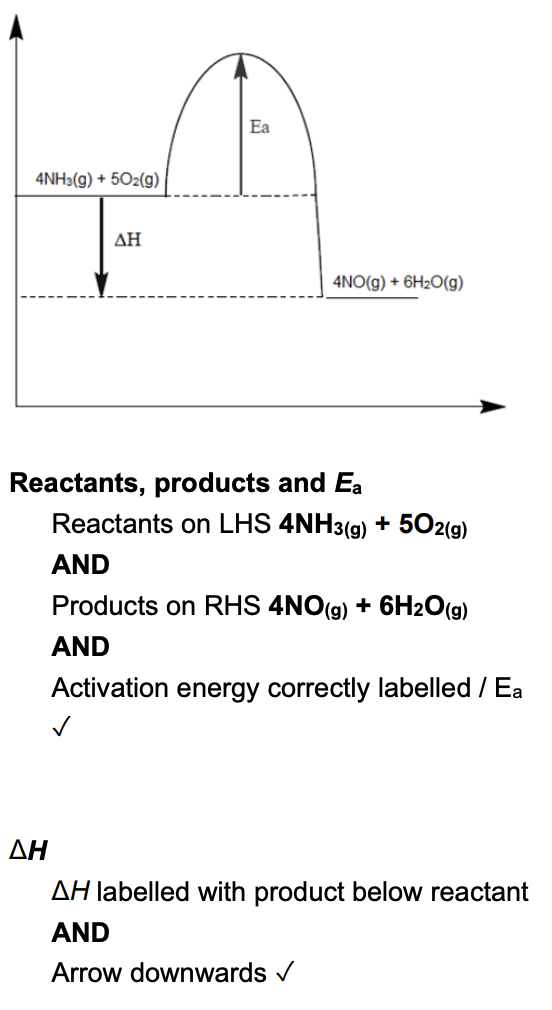
draw an enthalpy profile diagram for the forward reaction (2 marks)



In this experiment, the student uses a thermometer with an uncertainty of ±0.5 °C in each reading. Calculate the percentage uncertainty in the temperature rise. (1 mark)
(2×0.5) divided by 16.5 × 100 = 6%
Explain why this equation represents the standard enthalpy change of combustion of butane (1 mark)
One mole of butane completely combusts in oxygen

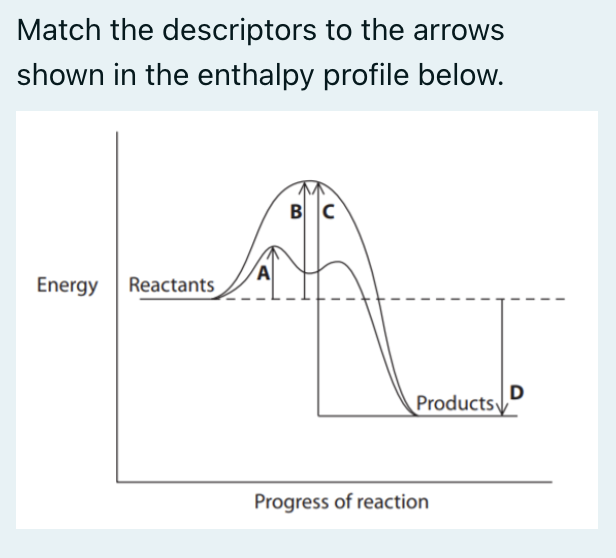
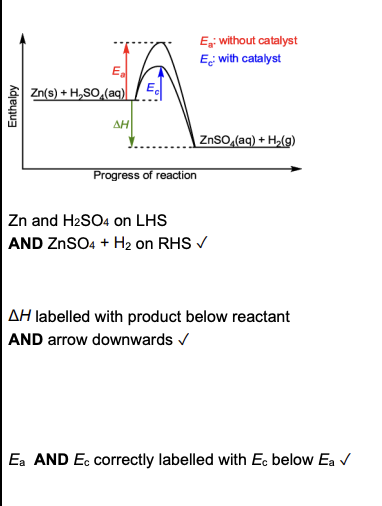
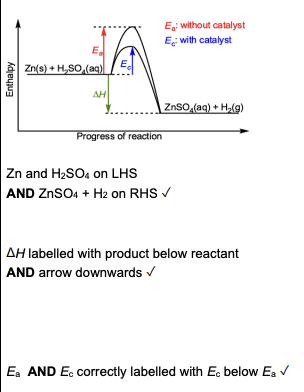
A: activation energy for a catalysed reaction
B: activation energy for the forward reaction
C: activation energy for the reverse reaction
D: enthalpy change for the forward reaction (exothermic)

B: 1 & 2

B: 1 & 2
1. Bond enthalpy of the C–H bond: Endothermic. Breaking bonds always requires an input of energy to overcome the attractive forces between atoms.
2. First ionisation energy of sodium: Endothermic. Energy must be supplied to remove an electron from the attraction of the nucleus in a gaseous atom.
3. Standard enthalpy change of formation of
: Neither. By definition, the enthalpy of formation for any element in its standard state is zero, so it is neither endothermic nor exothermic.

D: 1
1. It is equal to the standard enthalpy change of combustion of carbon: Correct. Both the standard enthalpy of formation of
and the standard enthalpy of combustion of carbon are defined by the same chemical equation
2. It is equal to twice the bond enthalpy of the
bond: Incorrect. Enthalpy of formation involves starting from elements in their standard states (solid graphite and gaseous
). Bond enthalpy calculations would also need to account for the energy required to atomize the carbon and break the
bonds.
3. It is the same for carbon dioxide produced from graphite and from diamond: Incorrect. "Standard" enthalpy of formation specifically requires using the most stable allotrope (graphite). Since diamond and graphite have different internal energies, burning them releases different amounts of heat.

D: only 1

C: 6C

B: only 1 & 2
1. Enthalpy change of formation: Correct.
This reaction shows exactly one mole of a compound () being formed from its constituent elements (
and
) in their standard states.
2. Enthalpy change of combustion: Correct.
This reaction shows exactly one mole of a substance () reacting completely with oxygen.
3. Enthalpy change of neutralisation: Incorrect.
While water is a product, neutralisation specifically refers to the reaction between an acid and an alkali (usually in aqueous solution) to produce one mole of water.
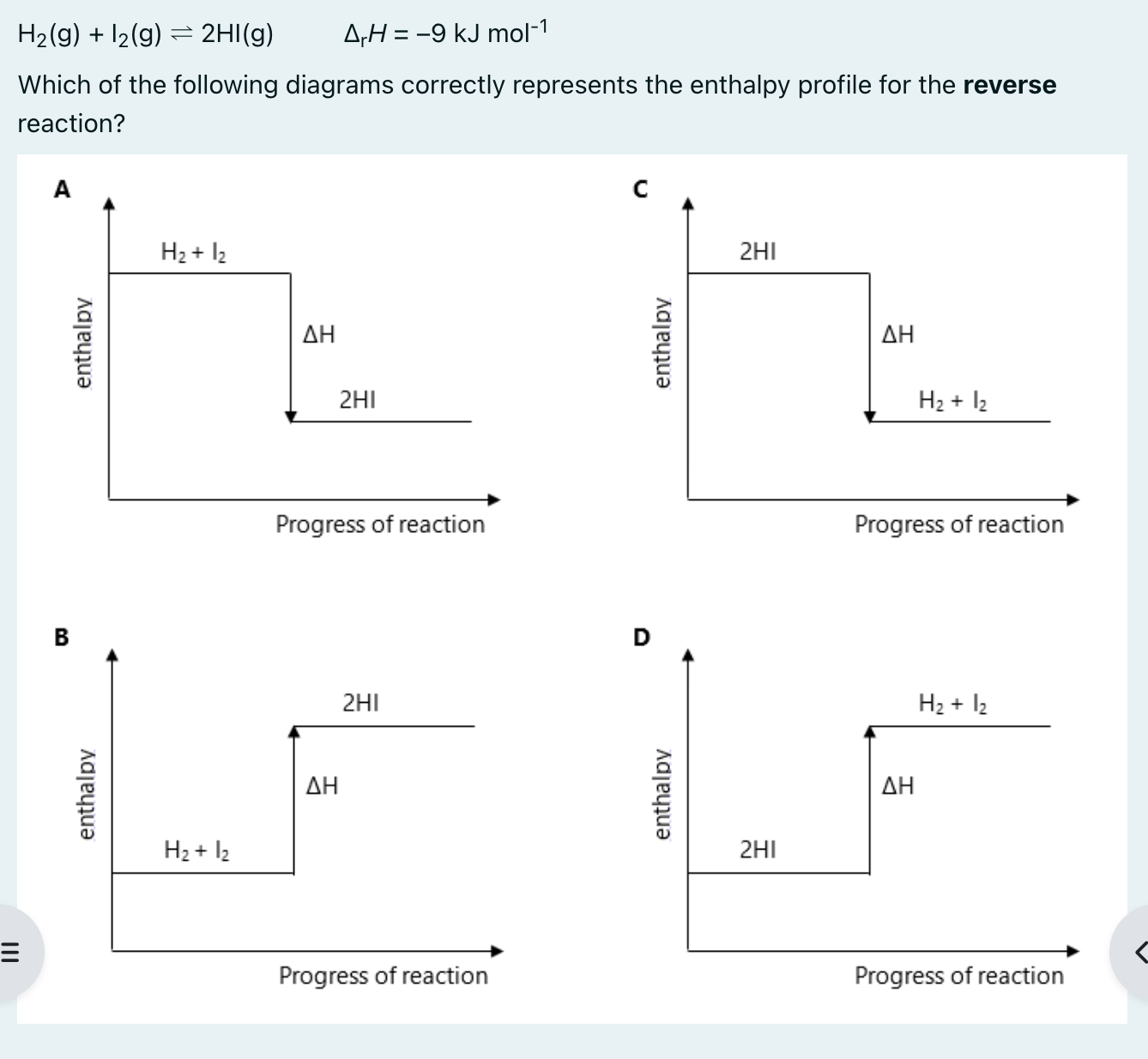
D

+298kJmol-1

For any reversible reaction, the enthalpy change () is equal to the difference between the activation energy of the forward reaction and the reverse reaction:
→ 173 - (-9) = 182 kJ mol-1
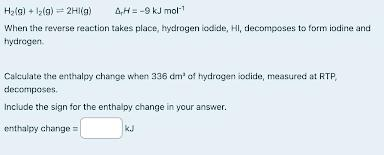
enthalpy change: +63kJ

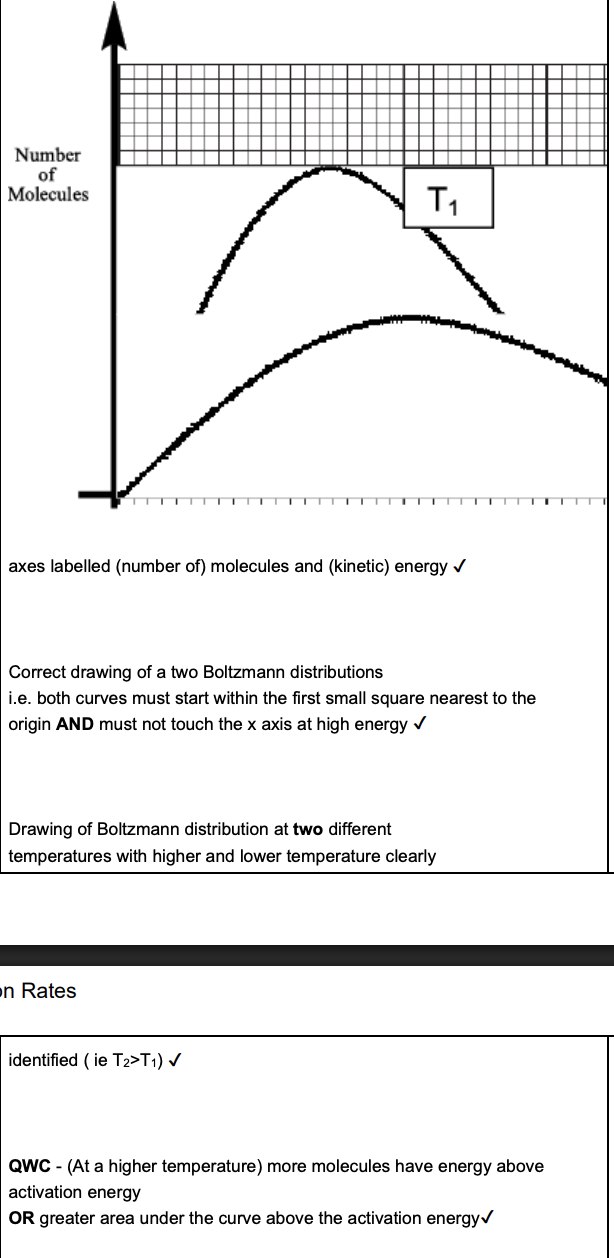
Rate of reaction decreases over time. Explain why, in terms of collision theory (2 marks)
acid concentration decreases
less frequent collisions per unit time
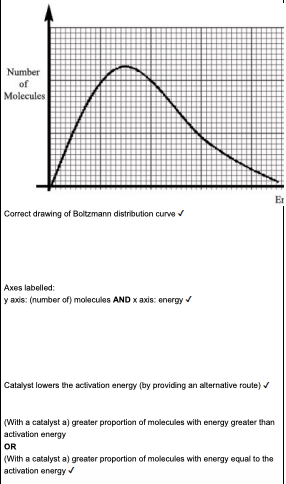
Explain how a catalyst increases the rate of reaction. (2 marks)
catalyst lowers the activation energy by providing an alternative route
a greater proportion of molecules have more energy greater than activation energy
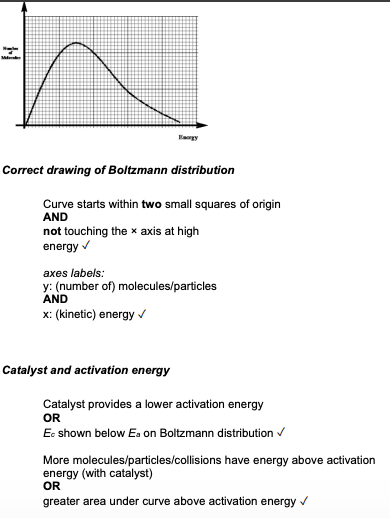
Draw the Boltzmann distribution model and explain how the rate of a reaction is affected by temperature. (4 marks)

Describe and explain the effect of increasing the pressure on the rate of this reaction. (2 marks)
increase in pressure increases rate of reaction AND because molecules are closer together
so there are more collisions per unit time
Using the Boltzmann distribution model, explain why the rate of a reaction increases in the presence of a catalyst. (4 marks)
Explain why use of the catalyst reduces energy demand and benefits the environment (2 marks)
lower temperatures
less fossil fuels
reduces CO2 emissions


Using a Boltzmann distribution, explain how a catalyst increases the rate of a chemical reaction. Include a labelled sketch of your Boltzmann distribution on the grid below. Label the axes and any other important features. (4 marks)

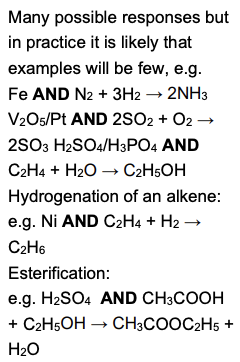
State an example of a catalyst used by the chemical industry and write the equation for the reaction that is catalysed. (1 mark)
State two ways that the use of catalysts helps chemical companies to make their processes more sustainable and less harmful to the environment. (2 marks)
lower temperatures
reduces CO2 emissions
different reactions can be used with less waste OR can reduce use of toxic reactants
Describe and explain the effect of increasing the pressure on the reaction rate. (2 marks)
increased rate AND greater concentration of molecules per unit volume
more frequent collisions per unit time
Explain why the use of a catalyst can reduce the demand for energy. (1 mark)
allows reactions to take place at lower temperatures
Describe and explain the effect of decreasing the pressure on the rate of a reaction. (2 marks)
decreases rate of reaction AND decreases concentration of molecules per unit volume
less frequent collisions per unit time
The use of catalysts in industrial processes can be beneficial to the environment. State one reason for this. (1 mark)
catalysts lower energy demand for reaction OR less combustion of fossil fuels and therefore lower carbon dioxide emissions