Food Science: Sugar Properties, Crystallization, and Candy Making Techniques
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
What is the primary source of granulated sugar?
Sugar beets and sugar cane
What is powdered sugar also known as?
Confectioners' sugar
What distinguishes brown sugar from other sugars?
It has a molasses film, which can be dark or light.

Name two types of syrups mentioned.
Molasses and maple syrup
What are low-calorie sweeteners also known as?
High intensity sweeteners
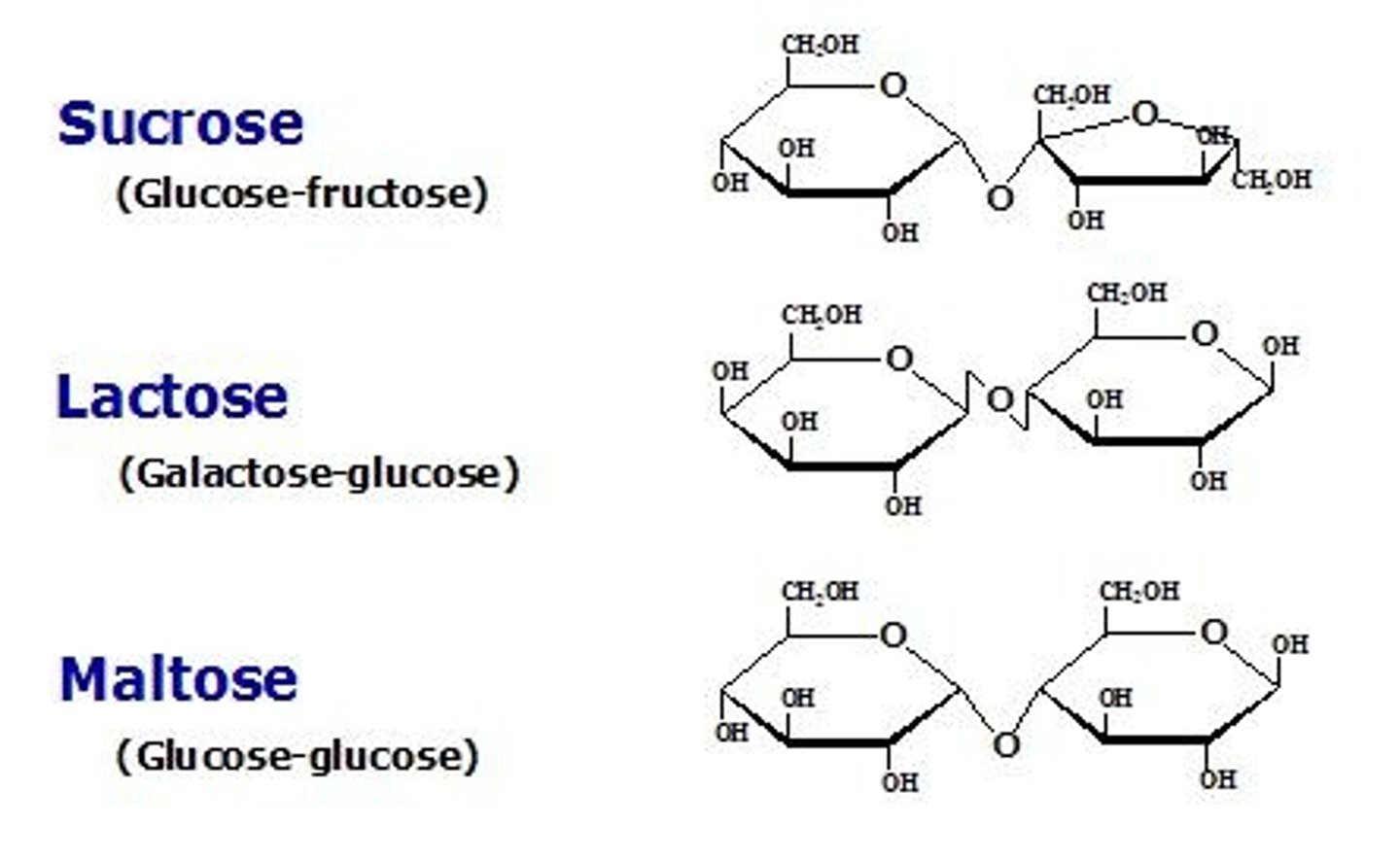
What is invert sugar?
Sucrose converted to fructose and glucose, more soluble and helps prevent crystallization.
What reactions do sugars participate in that result in browning?
Maillard browning and caramelization
What is the role of invertase in sugar processing?
It helps convert sucrose into invert sugar.
What is the relative sweetness of fructose compared to sucrose?
Fructose is 1.16 times sweeter than sucrose.
What happens to sugars when heated above 337°F (170°C)?
They dehydrate and decompose, creating flavors and brown pigments.
What is the main difference between caramelization and the Maillard reaction?
Caramelization does not require proteins, while the Maillard reaction does.
What is the caloric value of sugar?
4 calories per gram.
What is the impact of sugar concentration on boiling point?
Higher sugar concentration raises the boiling point.
How do different sweeteners affect texture in food products?
Different sweeteners vary in sweetness and solubility, impacting texture.
What is a common misconception about artificial sweeteners?
That they are worse than full sugar alternatives due to being 'not-organic'.
What is the effect of heating sugar on flavor?
Heating changes the flavor profile, making it more complex and sometimes bitter.
What is the significance of the cold-water test in sugar properties?
It helps determine the solubility and crystallization properties of sugars.
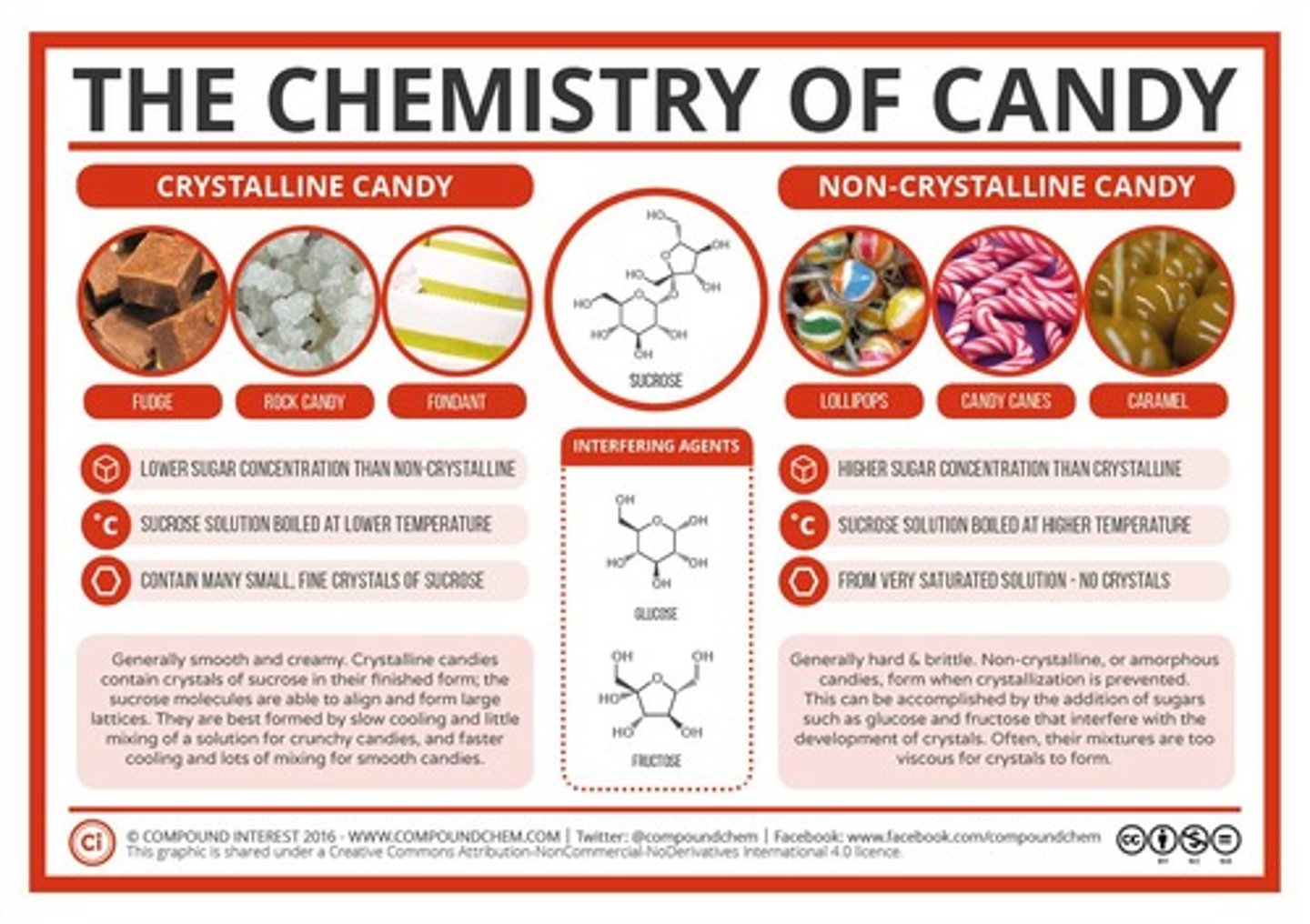
What are the two types of confections mentioned?
Crystalline and amorphous confections.
What is one factor that influences sugar crystal formation?
The type of sweetener used.
What is the primary function of invert sugar in confections?
To keep candies smooth and prevent graininess.
What is the role of amino acids in the Maillard reaction?
They react with reducing sugars to create flavors and brown pigments.
What is a fun fact about caramelization?
Maillard browning is responsible for the brown color of caramel, not caramelization itself.
What is the sweetness level of glucose compared to sucrose?
Glucose is 0.64 times as sweet as sucrose.
What is one question students have about reduced-sugar confections?
How do manufacturers decide which sweetener to use?
How does sugar concentration affect boiling point?
Higher sugar concentration increases the boiling point.
What is the effect of sugar on water molecules?
Sugar holds on to water molecules, disrupting crystallization.
What are colligative properties?
Properties that depend on the concentration of solute, such as boiling and freezing points.
What does hygroscopic mean in relation to sugar?
Hygroscopic means sugar can absorb water, making sugary foods sticky in humid conditions.
How does temperature affect sugar solubility?
Higher temperatures increase sugar solubility.
What is a supersaturated solution?
A solution that contains more solute than it can hold at a given temperature.
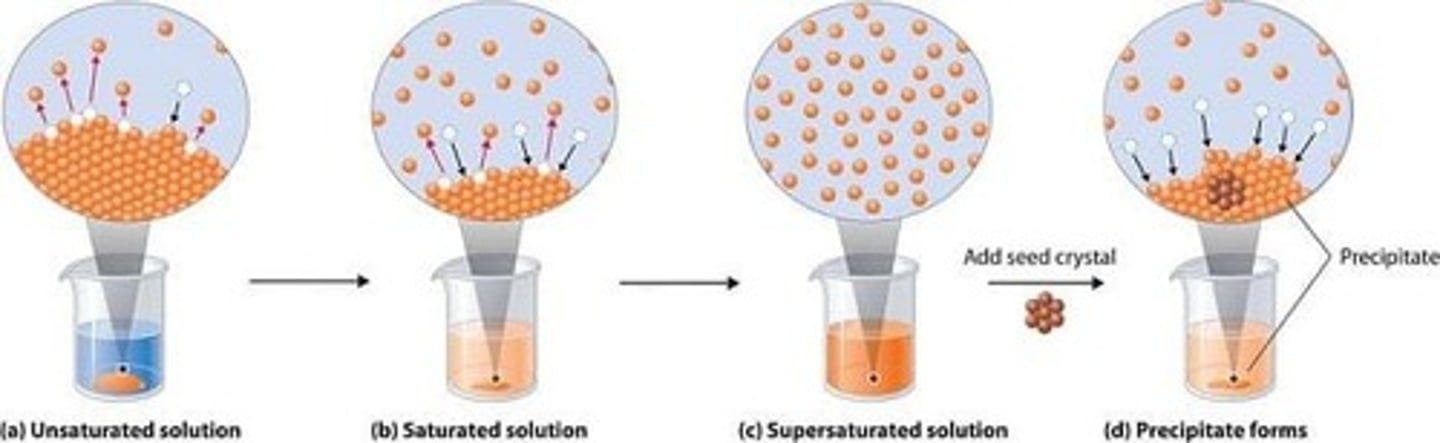
What happens when a hot sugar solution cools?
Sugar precipitates out of the solution.
What is the significance of controlling crystal formation in candy making?
Controlling crystal formation affects the texture and quality of the candy.
What is the cold-water test used for?
To assess sugar concentration in candy making.
What factors impact crystallization in candy making?
Seeding, sugar concentration, temperature, cooling method, agitation, and interfering substances.
What are interfering substances in candy making?
Ingredients that disrupt sucrose crystallization, such as fats and proteins.
What is the difference between crystalline and amorphous candies?
Crystalline candies have a structured pattern and form around seed crystals, while amorphous candies are too viscous for crystal formation.
What role do seed crystals play in candy making?
Seed crystals serve as a starting point for crystal growth.
How does cooling method affect crystal size in candy making?
Slow, undisturbed cooling allows for larger crystals, while agitation keeps crystals small.
What is the effect of invert sugar on crystallization?
Invert sugar has higher solubility than sucrose, which decreases crystallization.
What is the function of baking soda in peanut brittle?
It neutralizes acid, lightens caramelization pigments, and adds CO2 for a porous texture.
What is the purpose of calibrating thermometers in candy making?
To ensure accurate temperature readings for candy preparation.
What happens to a supersaturated solution when cooled?
It precipitates, causing sugar to crystallize.
What is the texture of fudge influenced by?
The texture of fudge is influenced by crystal size and the presence of interfering agents.
What is the impact of agitation on amorphous candies?
Agitation is not crucial for amorphous candies as they are too viscous for crystal formation.