Chemistry - Gases in our atmosphere
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Nitrogen percentage in dry air
78.1%
Oxygen percentage in dry air
20.96%
Argon percentage in dry air
0.9%
Carbon dioxide percentage in dry air
0.04%
Why do we measure in dry air?
The numbers will not change and are the same all around the world. Wet air changes based on the weather e.g. storm
Calculating the percentage of oxygen in the air by using the rusting of iron
4Fe + 3O2 + XH2O → 2Fe2O3.XH2O = Hydrated iron oxide. The iron has to be wet as it takes thousands of years to rust if its dry. Because there is both a solid reactant and product, it can stall as the product can coat the reactant.
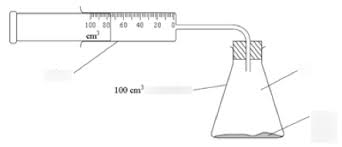
Using phosphorus to calculate the percentage of oxygen in the air
P2O5 + 3H2O → 2H3PO4. Calculate how much the height of the water changes to measure the volume. This method gives the best results. This is because the product is a non-metal oxide, which means there is an acidic gas, it then diffuses away and more phosphorus reacts.
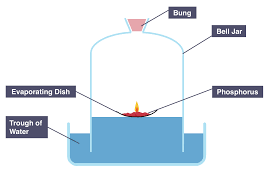
What happens when magnesium combusts?
There is a bright white light and a white solid is formed. 2Mg (s) + O2 (g) → 2MgO (s)
Equation for magnesium oxide with water?
MgO (s) + H2O (l) → Mg(OH)2 (aq)
What happens when sulfur combusts?
There is a blue / purple flame and a colourless gas forms. 2S + 3O2 → 2SO3
Equation for sulfur oxide with water?
SO3 (g) + H2O (l) → H2SO4 (l)
What happens when hydrogen combusts?
There is a squeaky pop. It’s used in rocket fuel. 2H2 (g) + O2 (g) → 2H2O (g). It’s a very hot exothermic reaction
How can carbon dioxide be produced?
By thermal decomposition of some metal carbonates. A → B + C
Carbon dioxide produced by thermal decomposition of metal carbonates experiment
The limewater goes cloudy. Ca(OH)2 (ag) + CO2 (g) → CaCO3 (s) + H2O (l).
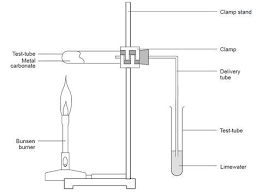
Why is carbon suitable for downwards delivery?
It’s more dense than air
Why is carbon used on fire extinguishers?
It acts like a blanket, pushing the air out to the sides, so that the fire is deprived of oxygen
Decomposition of copper carbonate
Copper carbonate → copper oxide + carbon dioxide
CuCO3 → CuO + CO2
Colour of CuCO3
Green
Colour of CuO
Black
Colour of CuSO4
Blue
Colour of Cu(OH)2
Pale blue
What is carbon dioxide?
A greenhouse gas and increasing amounts in the atmosphere may contribute to climate change