P5.2.3 Radioactive decay
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
What is radioactive decay?
Radioactive decay: a spontaneous, random change of an isotopes nucleus that can result in the emission of ionizing radiation
What happens to the nucleus during α-decay or β-decay?
A nucleus changes into a different element during α-decay or β-decay
Initial nucleus is called parent nucleus
Resulting nucleus is called daughter nucleus
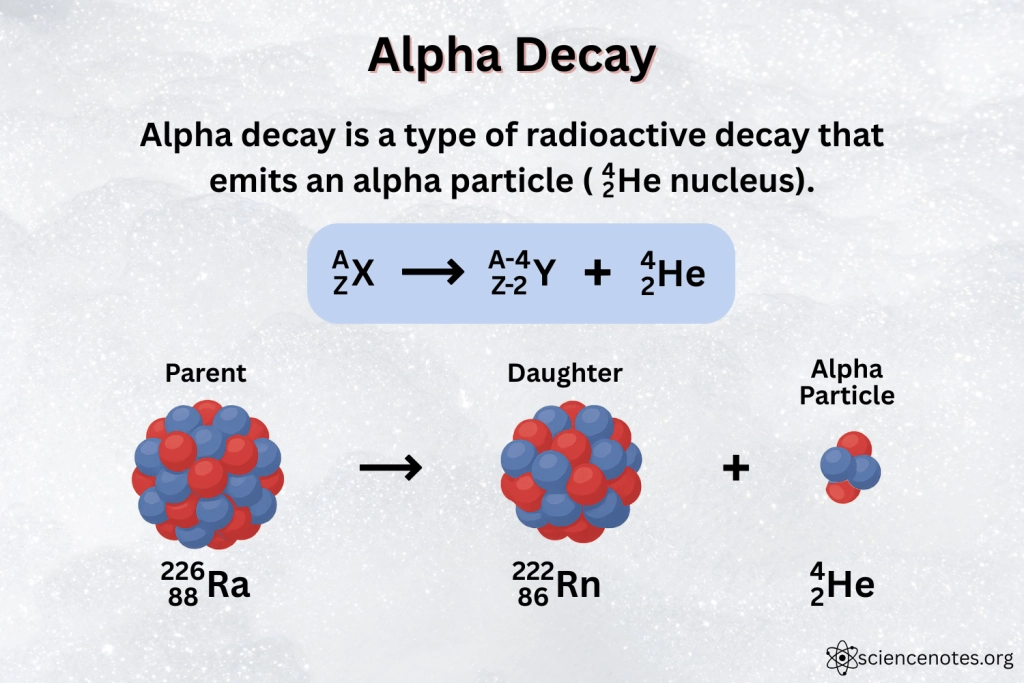
What happens during alpha decay?
When alpha particle is emitted from isotope, mass number and atomic number of nucleus changes
Mass number: decreases by 4
Atomic number: decreases by 2
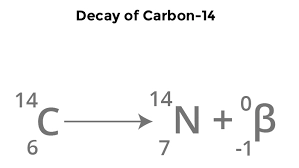
What happens during beta decay?
When beta particle is emitted from isotope, a neutron changes into a proton and an electron
Mass number: stays the same
Atomic number: increase by 1
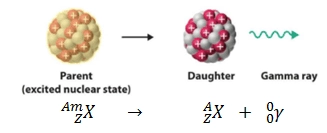
What happens during gamma decay?
When gamma ray is emitted from isotope, the nucleus becomes less energetic but there is no change to its structure
This is because gamma rays have no mass or charge