Chap 21C - Hydroxy
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Describe condensation of alcohols with carboxylic acids
Equation | alcohol + carboxylic acid ⇌ ester + water Eg. CH3CH2OH + CH3COOH ⇌ CH3COOCH2CH3 + H2O ethyl ethanoate |
Reagents and conditions |
NOTE:
|
Describe condensation of alcohols with acyl chlorides
alcohol + acyl chloride → ester + hydrogen chloride gas |
Just add acyl chloride
|
Describe tri-iodomethane formation
This oxidation OR oxidative cleavage reaction applies ONLY to alcohols with a methyl group and a hydrogen atom attached to the carbon atom at which the hydroxy group is also attached
Reagents and conditions: I2(aq) in NaOH(aq), warm
Observation: Yellow or pale yellow precipitate of tri-iodomethane (iodoform)
Draw reaction for tri-iodomethane formation (use CRHCH3OH) (overall)
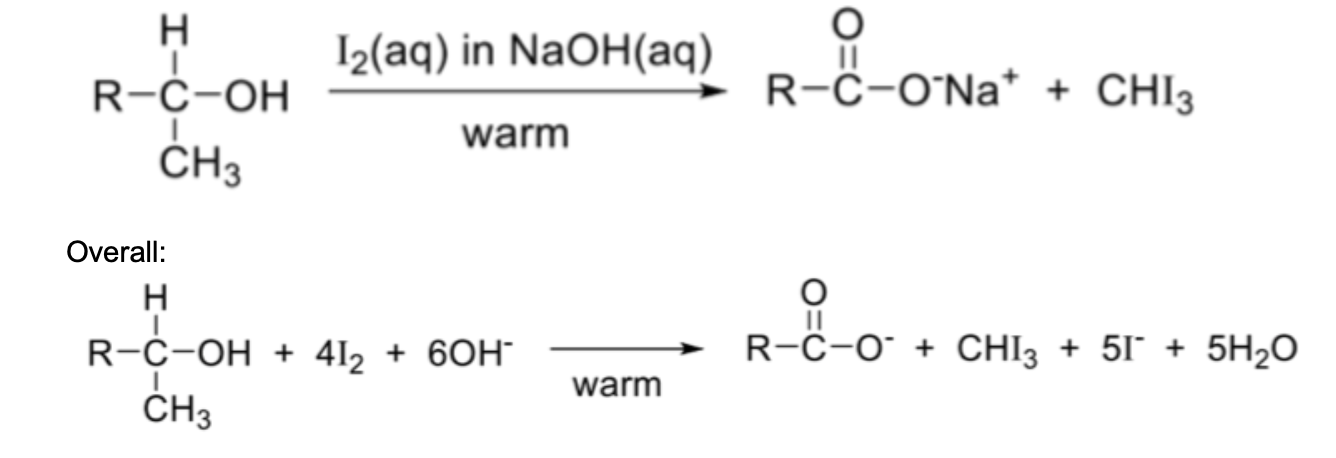
NOTE:
Ethanol is the ONLY primary alcohol to show positive iodoform test
The product RCOO– has one C atom less than the reactant RCH(OH)CH3 (step-down reaction) due to the formation of CHI3
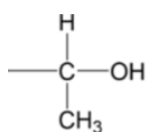
Test for alcohols which contain a (check image) group
Warm the alcohol with I2(aq) in NaOH(aq)
Observation:
If the alcohol contains a –CH(OH)CH3 group, a (pale) yellow precipitate (of CHI3) is formed
If –CH(OH)CH3 group is absent, no visible reaction is observed
NOTE: Carbonyl compounds containing a methyl carbonyl group also give a positive iodoform test
Describe phenols
Compounds containing a hydroxy group attached directly to an aromatic ring
Since the hydroxy group attached directly to the benzene ring -> lone pair of electrons on O is delocalised into the benzene ring -> C–O bond has partial double bond character
Describe appearance, bp and solubility of phenols
Appearance
Phenol is a colourless, hygroscopic and crystalline solid (m.p. 42 °C)
Boiling point
Higher bp than hydrocarbons with similar Mr due to intermolecular HB between molecules
Solubility
Partially soluble in water
The –OH group form HB with water but the large non-polar aryl group cannot
Solubility in water increases with temperature
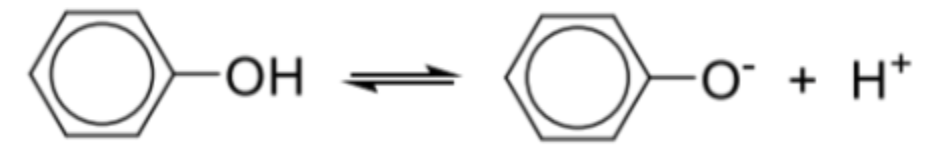
Describe aciditiy of phenol and why it is more acidic than water
Relative acidity: phenol > water > alcohol
Phenol can behave as an acid by donating protons
Greater acidity of phenol is due to the stabilisation of phenoxide anion (C6H5O–) by charge dispersal
The p-orbital of oxygen overlaps with the pi electron cloud of the benzene ring -> delocalisation of negative charge on the oxygen into the benzene ring -> disperses the negative charge on the O atom -> phenoxide anion is stabilised by charge dispersal
Hence, phenol is more acidic than water as such delocalisation effect and dispersion of charge is not found in water
Explain effect of electron-donating and withdrawing subsituents on acidity benzene ring
Electron-donating substituents | Electron-withdrawing substituents |
|
|

Explain which compound is more acidic
Comparing the conjugate bases of 2-chlorophenol and the conjugate base of phenol, the electron density on O atom is lower in the conjugate base of 2-chlorophenol due to the electron-withdrawing –Cl group
Hence, the conjugate base of 2-chlorophenol is more stable than the conjugate base of phenol -> 2-chlorophenol more acidic than phenol
State reactions of phenols
Reactions with hydroxy grp:
Redox with Na
Acid-base with NaOH
Acid-base with NaCO3
Elec sub at benzene:
Nitation
Halogenation