Ethers, Epoxides, & Sulfides Reactions
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
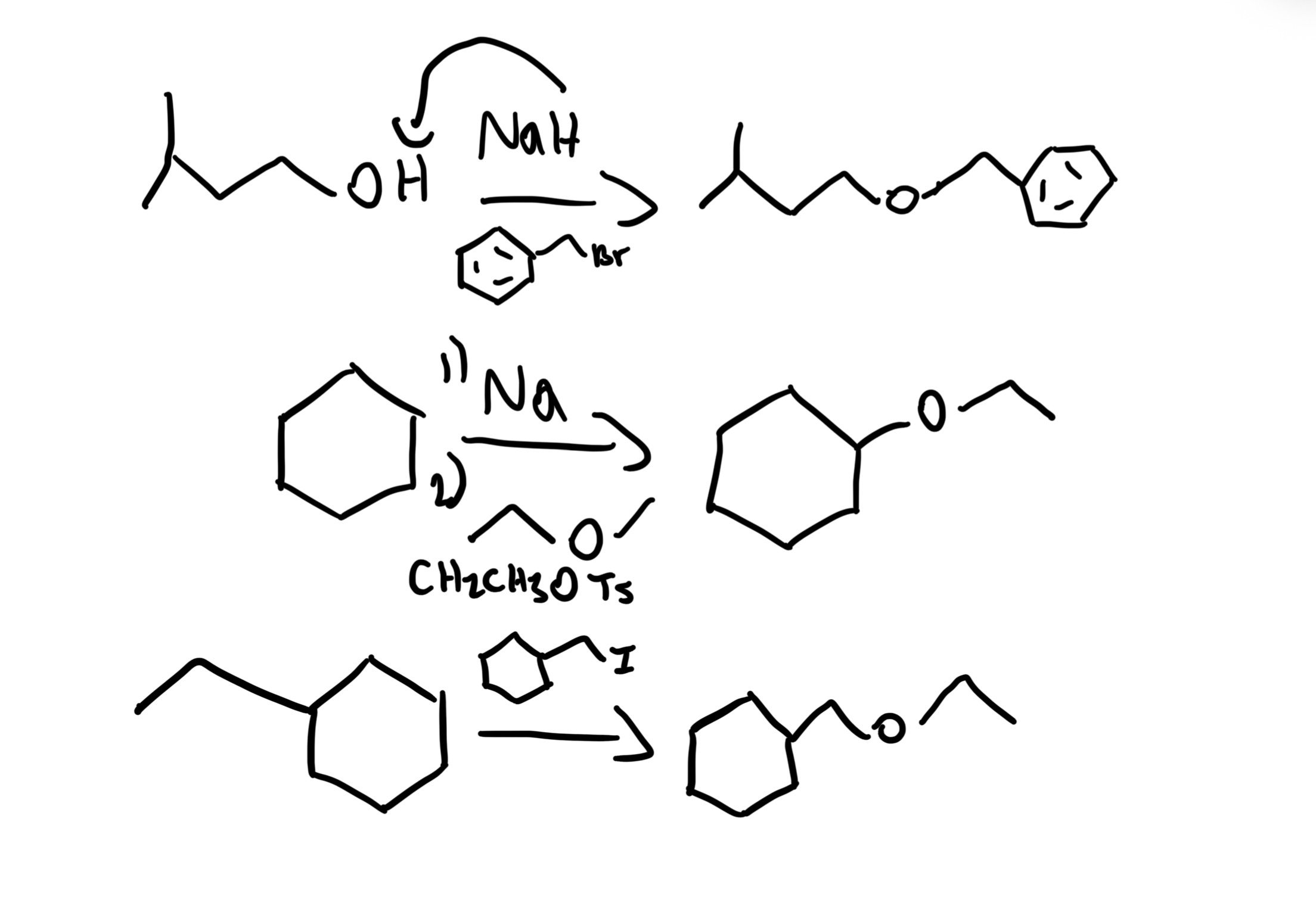
Williamson Ether Synthesis
Alkyl halide + Alkoxide (RO⁻) → R-O-R' (Ether) using NaH then R'X (SN2). Best with primary alkyl halides; secondary/tertiary give elimination.
Williamson Ether Synthesis (Phenyl)
Phenol + Alkyl halide → Ar-O-R (Aryl ether) using NaOH, then R-X. Phenoxide is nucleophile; works well with methyl/primary R-X.
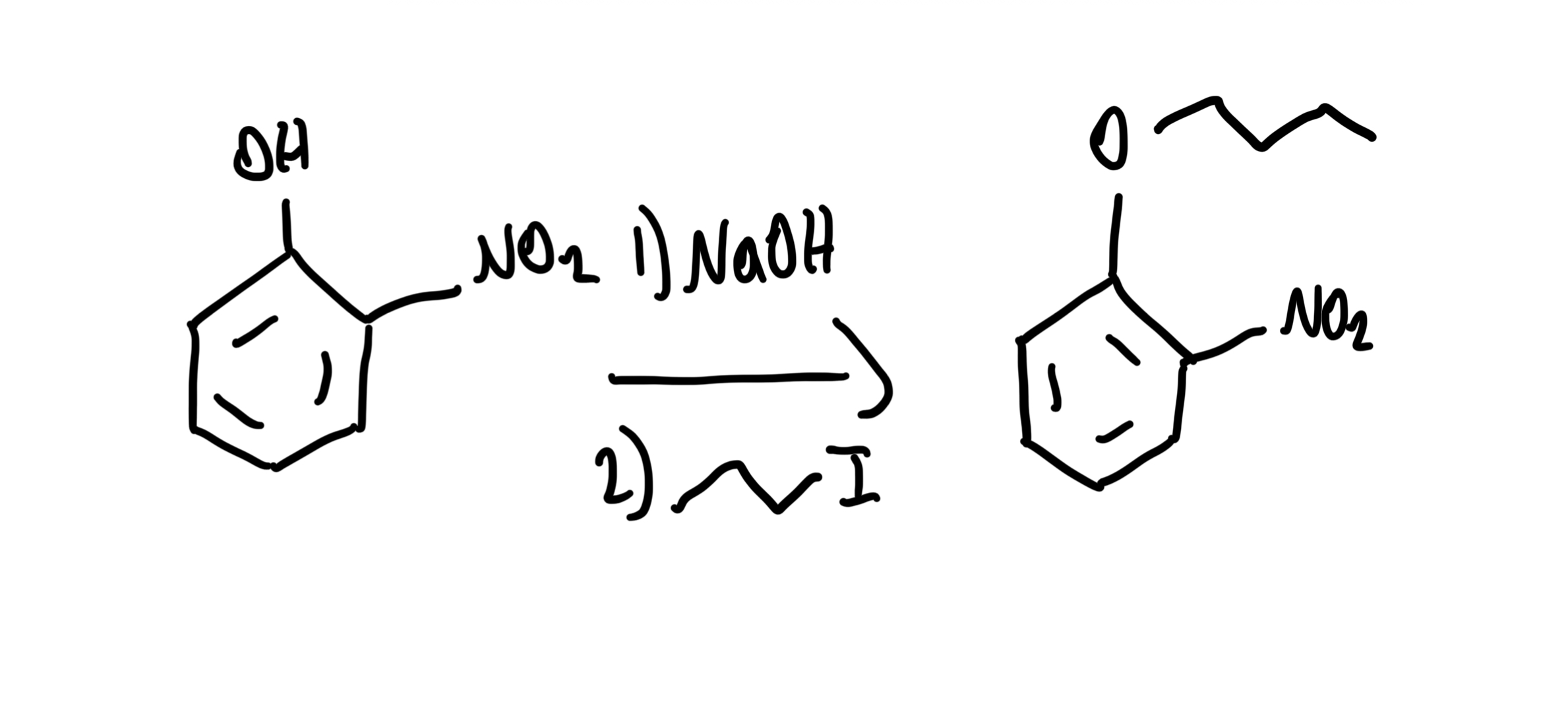
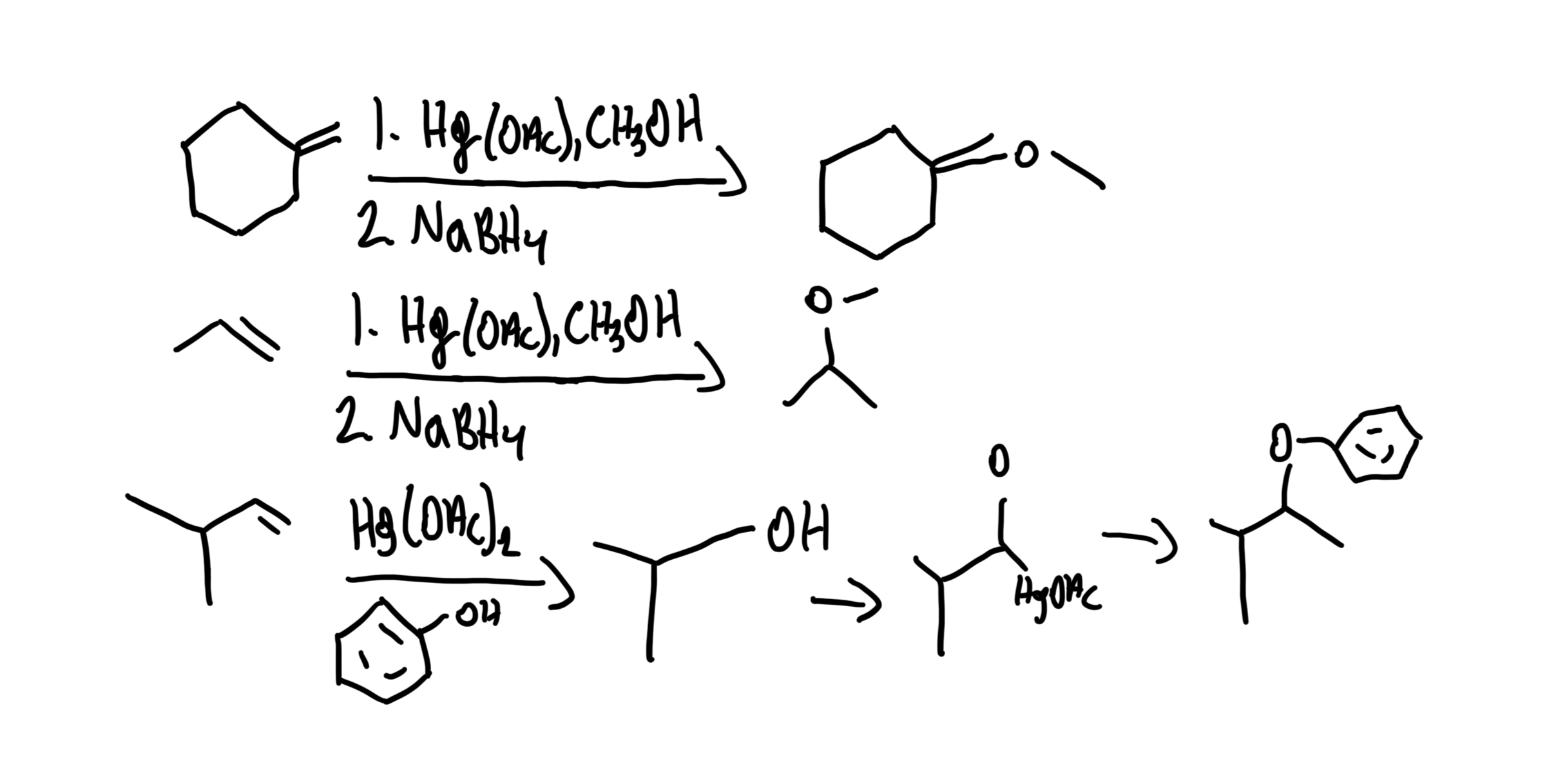
Alkoxymercuration-Demercuration
Alkene + Alcohol → Ether (Markovnikov) using 1) Hg(OAc)₂, ROH 2) NaBH₄. No rearrangements.
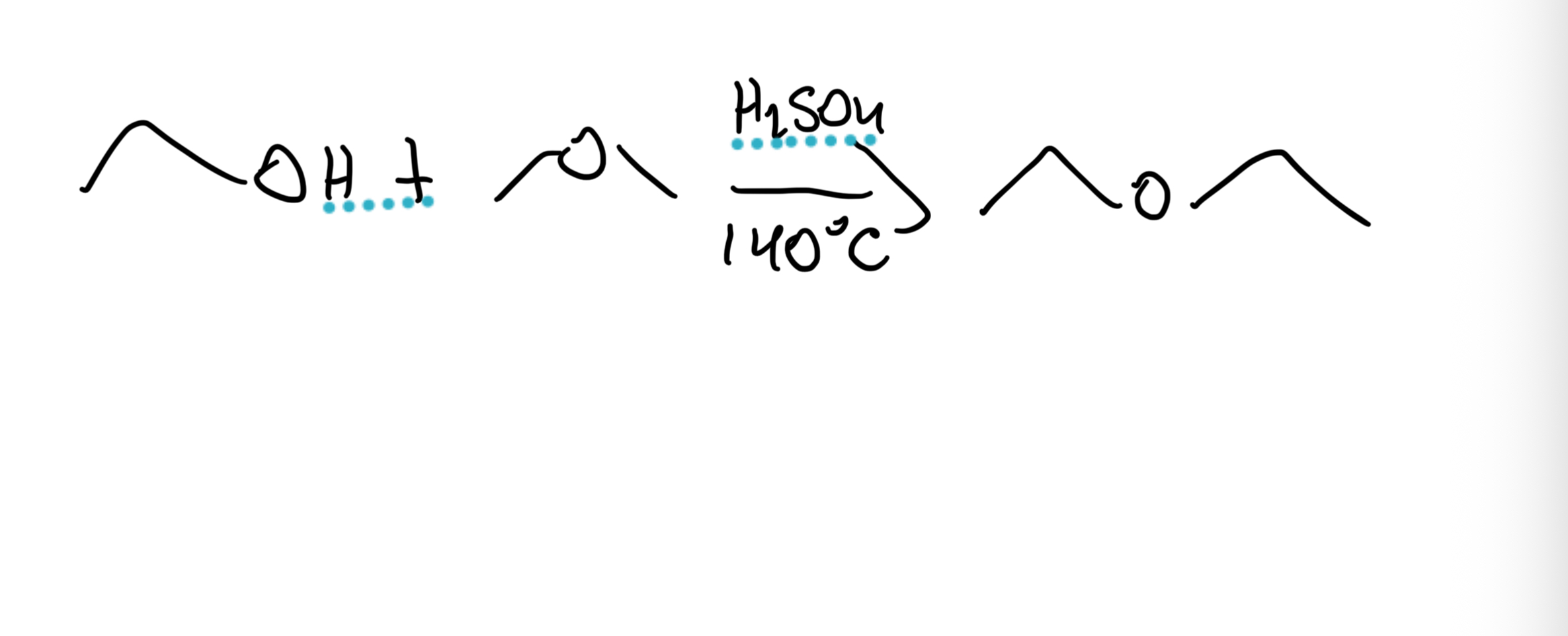
Industrial Ether Synthesis
Ethanol → Diethyl ether using H₂SO₄, 140°C. Condensation of 2 alcohols; works for symmetrical 1° ethers only.
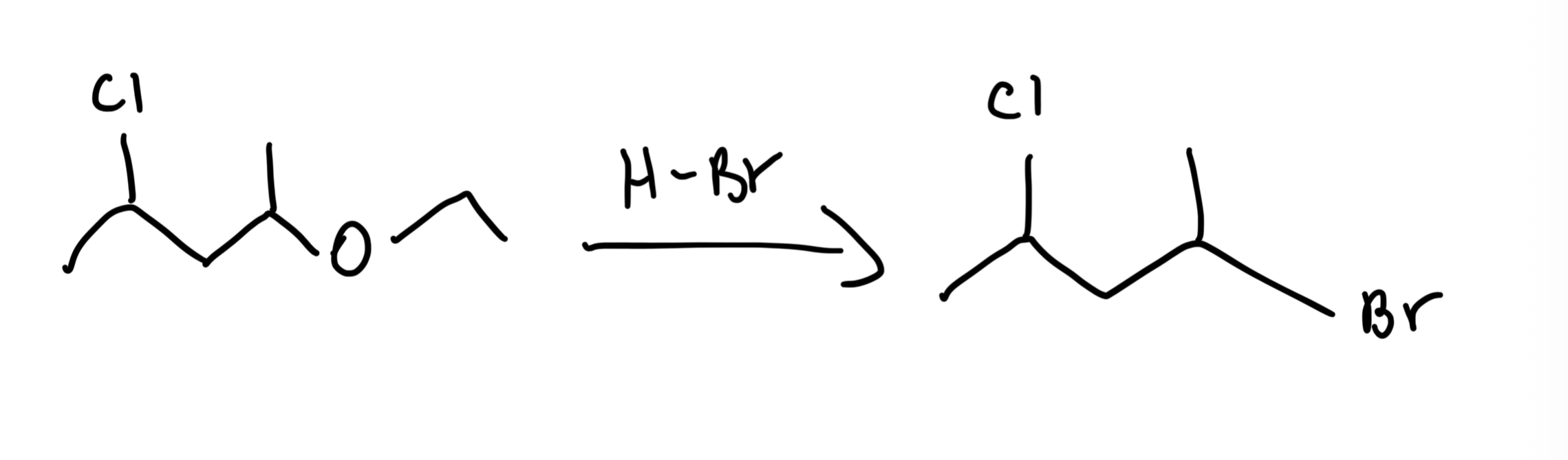
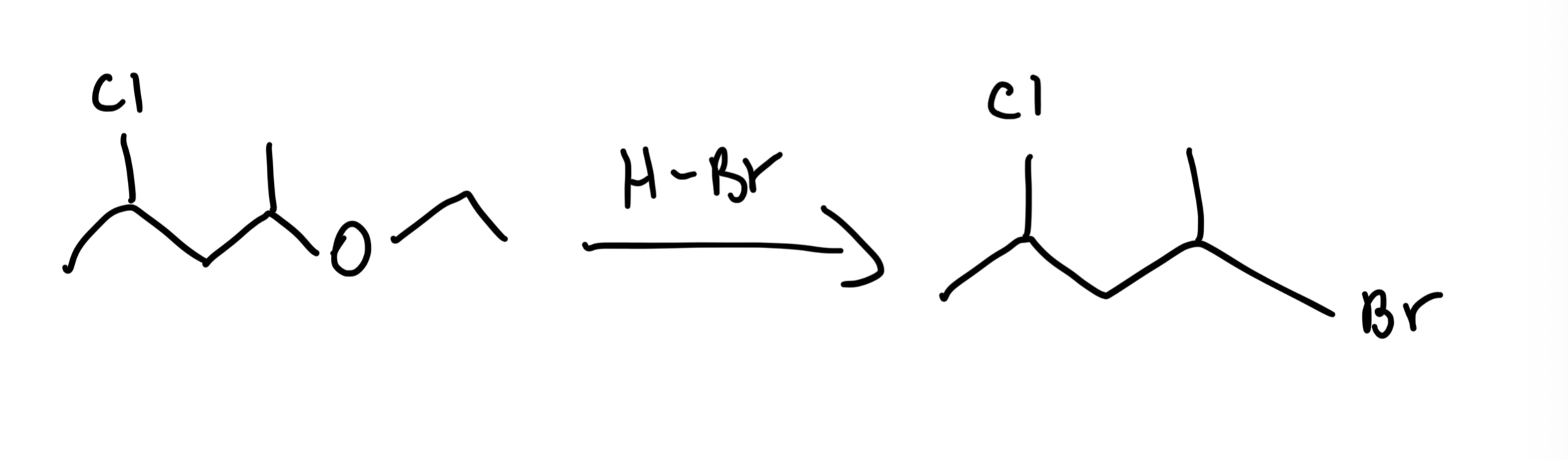
Ether Cleavage with HBr/HI
R-O-R' (Ether) → 2 R-X (Alkyl halides) using excess HBr or HI, heat. HI > HBr in reactivity; SN2 for 1°/2°, SN1 for 3°.
Ether Cleavage (Mixed)
R-O-R' (one 3° or aryl) → R-X + R'-OH using HBr or HI. Cleavage favors formation of more stable carbocation or phenol.
Autoxidation of Ethers
Diethyl ether / THF → Hydroperoxides (explosive) via O₂ (air) over time. Dangerous! Always test old ethers for peroxides before distilling.
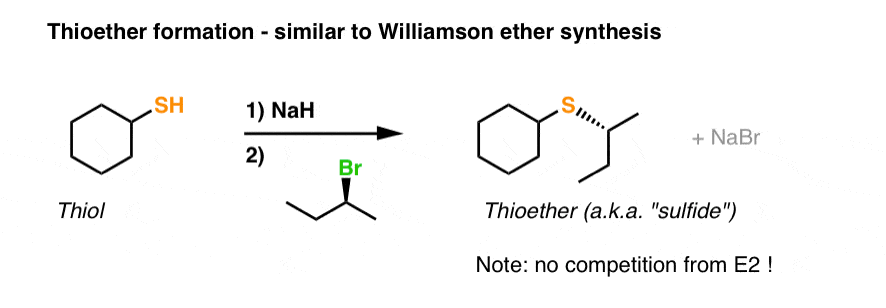
Thioether (Sulfide) Synthesis
Thiolate (RS⁻) + Alkyl halide → R-S-R' (Thioether) via Williamson-type SN2. Thiolates are better nucleophiles than alkoxides.
Sulfide → Sulfoxide
R-S-R' (Thioether) → R-SO-R' (Sulfoxide) using H₂O₂ (1 equiv). Mild oxidation; DMSO from DMS.
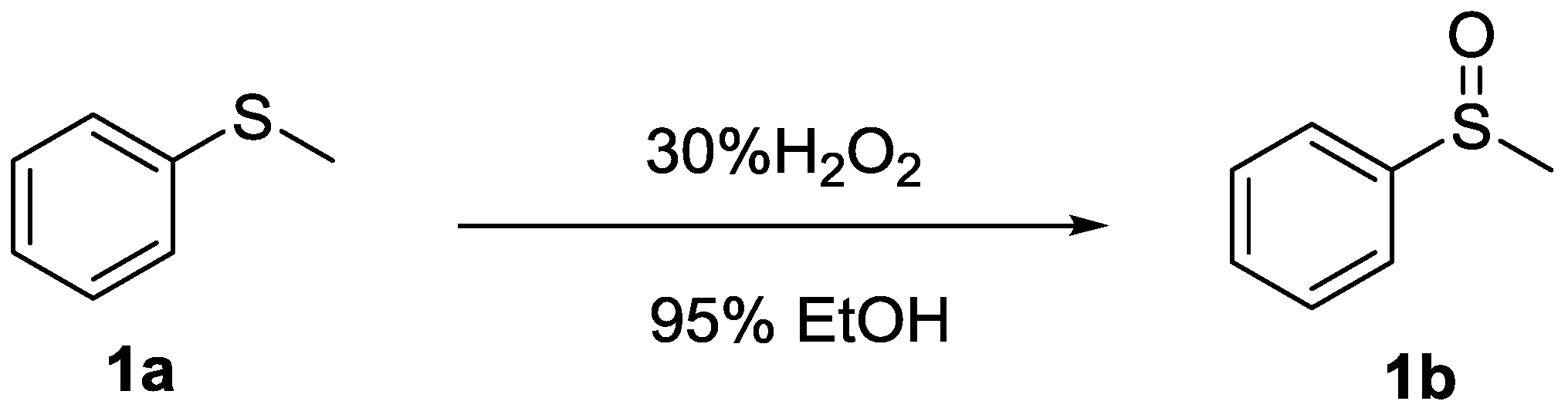
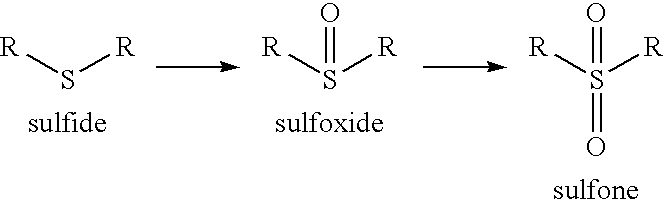
Sulfoxide → Sulfone
R-SO-R' (Sulfoxide) → R-SO₂-R' (Sulfone) using H₂O₂ (excess) or KMnO₄. Further oxidation of sulfoxide.
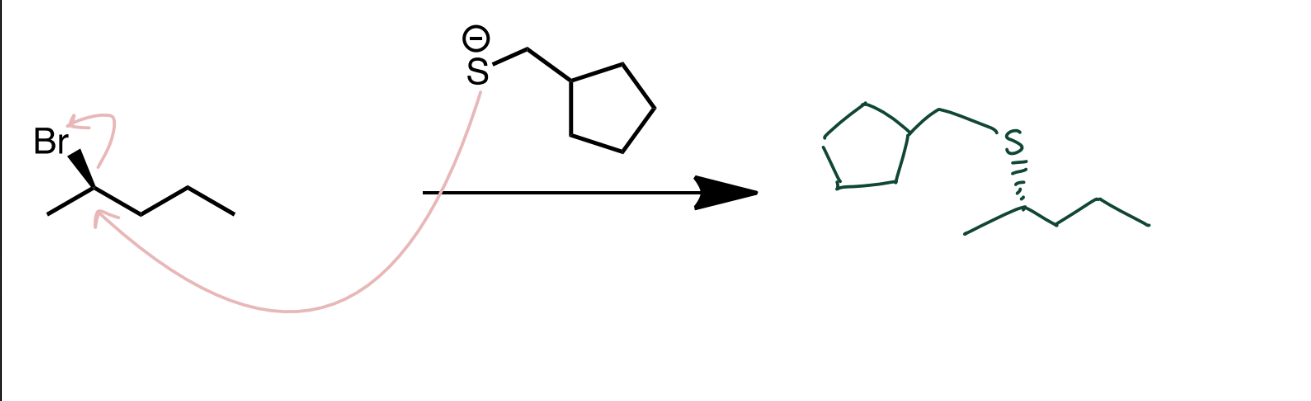
Sulfonium Salt Formation
R-S-R' (Thioether) → R-S⁺(R')(R'') (Sulfonium salt) using R''X. Sulfur acts as nucleophile.
Sulfonium Salt as Alkylating Agent
Sulfonium salt + Nu⁻ → R-Nu + R'-S-R'' via SN2. S-adenosylmethionine (SAM) is biological example.
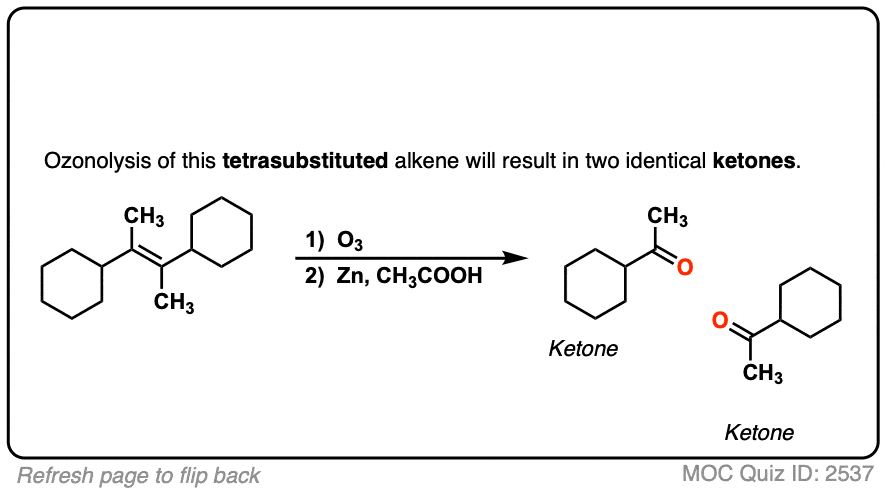
Ozonolysis Workup with DMS
Ozonide intermediate → Aldehydes/Ketones using (CH₃)₂S (DMS). DMS reduces ozonide; DMS oxidized to DMSO.
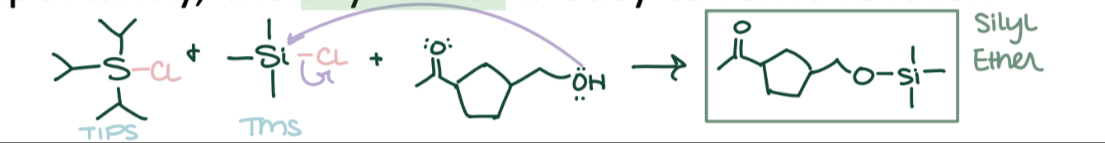
Silyl Ether Protection
Alcohol (R-OH) → R-O-TBDMS (Silyl ether) using TBDMSCl, imidazole. Protects -OH from reagents.
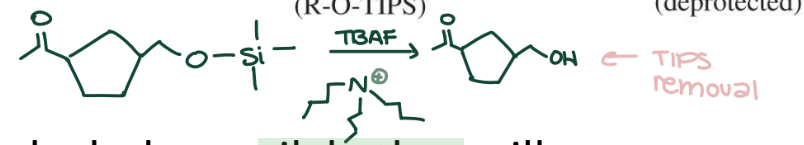
Silyl Ether Deprotection
R-O-TBDMS → R-OH (Alcohol) using TBAF (Bu₄N⁺F⁻) in THF. Fluoride has strong affinity for silicon.
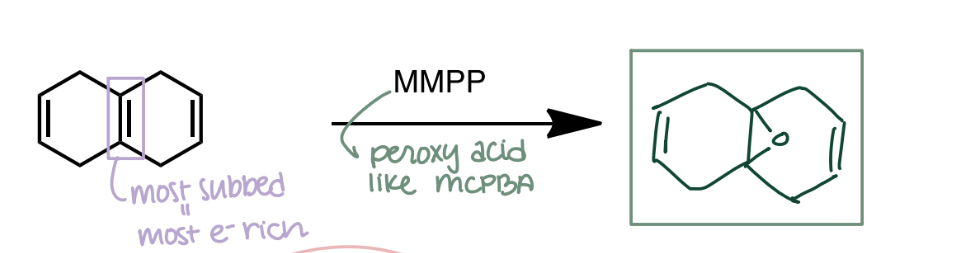
Epoxidation with Peroxyacid
Alkene → Epoxide using mCPBA. Stereospecific: syn addition; cis-alkene → cis-epoxide.
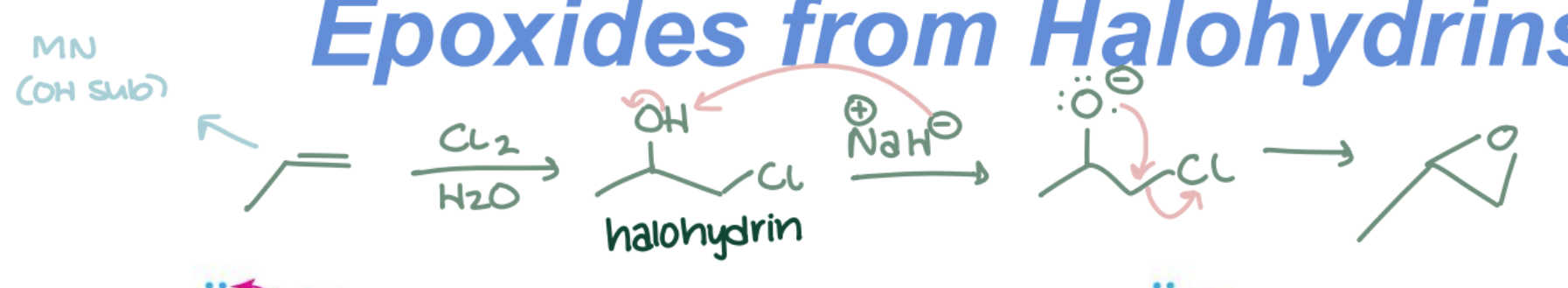
Epoxide from Halohydrin
Halohydrin → Epoxide using Base (NaOH). Intramolecular SN2 (backside attack); ring closure.
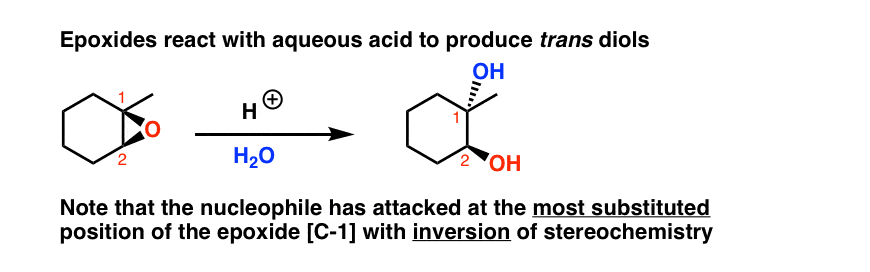
Acid-Catalyzed Epoxide Opening (H₂O)
Epoxide → anti-1,2-Diol using H₃O⁺/H₂O. Markovnikov regiochemistry; nucleophile attacks more substituted C.
Acid-Catalyzed Epoxide Opening (ROH)
Epoxide → β-Alkoxy alcohol (anti) using H⁺/ROH. Markovnikov; attack at more substituted carbon.
Base-Catalyzed Epoxide Opening
Epoxide → anti-1,2-Diol or β-Alkoxy alcohol using NaOH/H₂O or RO⁻. Anti-Markovnikov; nucleophile attacks less substituted C.
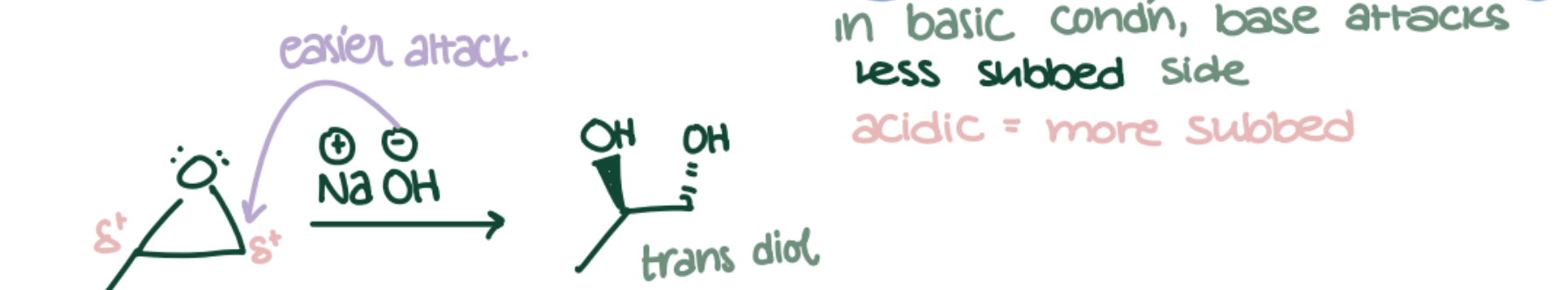
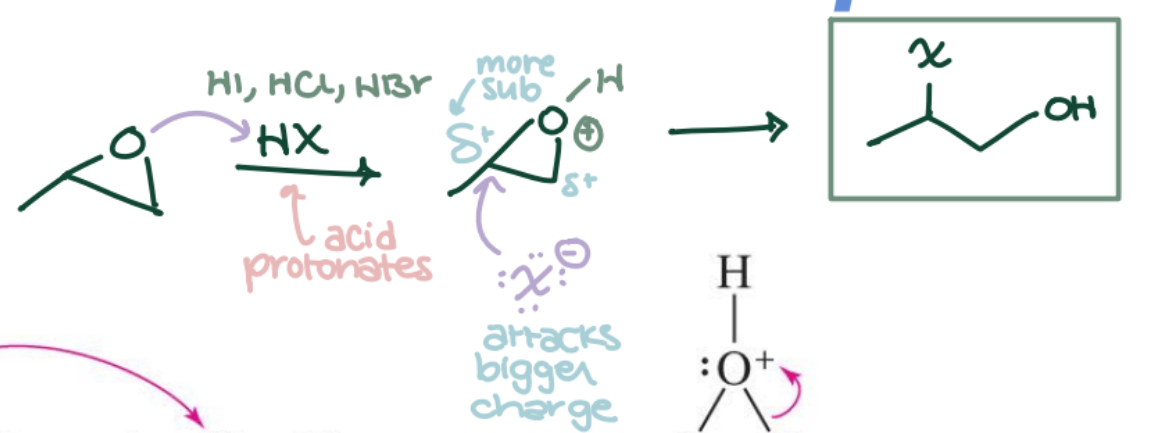
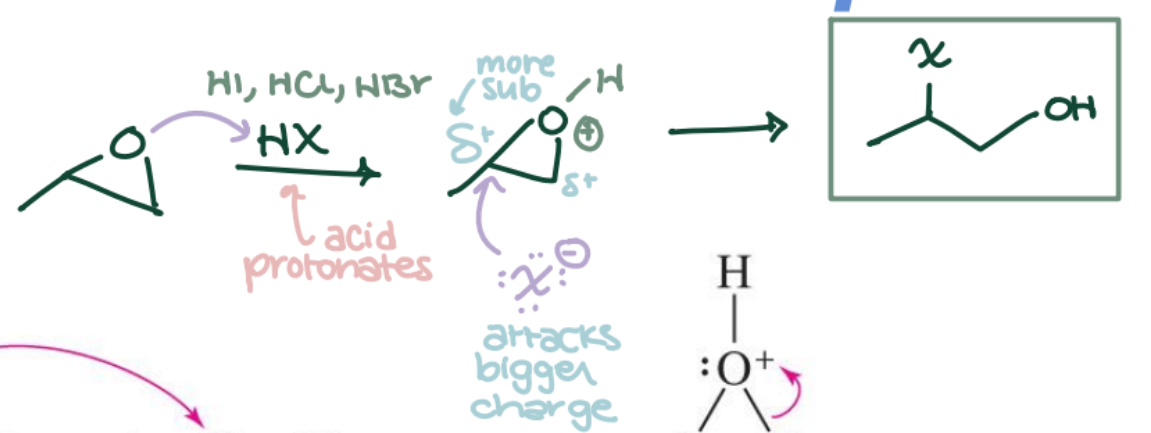
Epoxide Opening with HX
Epoxide → Halohydrin (anti, trans) using HBr or HCl. Markovnikov regiochemistry; anti stereochemistry.
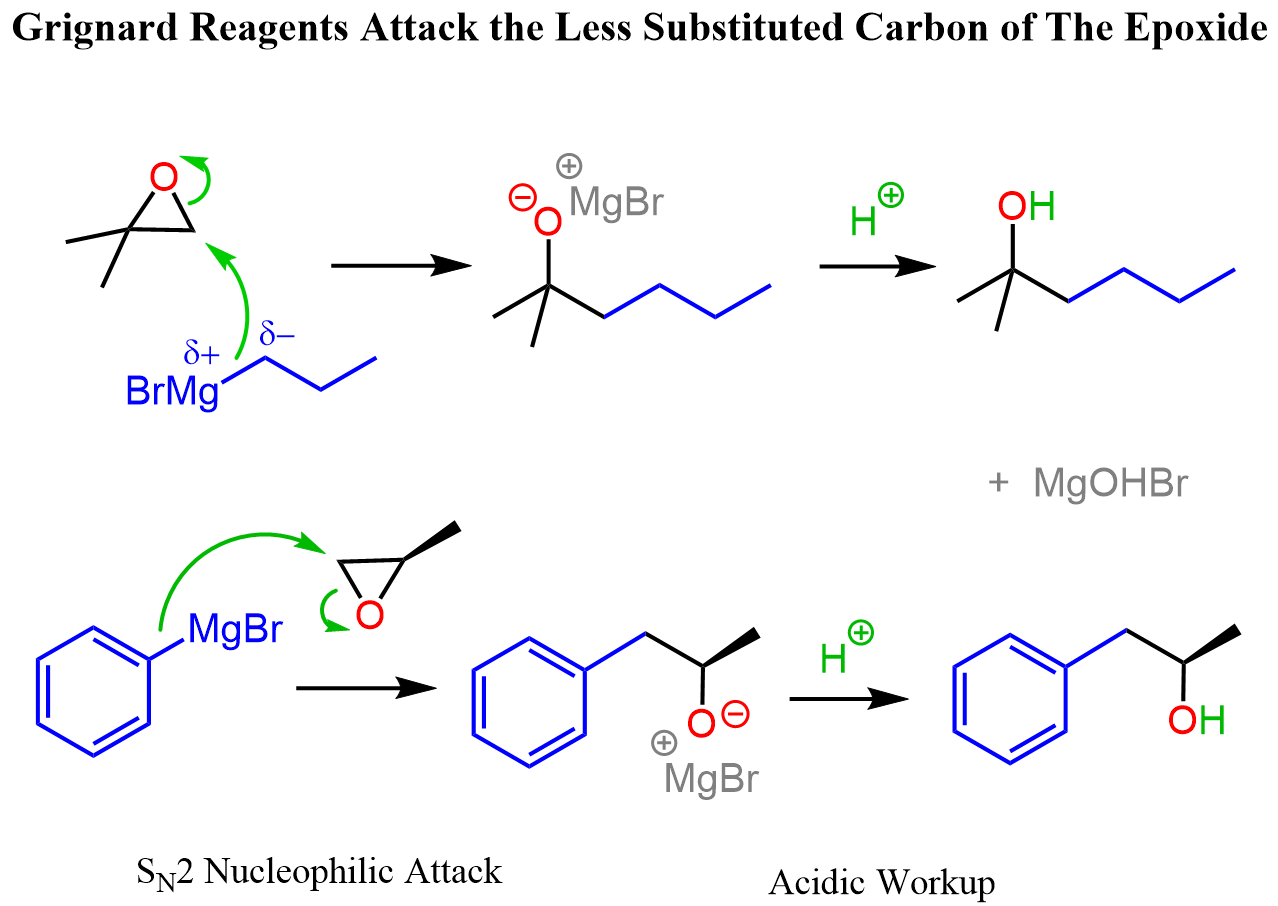
Epoxide Opening with Grignard
Epoxide → Alcohol (2C longer chain) using RMgBr or RLi, then H₃O⁺. SN2 at less substituted C; extends carbon chain.
Epoxide Regiochemistry Summary
Acid → more substituted C attacked; Base → less substituted C attacked. Both give anti stereochemistry.
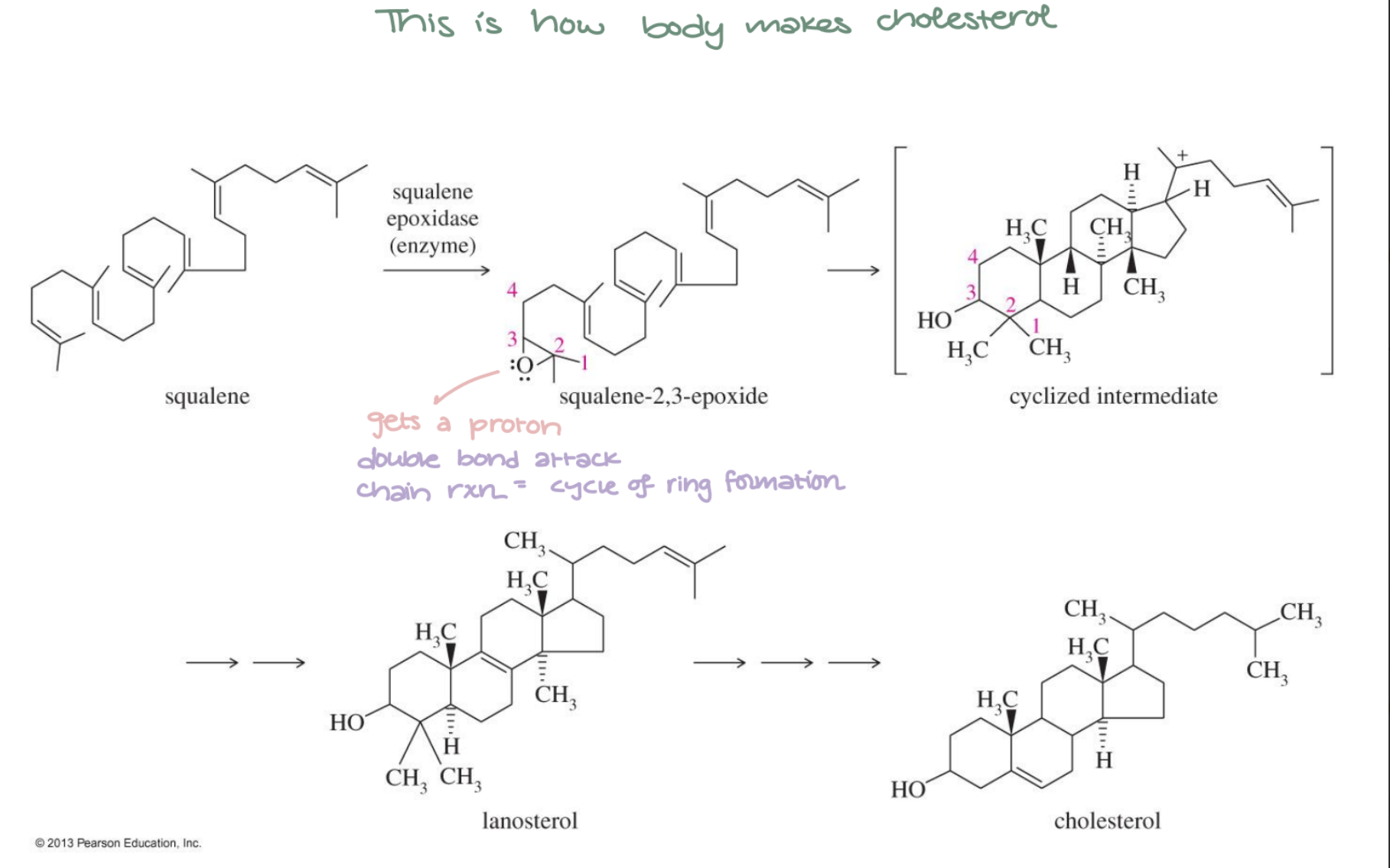
Squalene Epoxide → Lanosterol/Cholesterol
Squalene 2,3-epoxide → Lanosterol → Cholesterol via enzyme-catalyzed cyclization. Biological polyene cyclization; multiple ring formations in one step.