week 9
1/73
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
74 Terms
what is acid pollution
atmopsheric emmision, transport, chemical tansformation and deposition of acidying species
eg SO2, Nox, H2SO4
what does acid pollution manifest as
acid rain
acid deposition
acid mist
acid aerosols
acid precipitation
precipitation with ph <5.6
acid mist
fog/mist with acidity <2.5
acid deposition
total wet + dry deposition of acidifying species
why is normal rain slightly acidic
cabonic acid fomation
co2 dissolves into ain
critical load
deposition below which harmful effects do not occur
Critical level
atmospheric concentration above which direct adverse effects occur.
principal natural phenomena
emmisions from volcanoes, biological processes, soil microbes ect
principal human sources
industrial and power generating plants
transportation
factory farming
specific S pollutants
SO2
SO3
H2S
mercaptans (Foul odour)
how does glacial ice record the industrial pollution
decrease in pH
start of industrial revolution of 6 to 4.5
why has so2 emmisions decreased since 1989 in uk
changed from coal to gas in power station
natural gas has lot less sulphur
where are sox and nox conc concentrated
over urban and industrial areas
why do tall smoke sacks cause bad effects
gases chucked high up into the atmopshere
above inversion layer
where there are high winds
transports long distances→ cross boundary
only significant gas phase equation of so2 to acid
HO⋅+SO2(+M)→HSO3⋅(+M)
hydroxyl radical → highly reactive, photochemical decomposition of ozone
aqueous phase conversion of sulphur dioxide to acid
So2 dissolves and dissociates
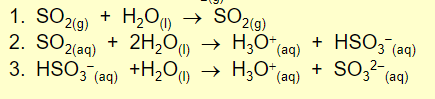
main equation for conversion of nitrogen oxides to acid
HO⋅+NO2(+M)→HNO3(+M)
gas phase
effects of so2 on plants
so2 can enter leaf stomata casuing
necrosis- cell death- black spots
chlorosis- bleaching of leaves- white and yellow
spring acid shock
snow and ice contain frozen acid
melts and transports frozen acid to lakes and river in high concentrations
spring is time of new plants and reproductive cycle
kills everything
damage to fish
vegetation impacts
SO₂ enters stomata → necrosis and chlorosis
Reduced photosynthesis
Increased susceptibility to frost, pests, disease
aquatic impacts
Spring acid shock
Mobilisation of Al³⁺ → fish toxicity
Loss of sensitive species (mayfly, trout, salmonids)
soil impacts
leaching of nutrients
plants can no longer grow
acid impact on built environment
Acid dissolution of carbonate stone (CaCO₃ → Ca²⁺ + CO₂)
Damage to limestone, marble, metals, paints
Examples: St Paul’s Cathedral, Acropolis
why is mercaptan added to natural gas
natural gas is odorless
add small amounts so people can smell leaks
what is the convention of long range trans boundary pollution convention
1979
Framework for transboundary pollution control
control emmisions though legally binding protocols
why do we want to enhance dispersion of emmisions
lowers pollutants conc to an acceptable threshold
methods for dispersion enhancement
Taller stacks- above inversion layer
Production curtailment- stop or reduce sulphur
Not a sustainable solution.
process change
modification of production process to reduce emissions
methods for process change
Fuel switching (coal → natural gas)
Efficiency improvements
Waste minimisation
Most economical option.
intervention
downstream pollution control
remove or destroy contaminants through chemical and mechanical processes
end of pipe intervention methods
Absorption (wet limestone scrubbing)
Adsorption
Low‑sulphur fuels
principal of absorption
relies on solubility of so2 in water
so2 dissolves and forms sulphurous acid (H2SO3)
forms sulfites and sulfates
prevents so2 from re-emitting into gas stream
most common so2 control technology
wet limestone scrubbing
absorption
so2 removed by aqueous limestone (CaCO3) slurry
limestone neutralises acid and forms calcium sulfite and sulfate
wet limestone scrubber chemistry
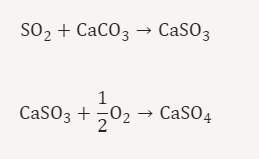
why is regenerative processes good
waste poducts can be comerically useful
eg gypsum
wall plaster
what is adsorption
Adsorption removes SO₂ by binding it onto the surface of a solid sorbent.
Typical sorbents include:
Activated carbon
Metal oxides
used for lower conc
limitations of adsorbent
Sorbents must be regenerated or replaced.
More expensive than wet scrubbing for large flue‑gas volumes.
low sulphur fuel control technology
pollution prevention by process change
most economical and preferred option
types of low sulphur fuels
Natural gas
Low‑sulphur coal
Low‑sulphur fuel oils
reduces SO₂ formation at source.
advantages of low sulphur fuels
Avoids the need for large end‑of‑pipe systems.
Reduces other pollutants (e.g., particulates, metals).
Aligns with modern environmental policy favouring prevention over intervention.
disadvantages of low sulphur fuels
Availability and cost of low‑sulphur fuels.
Not always feasible for legacy industrial infrastructure.
Does not address NOx or other pollutants.
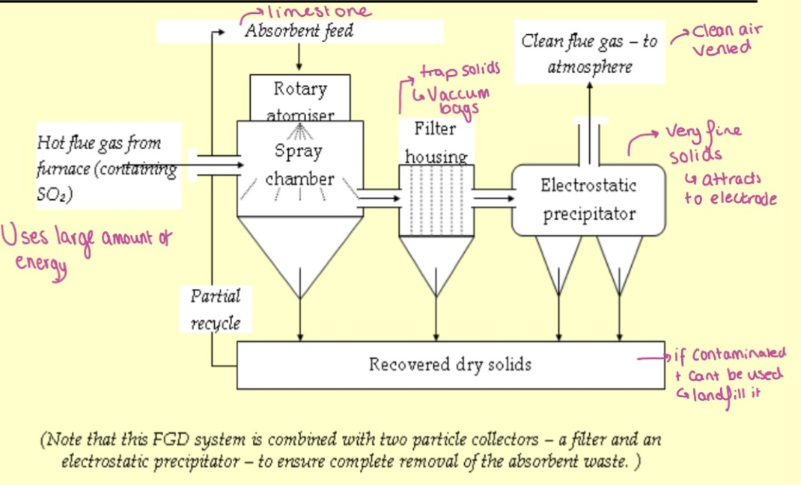
Flue Gas Desulfurisation (FGD)
end‑of‑pipe pollution control systems
remove SO₂ from flue gases using absorption, adsorption, or chemical reaction
commonly through wet limestone scrubbing, producing sulphite/sulphate by‑products.
schematic of spray drier absorbing system
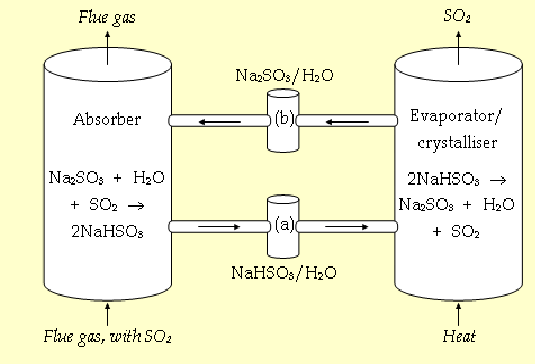
Wellman–Lord SO₂ Recovery Process
regenerative SO₂ removal system
Uses sodium sulfite (Na₂SO₃) solution to absorb SO₂
Produces sodium bisulfite (NaHSO₃)
Regenerated by heating to release pure SO₂, which can be used to make sulphuric acid- can have useful use
major industrial sources of pm
iron and steel mills
cement and asphalt
sulphuic and phosphoic acid
detergent
glass
instant coffee
collection efficiency equation
conc of exhaust gas/ conc of inlet gas
gravitational settling chambers
slows gas flow to allow large particles to settle under gravity
made of metals that can withstand high temps and corrosion
solid and liquid
used for dirty gases- smelters and metallurgical process
adv of gravitational settlers
simple
low cost
low maintenance
effective for big particles
disadv of gravitational settlers
ineffective for fine pm
schematic of gravitational settling chamber
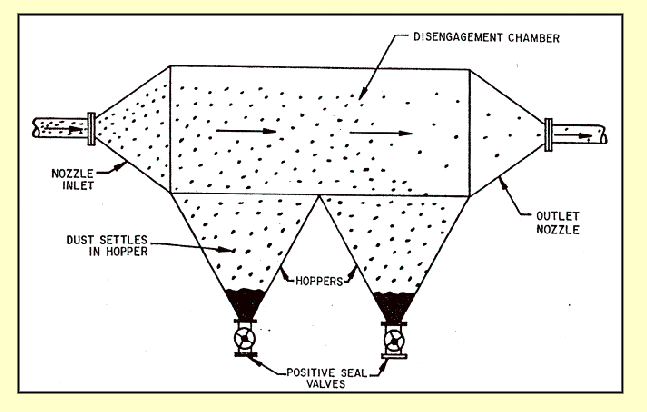
cyclone (centrifugal) collectors
gas spirals downwards
spiral diameter reduces
until gas tuns and spirals upward
particles driven by centrifugal force to wall
cannot turn fast enough
form lager agglomerates collect in gas hopper
multi-clones used for finer PM
advantages of cyclone collectors
more powerful than gravity
conc cyclones - 50% efficiency for 20um
high efficiency cyclones- 80% for >10um
disadvantages of cyclone collectors
poor for sticky particles (tar)
schematic of cyclone collector
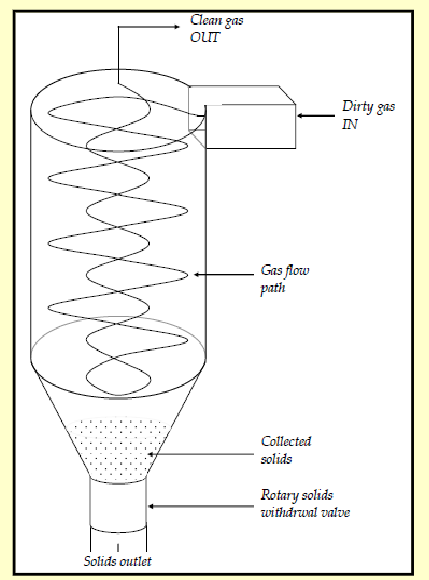
electrostatic precipitators
corona discharge (electrons) charges particles
charged particles migrate to electrodes forming cake
removed by rapping (tapping) or washing with liquid
conditioning agents (NH3) can improve performance
advantages of ESPs
high efficiency- 99% for >2um
well for medium-resistivity solid
widely used
disadvantages for ESPs
works poorly for low or high resistivity
schematic diagram of ESPs
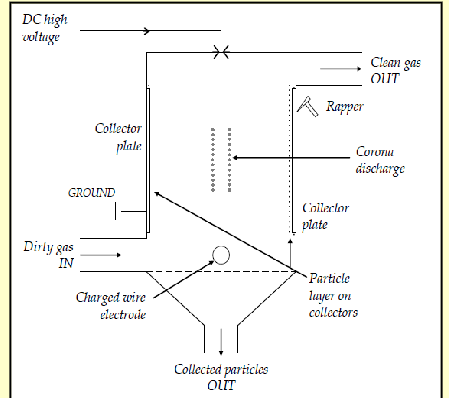
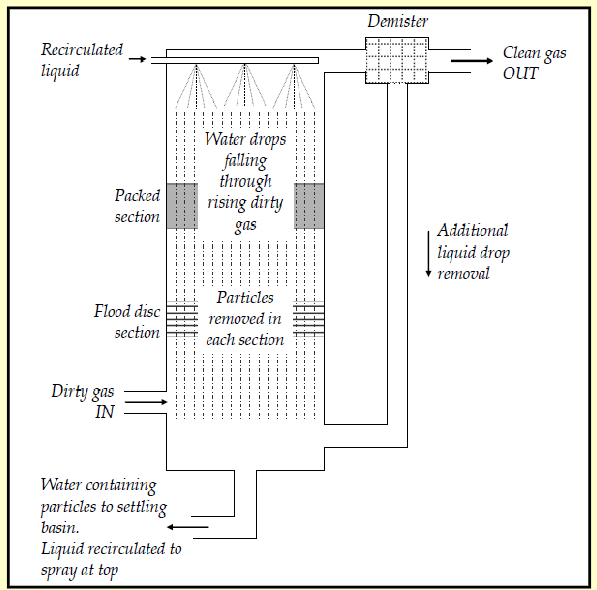
conventional scrubbers
falling water droplets collide with particles from upward flowing gases
liquid drains through disc- shower
liquid containing particles is collected
liquid recirculated; demister removes droplets
schematic of conventional scrubber
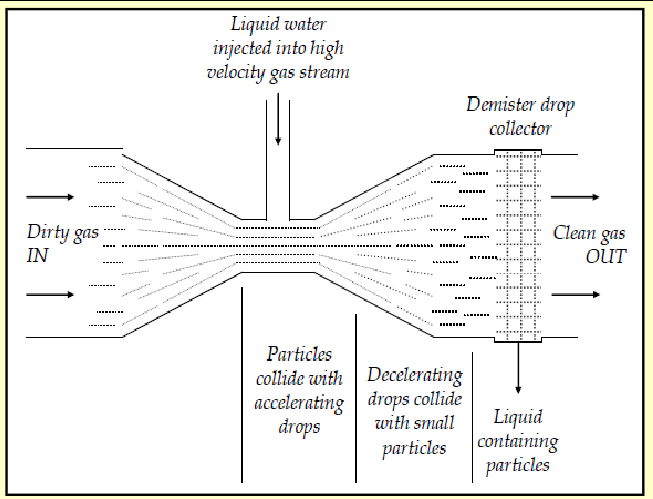
venturi scrubbers
particle laden gas accelerates in throat of device- narrows
liquid injected into throat
particles collide with liquid and form drops
drops containing particles removed in demister
used in steelmaking and smelting
efficiency up to 100% for 1um
schematic of venturi scrubber
surface filters (baghouse)
fabric bags trap particles on surface
clean air goes out
bags get shaken to clean
advantages of surface baghouse filters
high efficiency for small pm
inexpensive
disadvantages of surface baghouse filters
not suitable for wet or high temperature streams
depth filters
thick mats of tangled fibres capture particles throughout entire filter body
particles have many chances to impact upon individual fibres
liquid and solid particles
very fine droplets - sulphuric acid mist
emerging technologies
Fly ash leaching- FLUWA
acidic leaching of fly ash to remove contaminants
FLUREC
recovers valuable metals- Zn, Cu, Cd, Pb
waste incineration and wastewater
life cycle assessment
ISO-aligned method assessing environmental impacts across full life cycles
compare device impact, energy use, filter lifetimes and disposal routes
Material Flow Analysis (MFA)
quantifies inflows, outflows and stocks of materials
identifies emission hotspot and informs PM control strategies
combined with LCA for integrated assessment
dry flue gas desulphurisation
inject dry alkaline sorbents (hydrated lime) into flue gas
produces dry solid by-products
requires less water than wet scrubber
usually low efficiency
dry sorbent injection
simple and low cost
powdered sorbents injected into flue gas stream
moderate so2 removal
regenerative FGD system
uses regenerable sorbents→ activated carbon
sorbent regenerated
so2 recovered as concentrated stream and converted to sulphuric acid
high removal efficiency + minimal waste
high cost
post combustion techniques consideration
wet FGD produces wastewater→ requires treatment
dry systems avoid wastewater → produce more solid waste
regenerative systems minimise waste→ require more energy