I6: Bioinorganic Chemistry
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
What is a Cofactor ?
What do Metallobiomolecules do?
How can we determine the structure of proteins?
A cofactor is The metal in a protein
Metallobiomolecules comprise proteins, enzymes and non-proteins
we can determine the structure of proteins using “ Protein Crystallography “
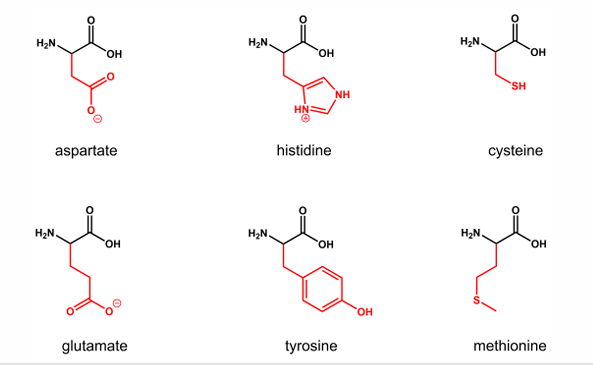
Name 6 Amino acid ligands
And draw 3 of your own choice
Amino acid ligands: glutamate, aspartate, histidine, tyrosine, cysteine, methionine
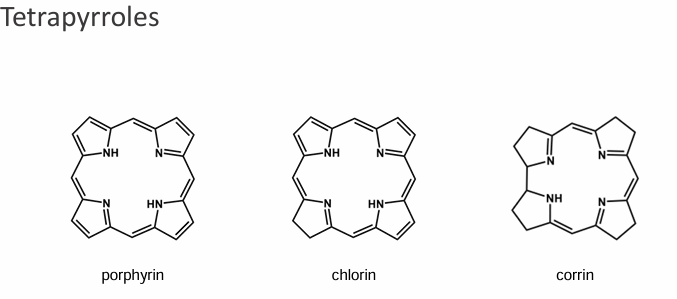
Draw 3 core structures of tetrapyroles, and where are they found
Tetrapyroles are found in plants and animals
What are the common OS of Fe?
What does Ferritin do?
What does Transferrin do?
What does Ceruloplasmin do?
What do Siderophores do?
What is the greatest known chelator?
Common oxidation states of iron are +II and +III
Ferritin stores iron in the body as iron oxide
Transferrin transports iron in/out of cells
Ceruloplasmin is the copper transport protein
Siderophores chelate iron in microorganisms
Enterobactin is the greatest known chelator
Which have a higher reduction potential σ-/π-donors or π-acceptors and why?
What is a heme?
Give 2 common cofactors
σ-/π-donors favour high oxidation states and lower the reduction potential
π-acceptor ligands require low oxidation states (i.e. electron rich metals) and produce high reduction potentials
Heme = iron + porphyrin
Cofactors ( M in a protein), common once are Fe and Cu
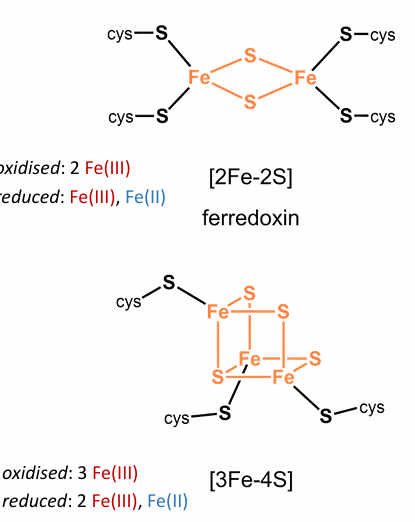
Give Structure and function of iron-sulfur clusters
Fe-S, is Fe2+ and Fe3+ ions Tetrahedrally coordinated by sulfide (S2–) and thiolate (cys-S–)
They Mediate photosynthesis, cell respiration, nitrogen fixation, hydrogen metabolism, nitrate and sulfate oxidation/reduction
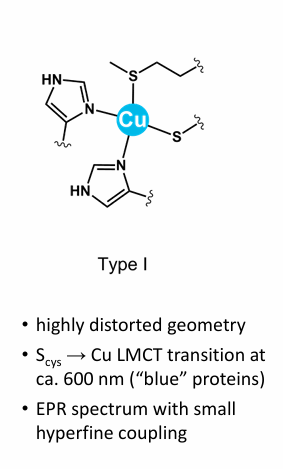
Draw Type 1, copper what is it used for and what colour is it?
Type 1 Copper/ Plastocyanin, “Blue Copper” is an Electron transfer proteins (Provides/removes electrons from neighbouring enzyme cofactors)
Draw Type 2 and Type 3 Copper
What colour is Type2?
Type 2 (Azurin) Colourless
Type 3 CuA
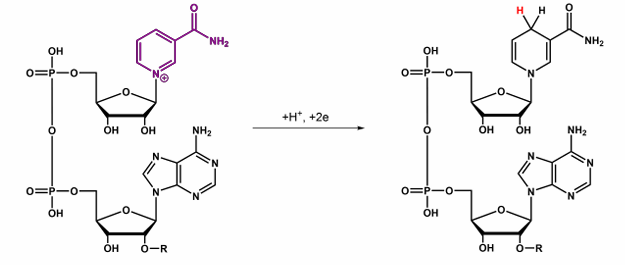
Give the abbbreviation and name for this, what does it do?
NAD(P)+ → NAD(P)H , named Nicotinamide
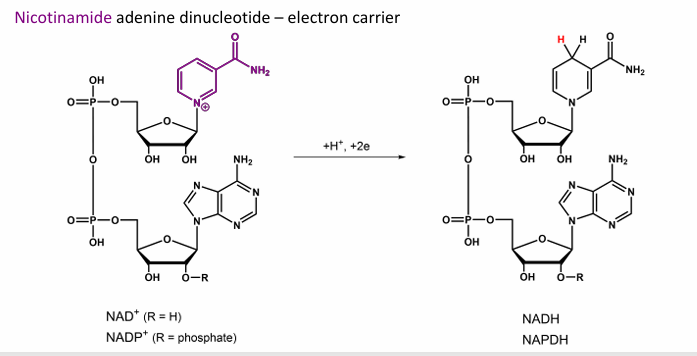
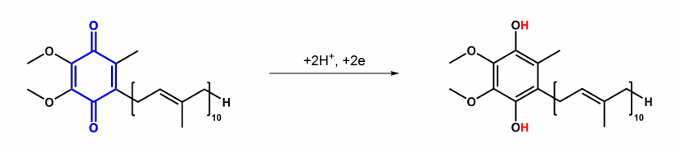
Give the name for this, what does it do?
Ubiquinone → Ubiquinol , Quinone
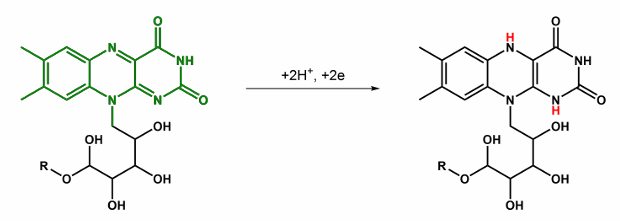
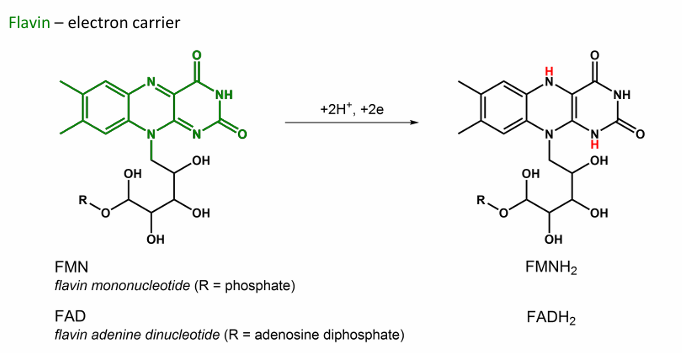
Give the abbbreviation and name for this, what does it do?
FAD/FMN → FADH2/FMNH2 , Flavin
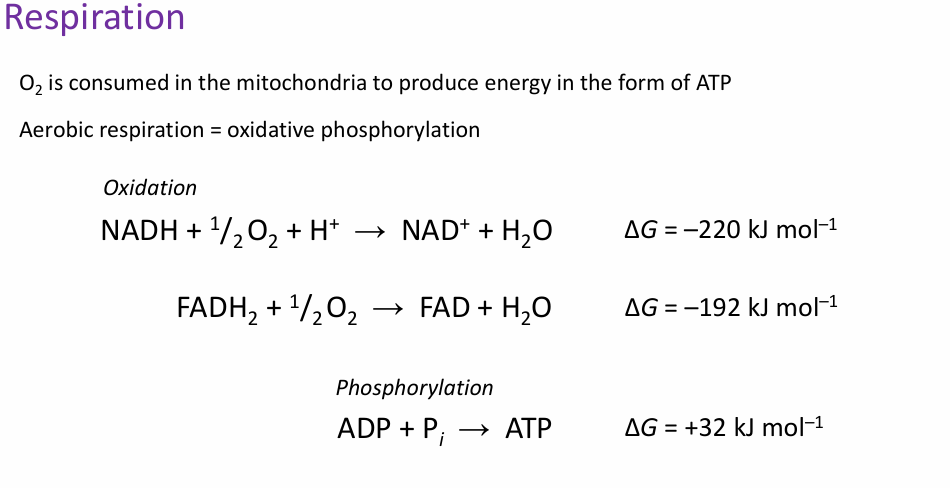
Give the overall formula for oxidative phosphorylation
Draw Hemoglobin reaction with O2
What does Hemoglobin do
Where is O2 saved
Which binds more strongly to Hemoglobin O2 or CO
Hemoglobin transfers O2 in general, It also transfers O2 to Myglobin for storage
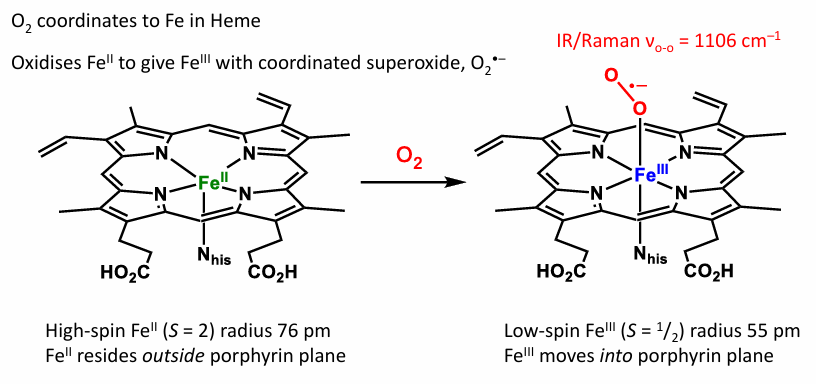

(i) Deoxyhemoglobin
Oxidation state: Fe²⁺
Spin state: High spin (HS, d⁶)
(ii) Oxyhemoglobin
Oxidation state: Fe³⁺ (with O₂⁻ superoxide)
Spin state: Low spin (LS, d⁵)
(iii) Carboxyhemoglobin
Oxidation state: Fe²⁺
Spin state: Low spin (LS, d
(c) Highest Mössbauer isomer shift
Highest isomer shifts is related to higher e- density ( high e- number) meaning lower O.S
and HS complexes , so Deoxyhemoglobin has the highest isomer shift
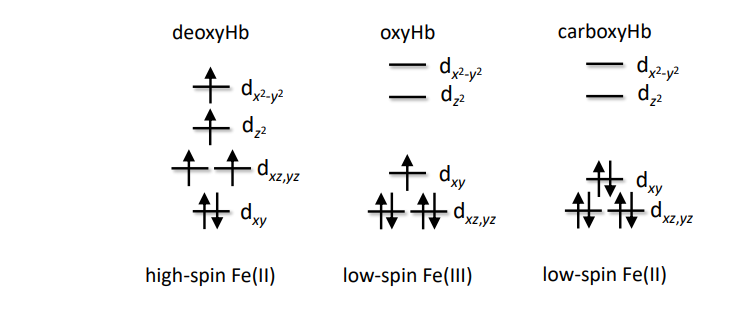
What is Mössbauer spectroscopy for and what does it determine?
Mössbauer spectroscopy is only for “Iron”
It deretermines: Oxidation State using Isomer Shifts
Coordination Number, and provides infos on Magnetic Properties
How to find the isomer shift (δ) and quadrupole splitting (ΔEQ) from the table?
How the isomer shift (δ) trends with the iron oxidation and spin state?
δ = (v1+v2)/2
ΔEQ = ∣ v2−v1 ∣
Big δ → Means High e- density so high O.S. Fe²⁺ or high spin
Small δ → Lower O.S coz lower e- density Fe³⁺ or low spin
Big ΔEQ → distorted / high spin
Small ΔEQ → symmetric / low spin
What is a Cytochrome P450, and what does it do write a rxn for it
Cytochrome P450 inserts an oxygen atom into the C–H bond of a substrate
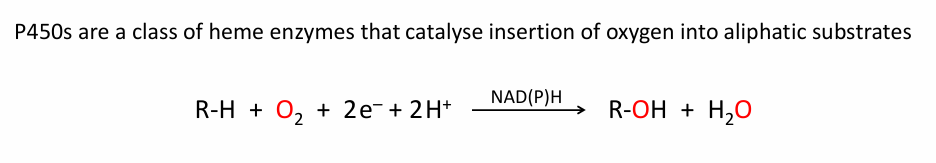
Draw the structure of Cytochrome P450
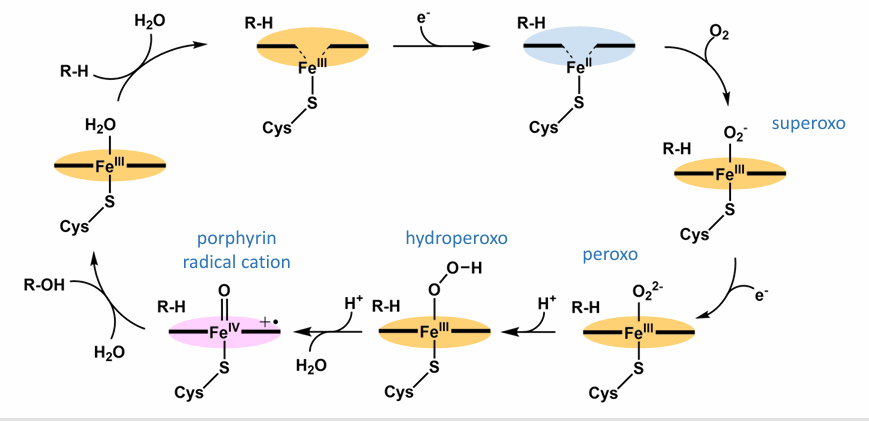
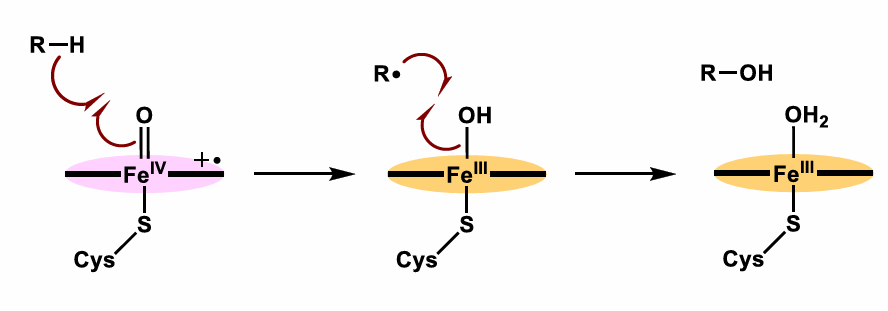
Draw the Catalytically active oxo-iron(IV) species cycle
Also what is produced when O2 is used as an oxidant?
Superoxide (O2^–.) and Peroxide (H2O2) are produced when O2 is used as an oxidant
Draw the rebound mechanism
What is the equation of disproportion of Superoxide?
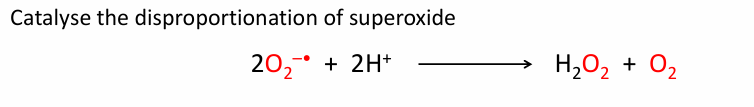
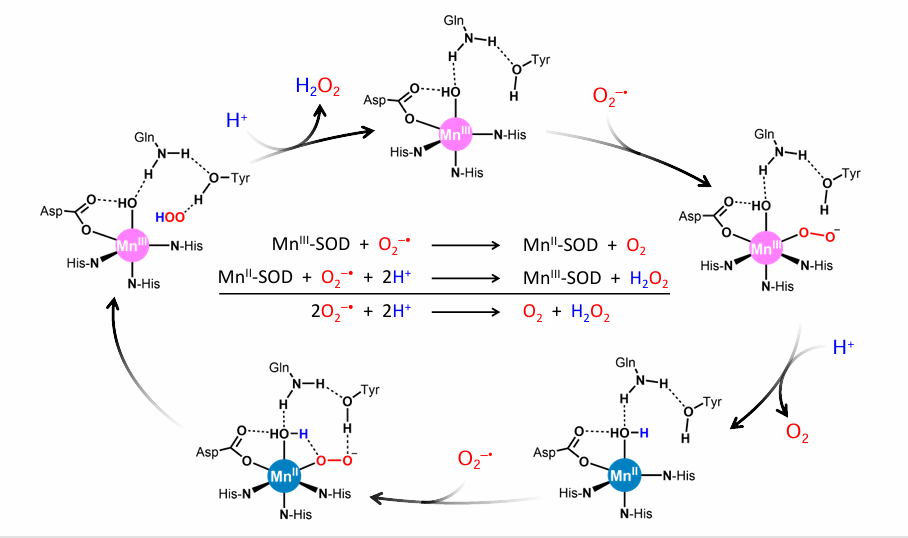
Draw a Superoxide Dismutase cycle
What is Peroxidase and a catalase, write down the rxn
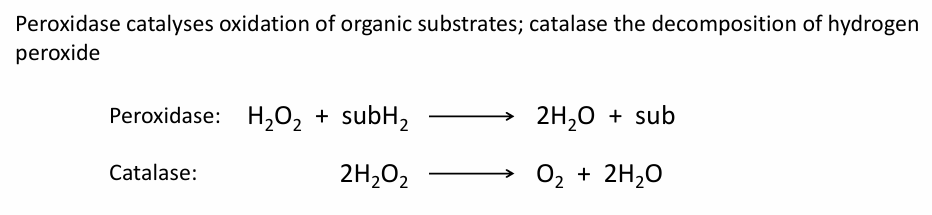
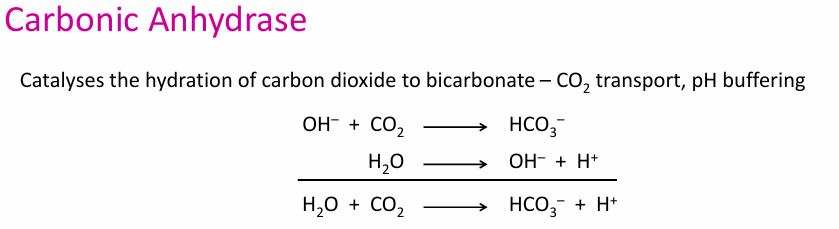
In Carbonic anhydrase: Write the rxn of CO2 transport and at what pH the turnover happens
Carbonic anhydrase: CO2 transport and pH buffer (CO2 ⇌ HCO3–)
Phenomenal turnover rate of 106 s–1 (pH 9 and 25 °C)
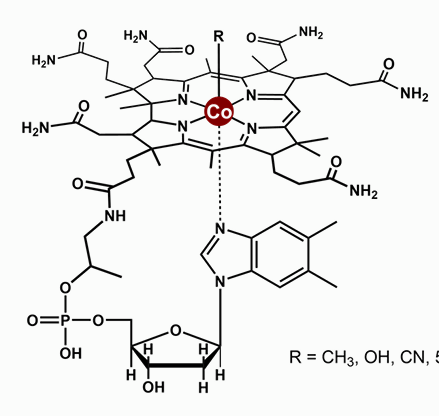
What does Cobalamin (B12) have?
And what is it?
B12 = Cobalt + Corrin
Cobalamin is a coenzyme – group transfer reagent (e.g. CH3+)
(No need to memorize the structure just recognise it)
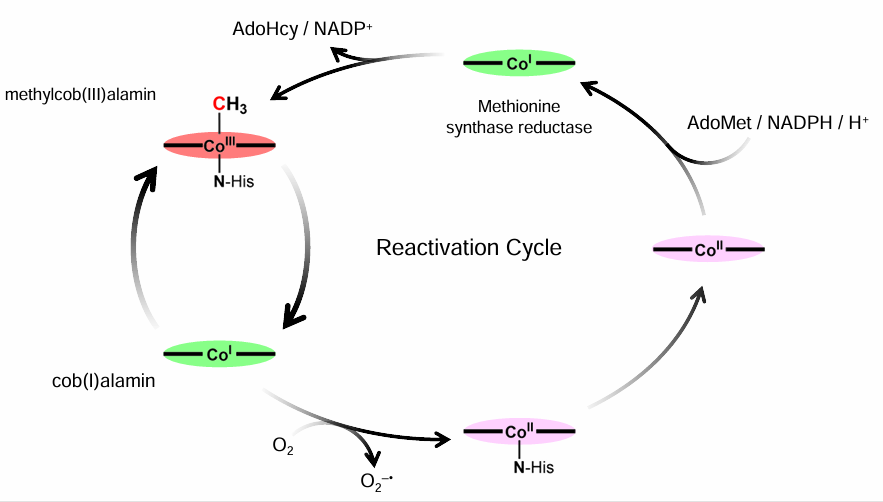
Draw the Cycles between Co(III), Co(II), Co(I)
and what is Co(I)?
Co(I) d8 is a Supernuecleophile
Give the 2 equations of photosynthetic light reactions
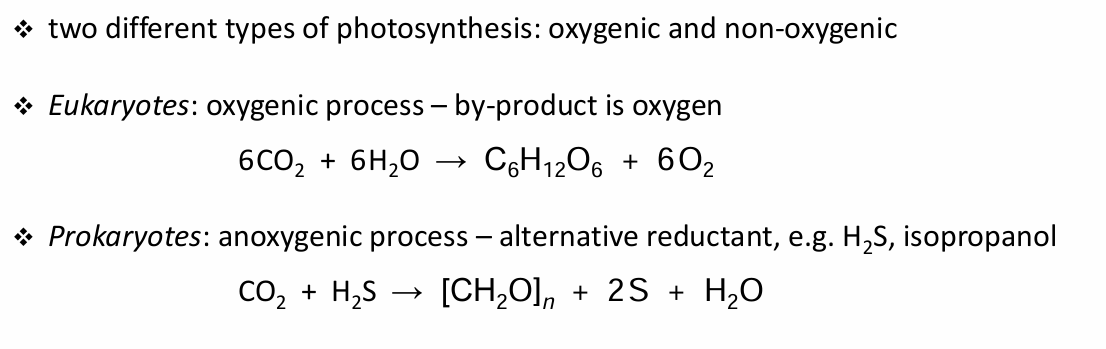
Give the steps to light rxn
What is Chlorophyll and what is it used in?
LIght rxn steps:
1.Light → 2.PSII → 3.ETC → 4.PSI → 5.ATP made
Chlorophyll acts like a cofactor
Chlorophyll = magnesium + chlorin
PSI and PSII use chlorophyll for light harvesting and reaction centre
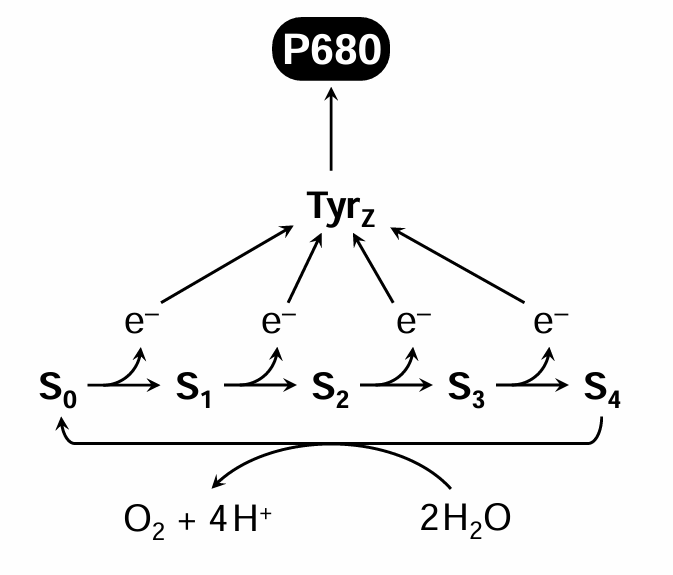
Draw a representation and mention the steps to a Kok cycle
Kok cycle: stepwise release of four electrons and four protons to make one O2 molecule; electrons transferred to PSII reaction centre
Use the Hückel’s rule to assign the prosthetic group of hemoglobin as aromatic, anti-aromatic, or non-aromatic.
Huckel’s rule should follow this rule
4n+2
n= number of C=C double bonds eg. for C=C, n=2,
for hemoglobin 4n+2=18, so n=4 so it is Aromatic