CHEM 1112- Electro/Nuclear/Organic Chemistry
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What is Oxidation Number?
The charge that an atom (or Ion0 has, or formally assigned.
What is the Oxidation Number of Oxygen?
Oxygen is -2 in most compounds. However Oxygen can have a -1 oxidation number when it is in a Peroxide (H2O2). Oxygen can also have +1 oxidation number when bonded with Fluorine.
What is the Oxidation Number of Hydrogen?
What is the Standard Potentials (E*) equation?
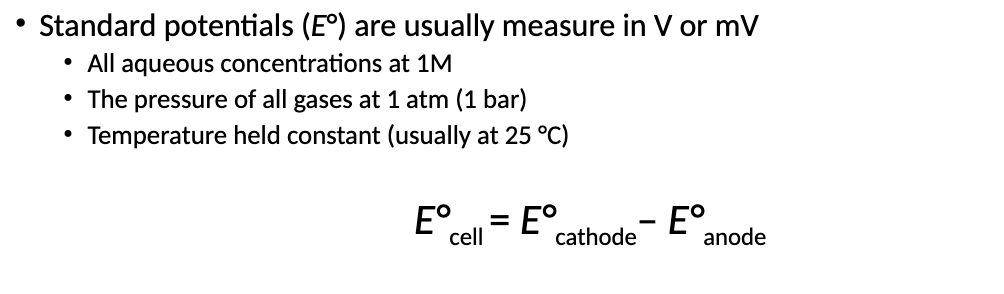
What is the function of a Salt Bridge in a Galvanic Cell?
Allows migration of ions without mixing of solutions. It also maintains electrical neutrality of the half cells.
What are the the Electrodes in the Galvanic Cell?
Anode: the electrode at which oxidation occurds
Cathode: the electrode where reduction occurs.

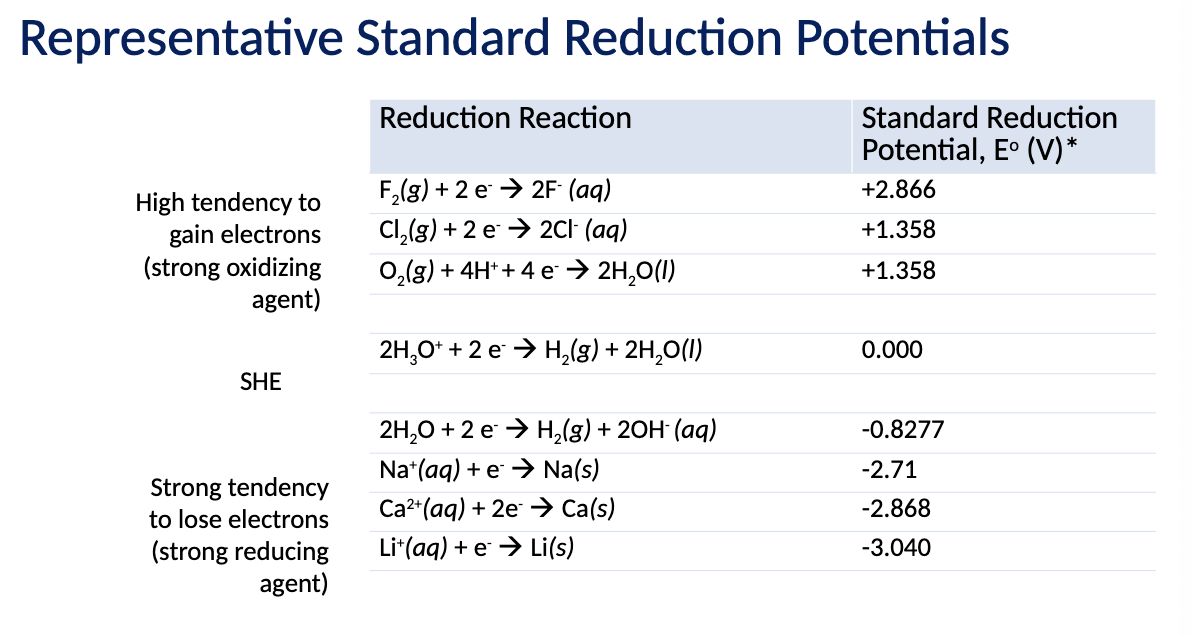
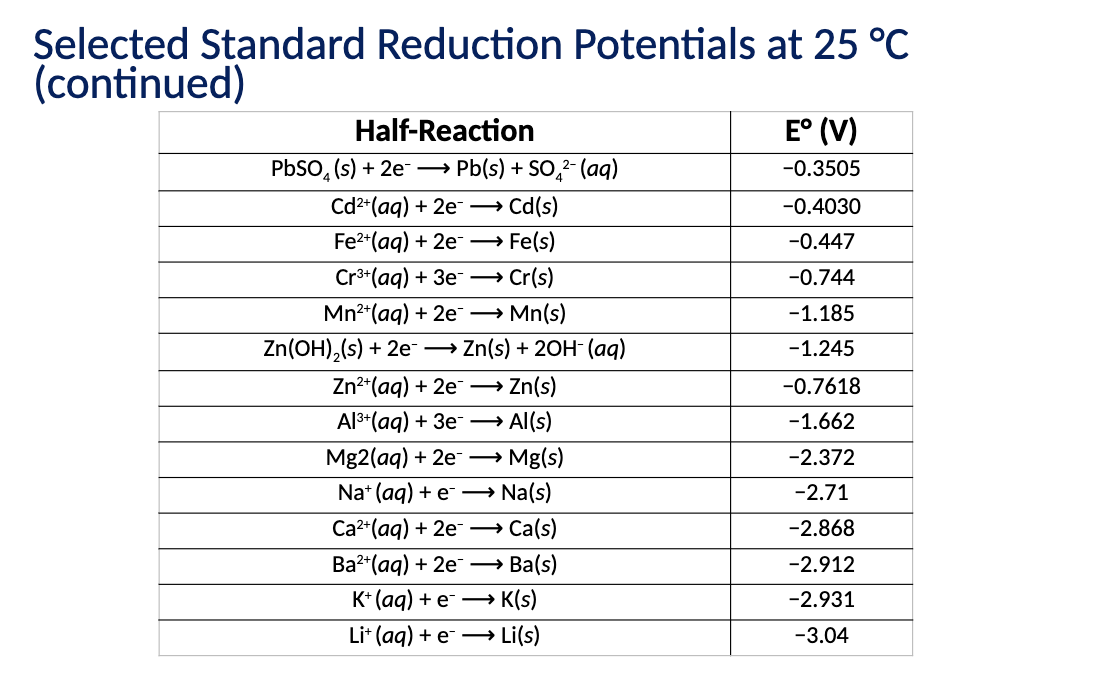
What is the relationship between E and Delta G (Equation). What does n represent?
n = number of electrons transferred, look at the constant of e. (LOWEST COMMON MULTIPLE)

In relation to Delta G and Ecell, How do you know if its a spontaneous reaction?
If Delta G < 0 and if Ecell > 0, it is a spontaneous reaction
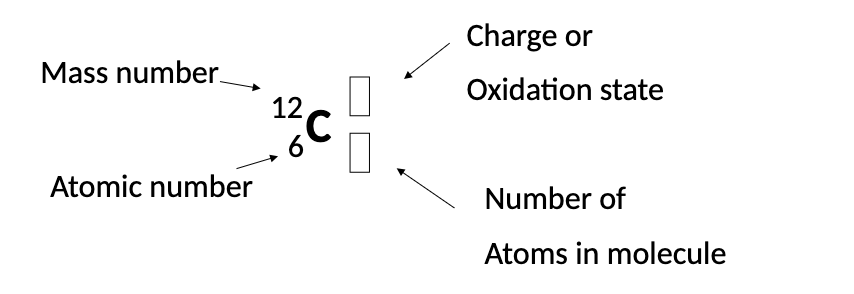
What is a Nuclide?
A particular nucleus
What is the most stable nuclide?
Ni where (atomic number =28 and Atomic mass = 62)
What is an Isotope?

WHat is Nuclear Decay?
the decomposition of unstable nuclides into more stable ones
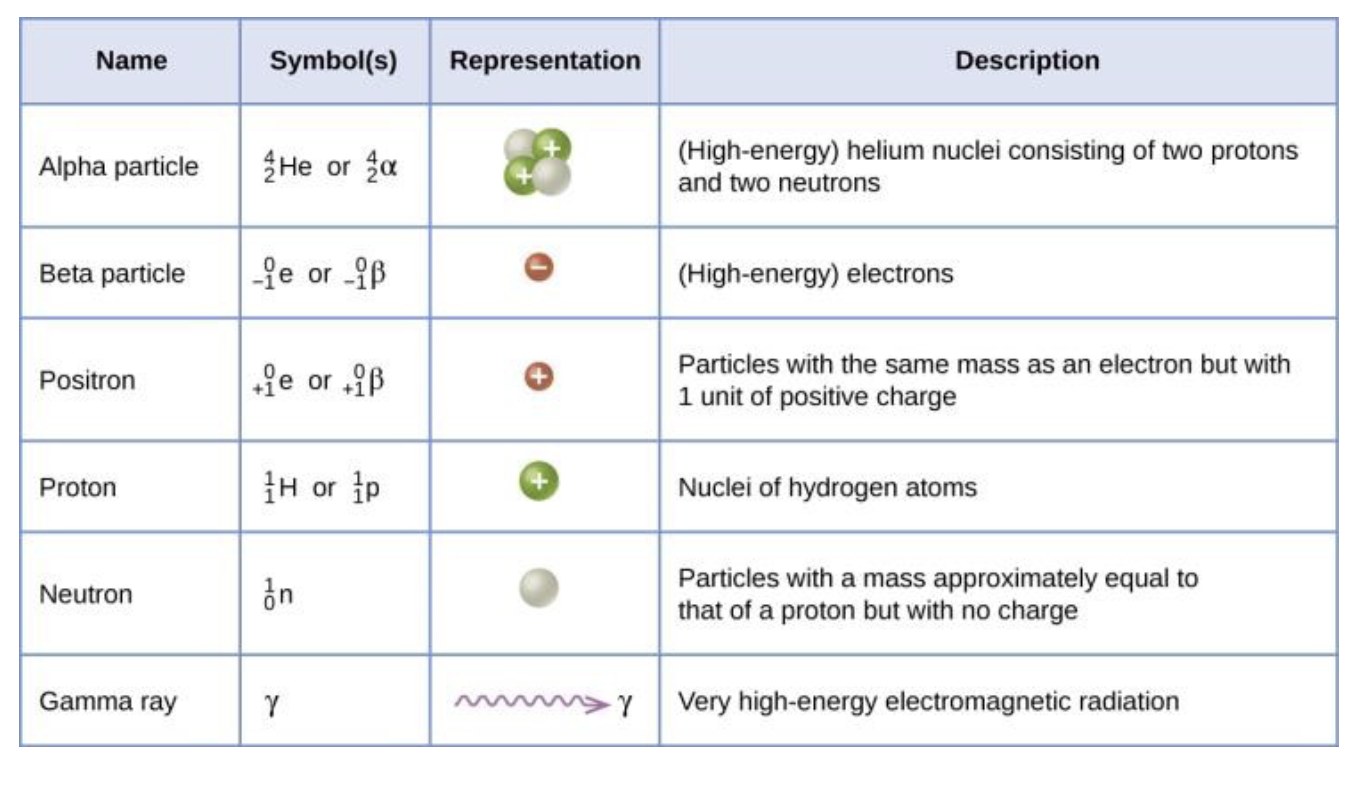
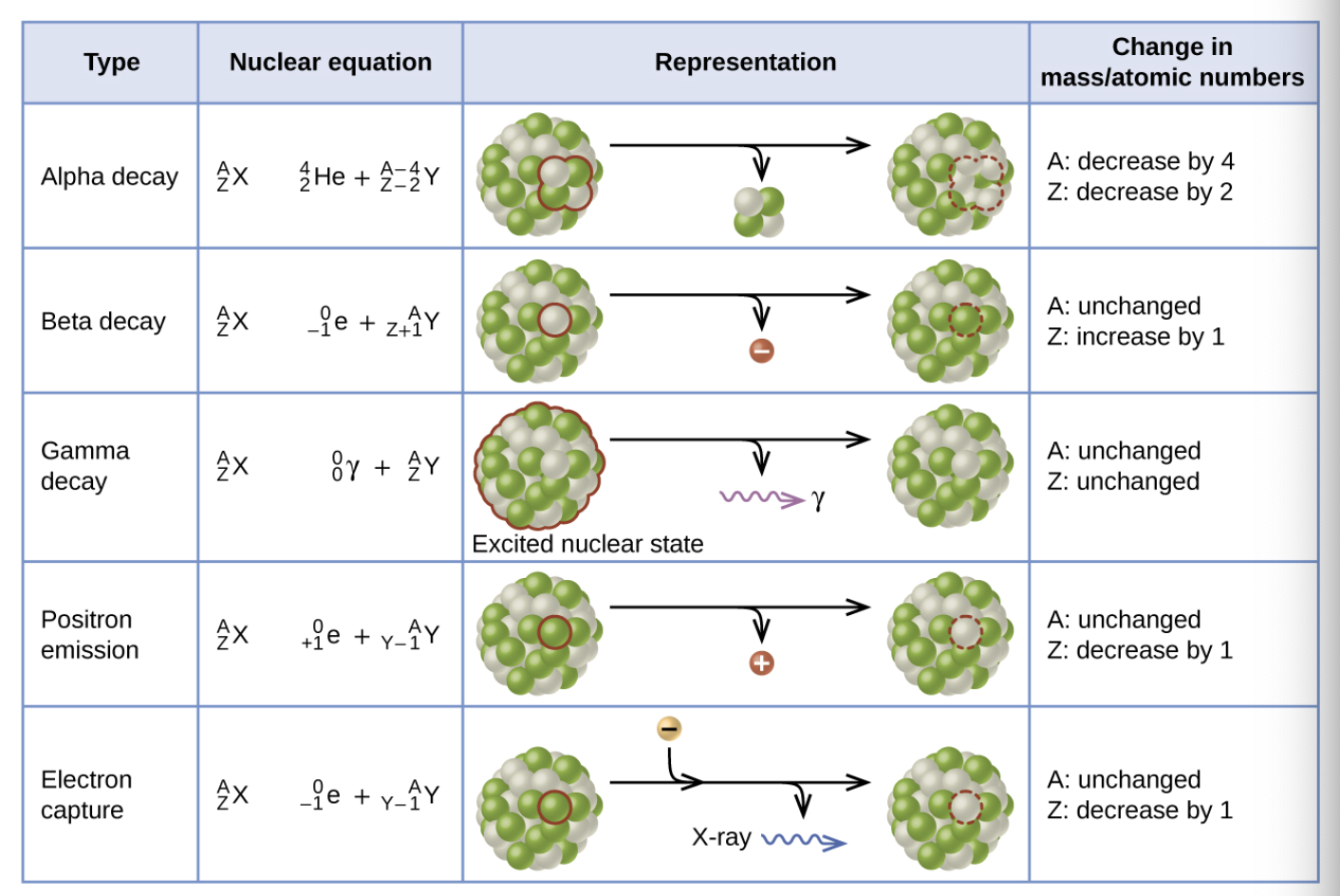
When the atomic number of a particle is greater than 83, what nuclear reaction occurs?
Alpha decay
If the particle has greater ration of Neutrons compared to Protons, what nuclear reaction would occur?
Beta emission: the conversion of a neutron to a proton
If a particle has a larger ratio of protons compared to Neutrons, what nuclear reaction would occur?
Beta + emission (position emission): the conversion of a proton to a neutron.
Fusion vs. Fission
Fusion: when LIGHT nuclides combine
Fission: when heavy nuclides fragment (decay)
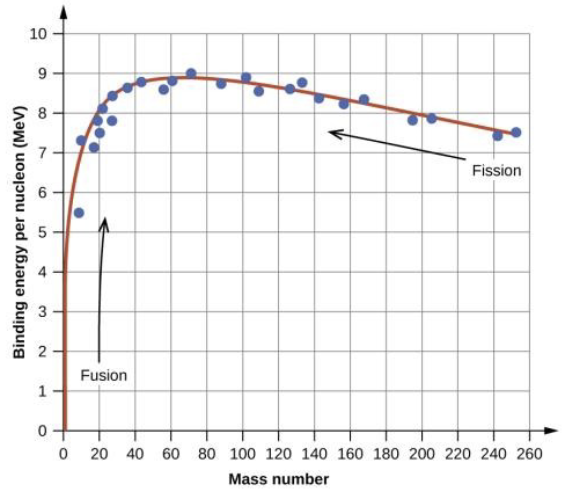
What are the Advantages and Disadvantages of Fission
Advantages: generates tremendous amount of electricity from a small amount of fuel
Disadvantages: potential for nuclear accidents (fission reaction can overheat)
What are the Advantages and Disadvantages of Fusion
Advantages: provides about 10 times more energy per gram of fuel than fission
Disadvantages: very high temperatures for fusion to occur (no material can withstand those temperatures for long)
When a particle is “bombarded” with alpha particles, what are the result?
Add the alpha particle to the particle but subtract 1 Neutron. A neutron is most likely emitted as an alpha particle is absorbed.
What are Hydrocarbons?
Organic compounds composed of Hydrogen and Carbon
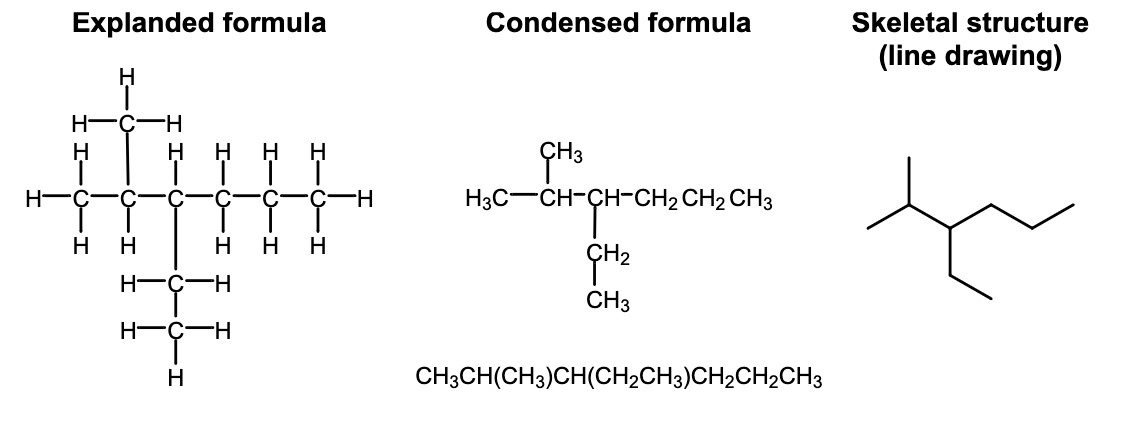
What are the Chemical Classes of Hydrocarbons?
Alkanes
Alkenes
Alkynes
Aromatic
What are Alkanes?
Molecules that contain only SINGLE covalent bonds between carbon atoms (no double/triple bonds)
What are the Properties of Alkanes?
Alkanes are not very soluble in water, and are highly combustible. The names are based on how many carbons it has
Name the first 10 Alkanes
Methane
Ethane
Propane
Butane
Pentane
Hexane
Heptane
Octane
Nonane
Decane
What is the General Formula for Alkanes?
CnH2n+2
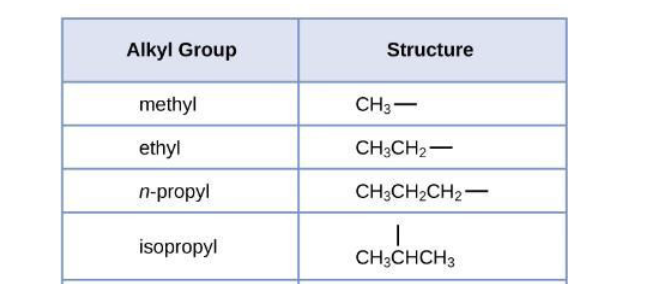
What are the common Substituents that will be tested on?
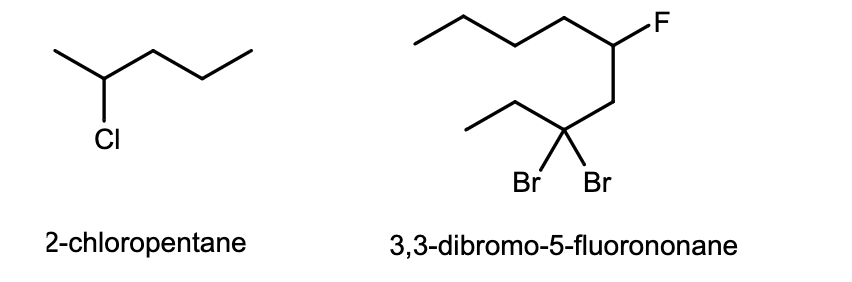
What are OTHER NON-Akyl groups of Substituents?
What are Alkenes?
Molecules that contain at least ONE DOUBLE Covalent bonds between carbon atoms