Pathogenesis of Periodontal Disease & Host Response
1/98
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
periodontal disease pathogenesis
periodontal disease results from a complex interplay btwn subgingival biofilm (bacteria) and the host immune-inflammatory response... which ultimately causes tissue destruction
cardinal signs of inflammation
redness
heat
swelling
pain
loss of function
what initiates inflammation in periodontal disease
tissue injury or bacterial infection
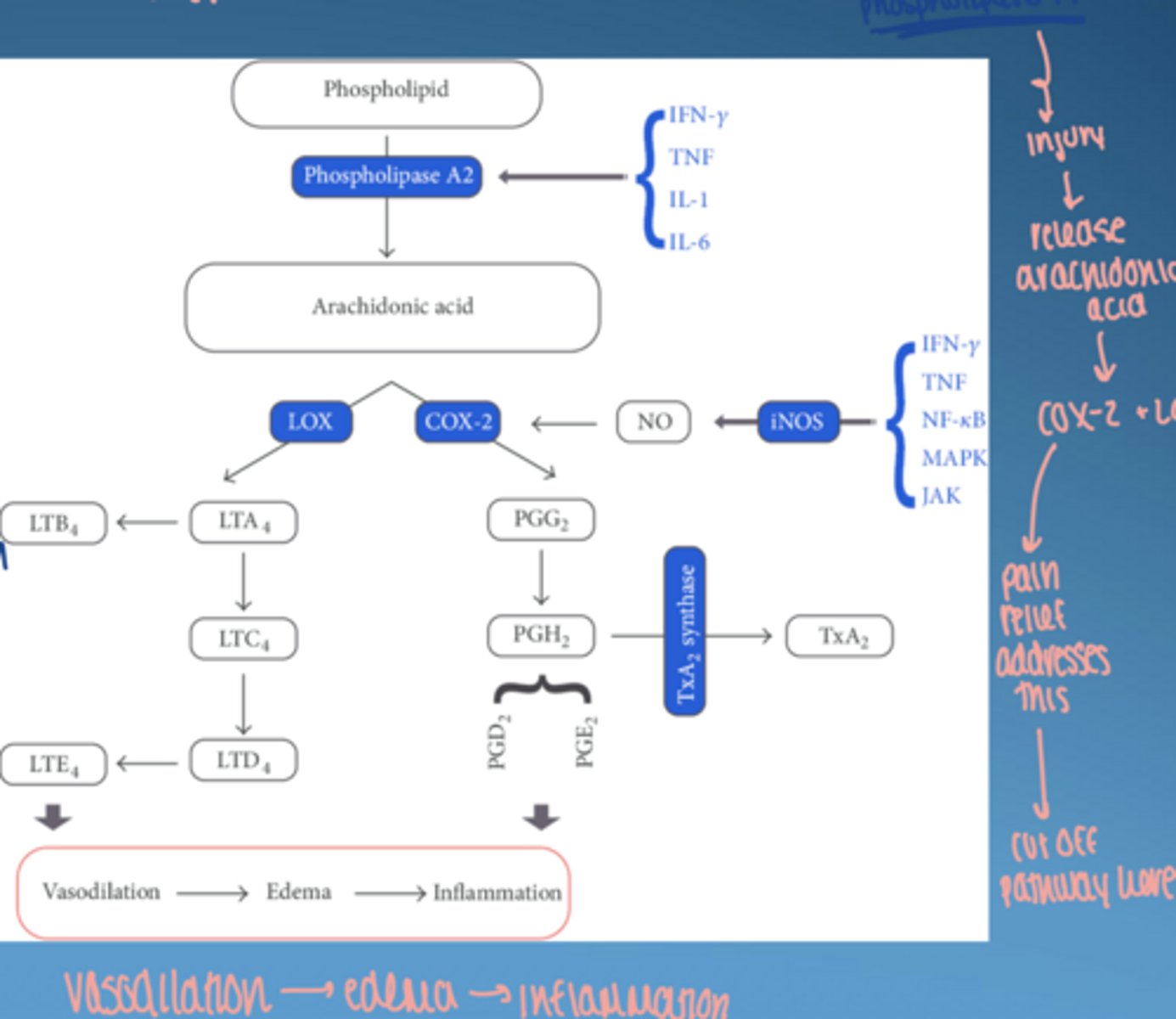
what is arachidonic acid
polyunsaturated fatty acid in phospholipids of cell membranes & released during cell injury
abundant in brain, muscles, and liver
what does arachidonic acid produce
prostaglandins
leukotrienes
thromboxanes
prostacyclin
role of COX-2
converts arachidonic acid into prostaglandins → inflammation + vasodilation
what is clinical significicance
pathway drives inflammation, edema, and bone resorption
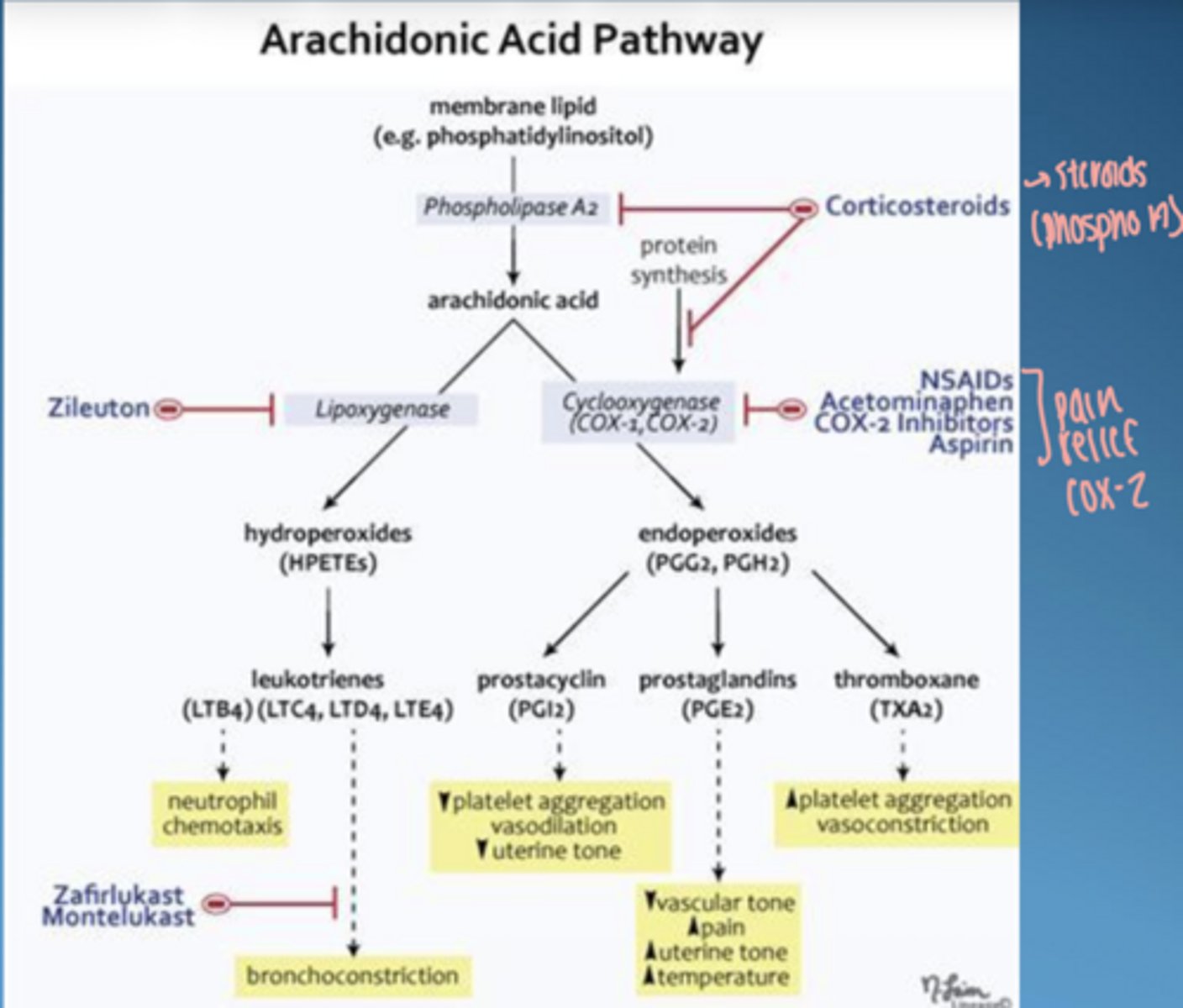
tissue injury or infection initiates activation of the ____ system and the ____ ____ pathway
kinin, arachidonic acid
what is the kinin system
blood protein system involved in (hormonal):
- inflammation
- pain
- blood pressure
- coagulation
key mediators of kinin system
bradykinin & kallidin
roles of bradykinin & kallidin
vasodilation
increase vascular permeability
promote pain
*vasodilation = slow down blood flow = when you want things to come out of vessels*
periodontal disease results from a complex interplay between _____ biofilm and the host ____-_____ response
subgingival, immune-inflammatory
*need control of inflam response to decrease colateral damage*
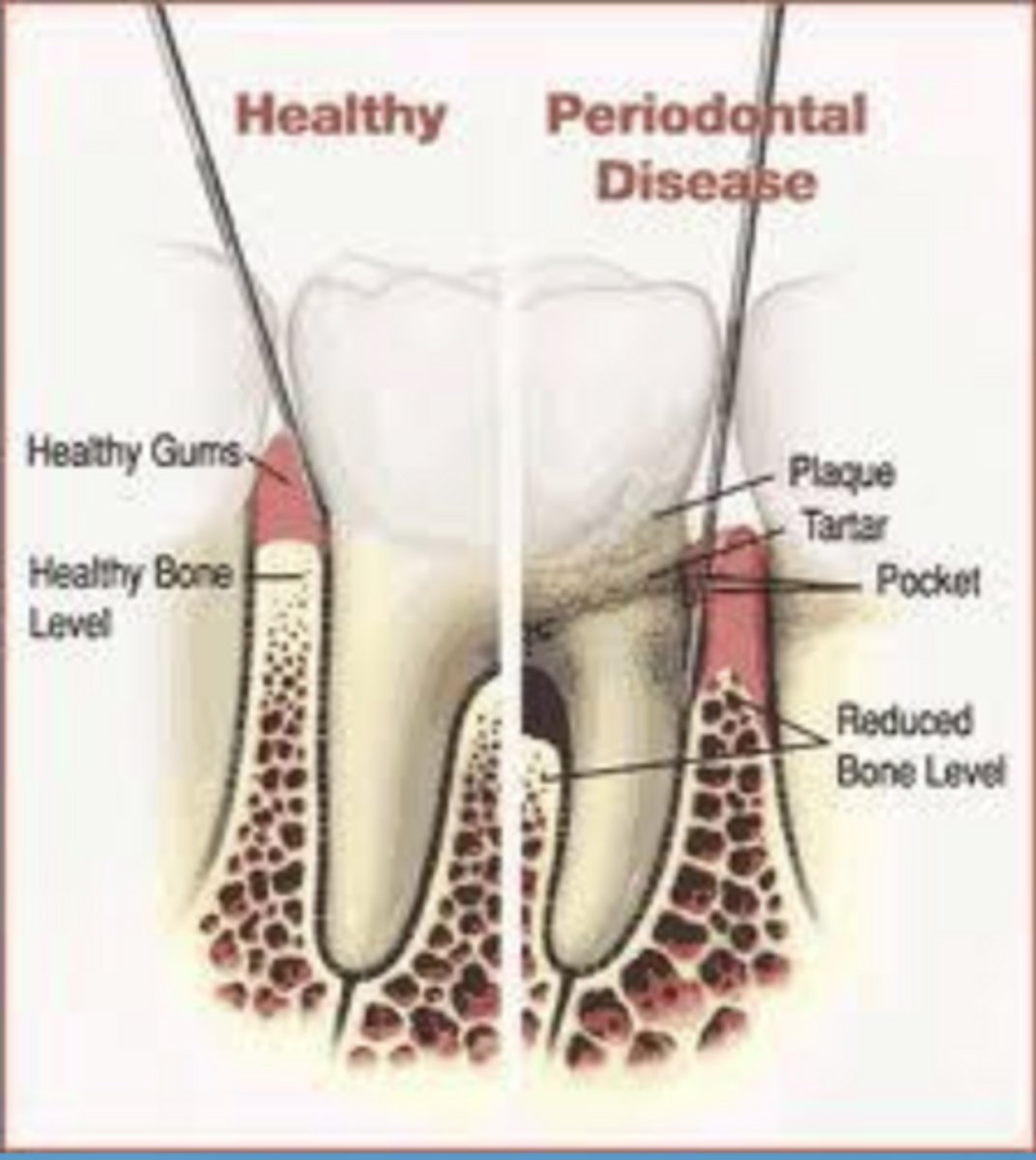
what is dysbiosis
imbalance in the microbial community → pathogenic bacteria dominate
how fast does biofilm form
within minutes after bruhsing (~30 seconds)
how many bacteria exist in oral biofilm
~500 species, but only a few cause disease
what determines disease severity
host response, not just bacteria
direct recognition of pathogenic bacteria is mediated by ____ ____ ___ ___ (MAMPs) or pattern recognition receptors on cell surfaces
microbial-associated molecular patterns
what is the red complex
group of highly pathogenic bacteria strongly associated with periodontitis
includes:
- P. gingivalis
- T. forsythia
- T. denticola
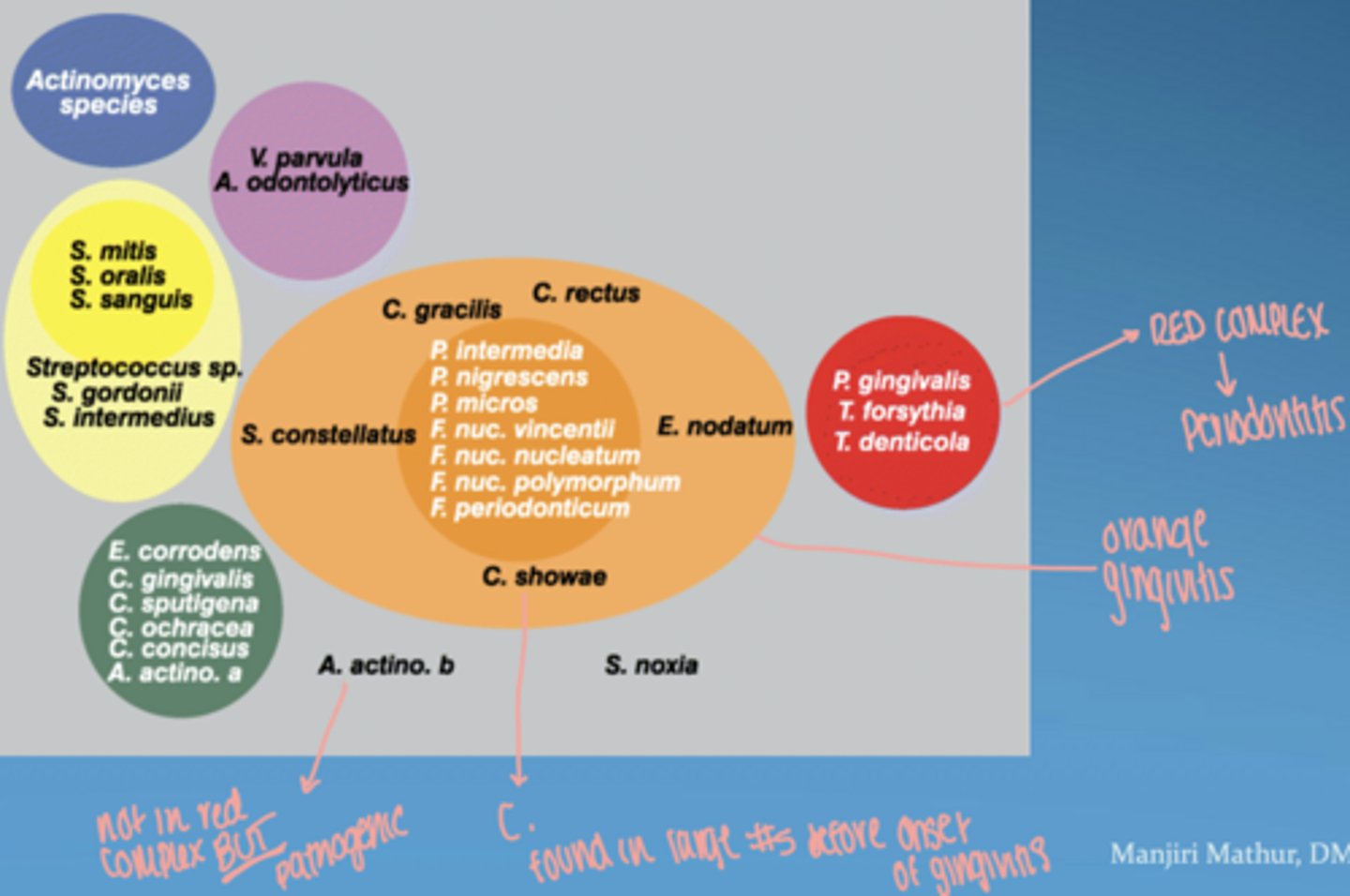
orange complex
bridge between gingivitis and more severe periodontitis
proliferate as plaque accumulates, leading to increased gum inflammation
Fusobacterium, Prevotella, and Campylobacter species
visual of pathogenesis of periodontal disease
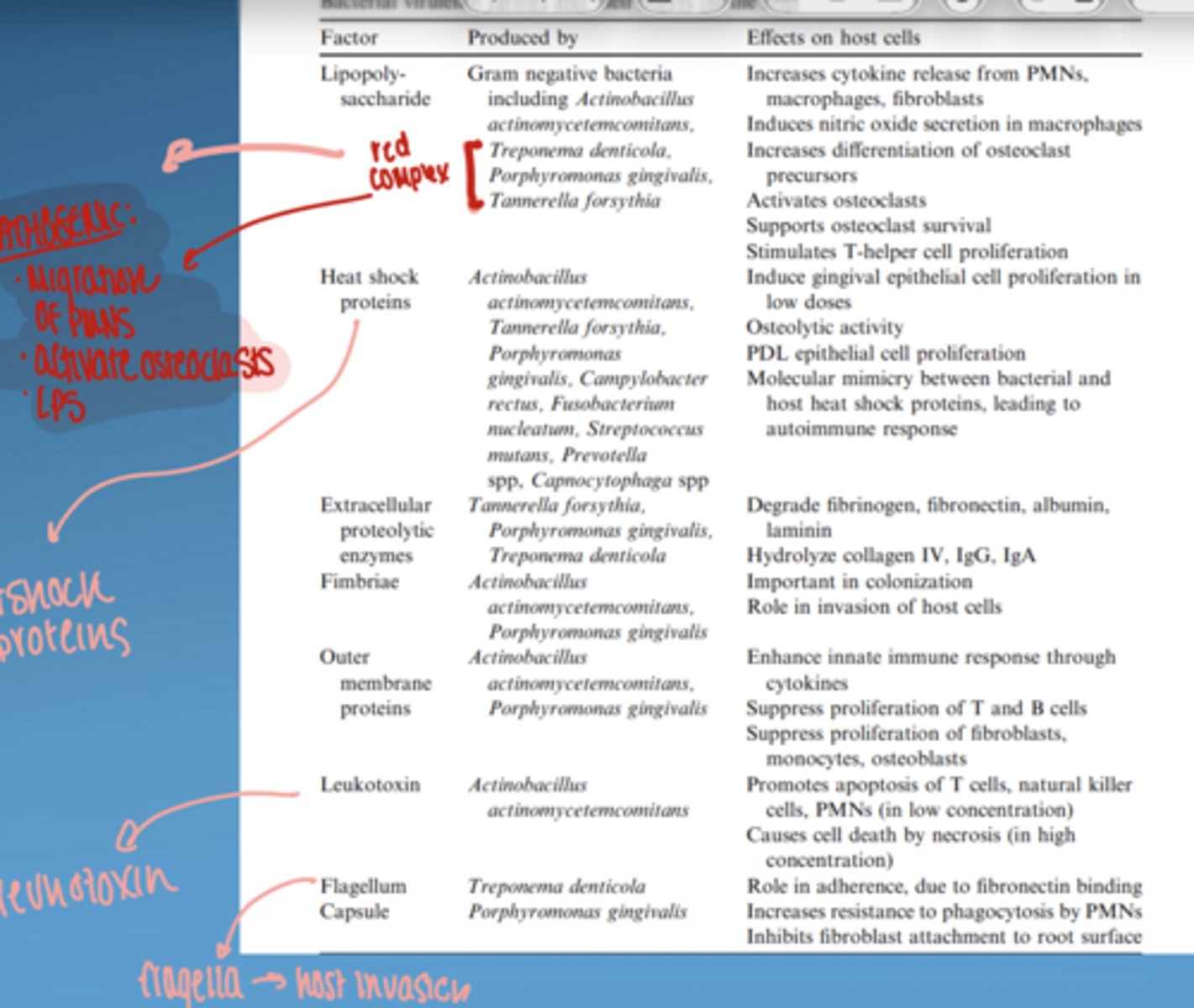
microbial virulence factors
- lipopolysaccharide (LPS)
- bacterial enzymes
- microbial invasion
host derived inflammatory mediators
- cytokines
- prostaglandins
- matrix metalloproteinases (MMPs)
host responses = mainly protective
mainly responsible for tissue damage in peridontal disease
what is LPS
lipopolysaccharide (endotoxin) from gram - bacteria
effect of LPS
- strong immune activation
- cytokine release
- tissue destruction ( indirect via host response)
how do bacteria cause damage
direct: toxins, enzymes
indirect: triggering host immune responses
bacterial virulence factors ex.
- LPS
- heat shock proteins
- extracellular proteolytic enzymes
- fimbriae
- outer membrane proteins
- leukotoxin
- flagellum
- capsule
what are the first-line defenses in gingiva (innate and structural)
- activated rapidly (within mins)
- junctional epithelium + sulcular epithelium: intact barrier
- gingival crevicular fluid (GCF)
- neutrophils + macrophages in sulcus (phagocytose bacteria)
- antibodies (in GCF)
increase in plaque + host response = inflammation
role of gingival crevicular fluid (GCF)
- flushes bacteria
- delivers immune cells
- increases in inflammation
* outflow of GCF from sulcus has dilution effect and flushing action*
inflammatory cells involved in innate immunity "professional"
- neutrophils/PMN/polymorphonuclear → phagocytosis
- macrophages → phagocytosis + signaling
- dendritic cells → antigen presentation & activate adaptive immun.
- NK cells → kill infected cells; host cells that are altered (ex. tumor)
"non-professional" inflammatory cells involved in innate immunity
fibroblasts
osteoblasts
epithelial cells
what do fibroblasts and osteoblasts do in inflammation
produce:
- variety of cytokines (IL-6)
- MMPs
- prostaglandins (E2)
- RNAKL
*fibroblasts = constantly making & breaking down collagen*
epithelial cells
junctional or sulcular epithelium
initial point of contact with microbial-associated molecular pattern (MAMPs)
what characterizes healthy gingiva histologically
CT:
-densely packed collagen fiber bundles
-few inflammatory cells (neutrophils)
-balanced host-bacteria interaction
- gingival crevicular fluid in small amounts
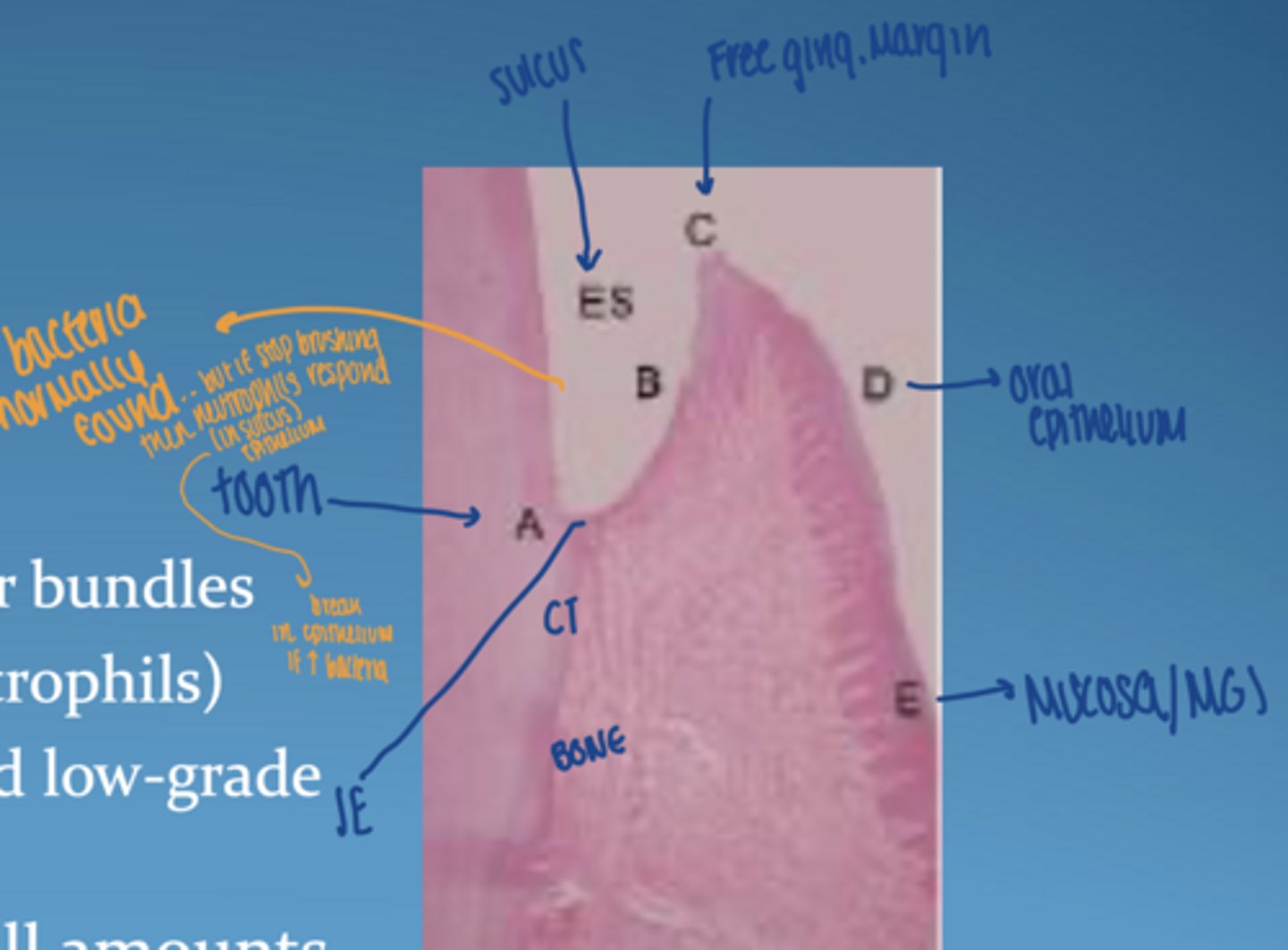
what is unique about junctional epithelium
- non-keratinized
- "leaky" = allows immune cells to pass; neutrophils & macrophages from CT to sulcus
get ingress of bacterial products and antigens
- weak attachment via hemidesmosomes
where does gingival crevicular fluid originate
from postcapillary venules
GCF
- flushing action in gingival crevice
- brings blood components of the host defense into sulcus
- fluid flow increases in inflammation (edema/swelling)
- neutrophils are important component
saliva
-prevents attachment of bacteria to teeth and oral mucosa
- inhibit virulence factors, bacterial cell growth
- provide IgA antibodeis to peridontal pathogens
what happens to GCF during inflammation
increases → contributes to edema
what recognizes bacteria
pattern recognition receptors (PRRs)
especially toll like receptors (TLRs)
detect MAMPs (microbial associated molecular patterns)
patterns recognition receptors
on macrophages & dendritic cells
recognize MAMPs including:
-LPS
-bacterial lipoproteins/ lipoteichoic acids
- flagellin
- CpG DNA of bacteria and viruses
- ds RNA
- ss viral RNA
* to signal an immune response*
toll like receptors
pathogen related receptors
regulate apoptosis, inflammation, and immune responses leading to production of cytokines, chemokines, & anti-microbial peptides
expressed by epithelial cells, dendritic cells, lymphocytes, osteoclasts, and osteoblasts
neutrophil function
- feature of healthy gingiva
- forms barrier between subgingival plaque and gingival tissues
- phagocytes
-secrete enzymes = degradation of junctional epithelium BM
- migrate from gingival plexus to CT in response to chemotactic gradient (pathway to bacteria)
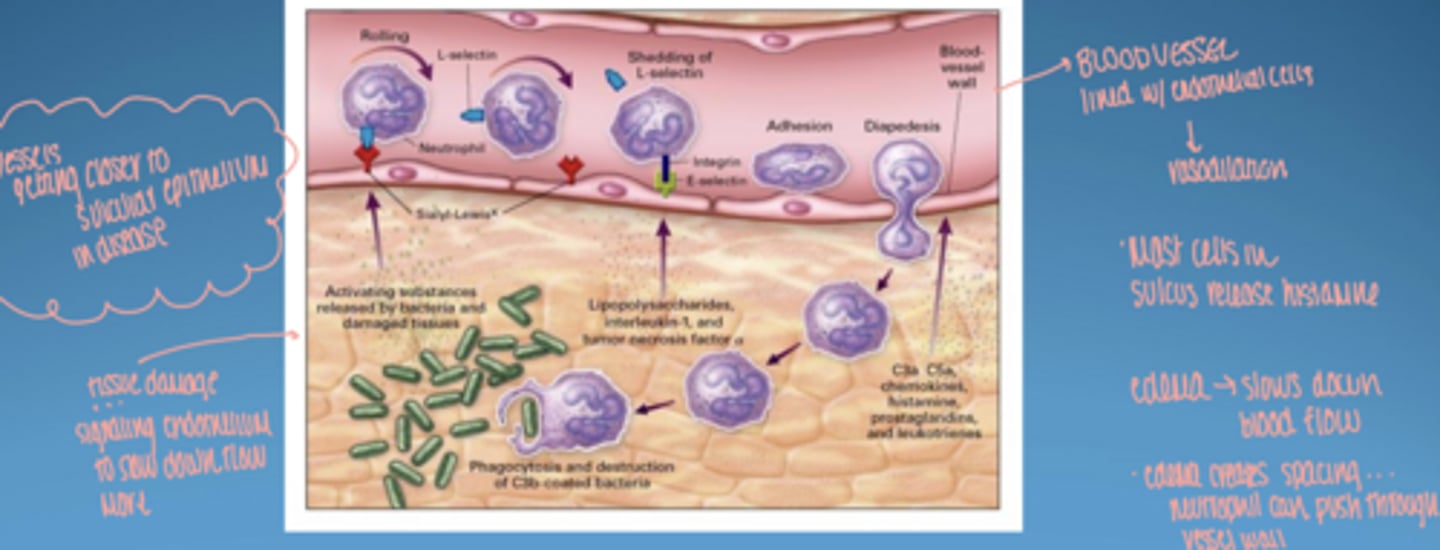
steps of neutrophil migration (1-4)
1. margination-- injury slows down blood flow, neutrophils move to periphery of blood vessel
2. adhesion-- neutrophils attach to adhesion molecules within endothelial wall (ICAM-1, ELAM-1, CMP-140)
3. emigration-- neutrophils exit vessel and go into CT
4. chemotaxis-- move toward bacteria; move through CT to BM and then junctional epithelium to bacteria
steps of neutrophil migration (5-8)
5. opsonization-- recognition & attachment to the target
6. phagocytosis-- engulfing target
7. killing-- respiratory burst or vacuole degranulation
8. digestion of dead bacteria/cells
what causes tissue damage from neutrophils
oxygen free radicals
enzymes
HOST causes damage (collateral damage)
what is chemotaxis (neutrophil migration)
movement of a cell in response to a chemical stimulus
complement dependent:
- C3a, C5a
complement independent:
- leukotriene
- products of bacterial metabolism
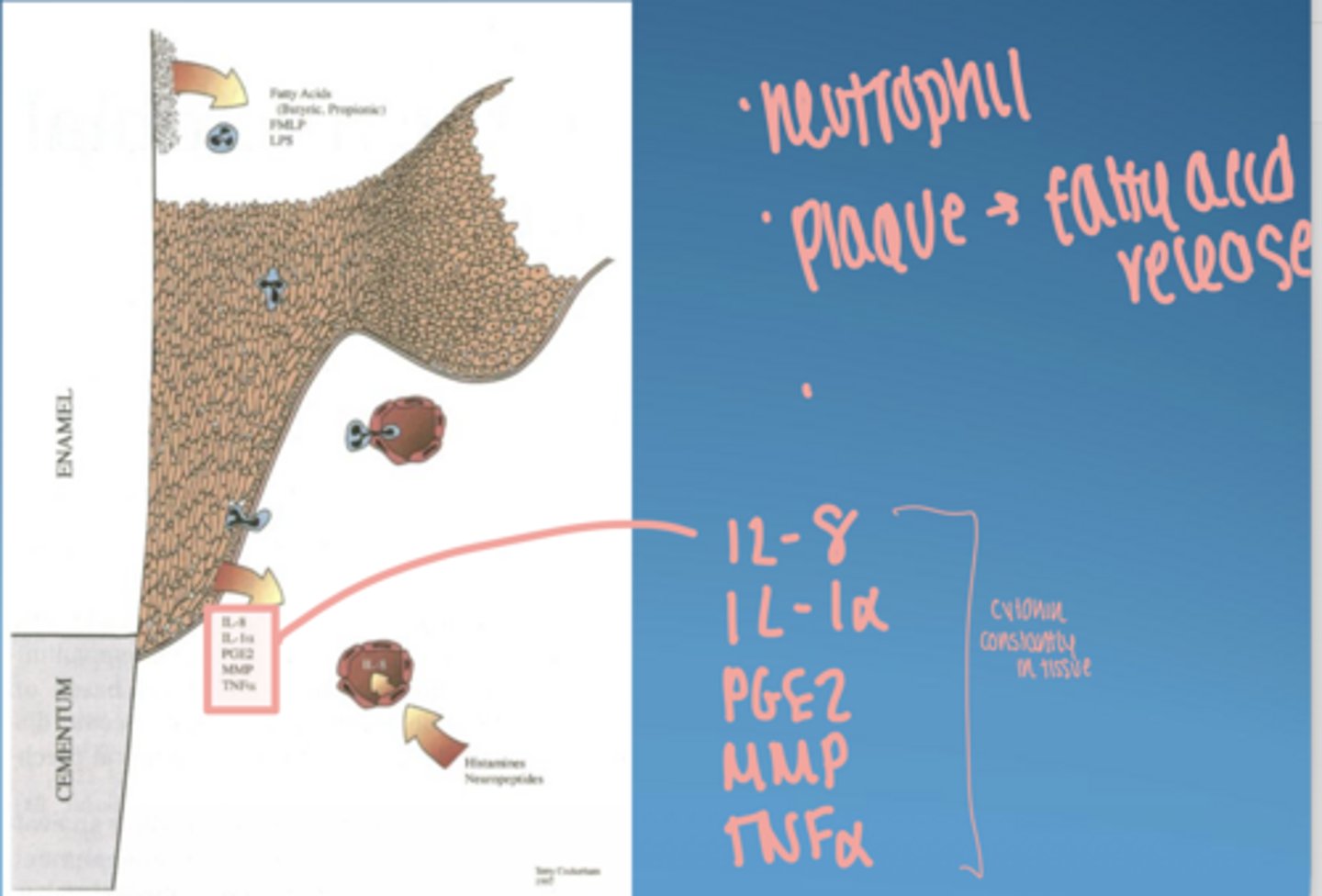
- endotoxin = activated macrophage = IL-8
- tissue injury
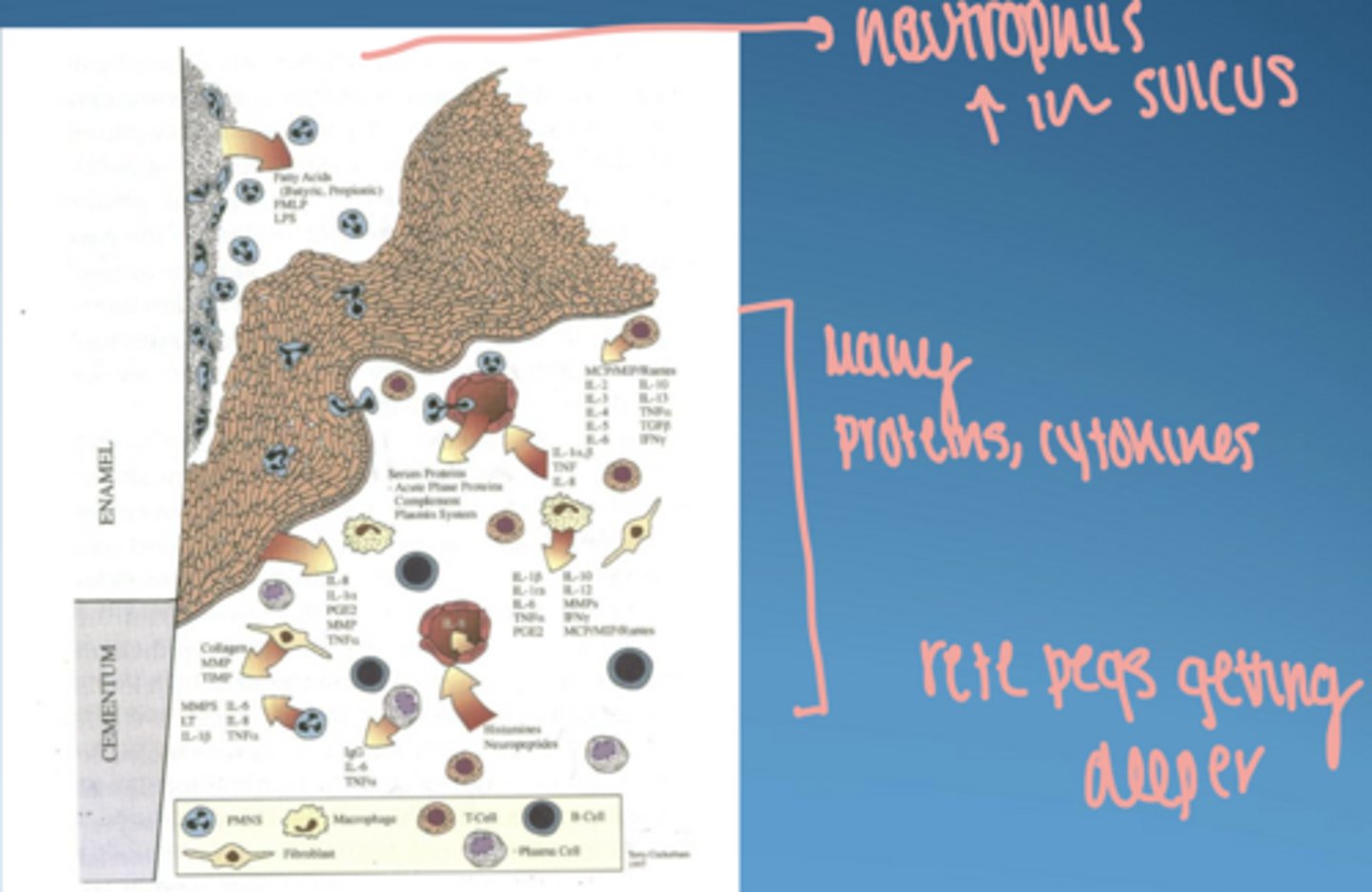
visual of neutrophil migration & chemotaxis
vessels getting closer to sulcular epithelium in disease
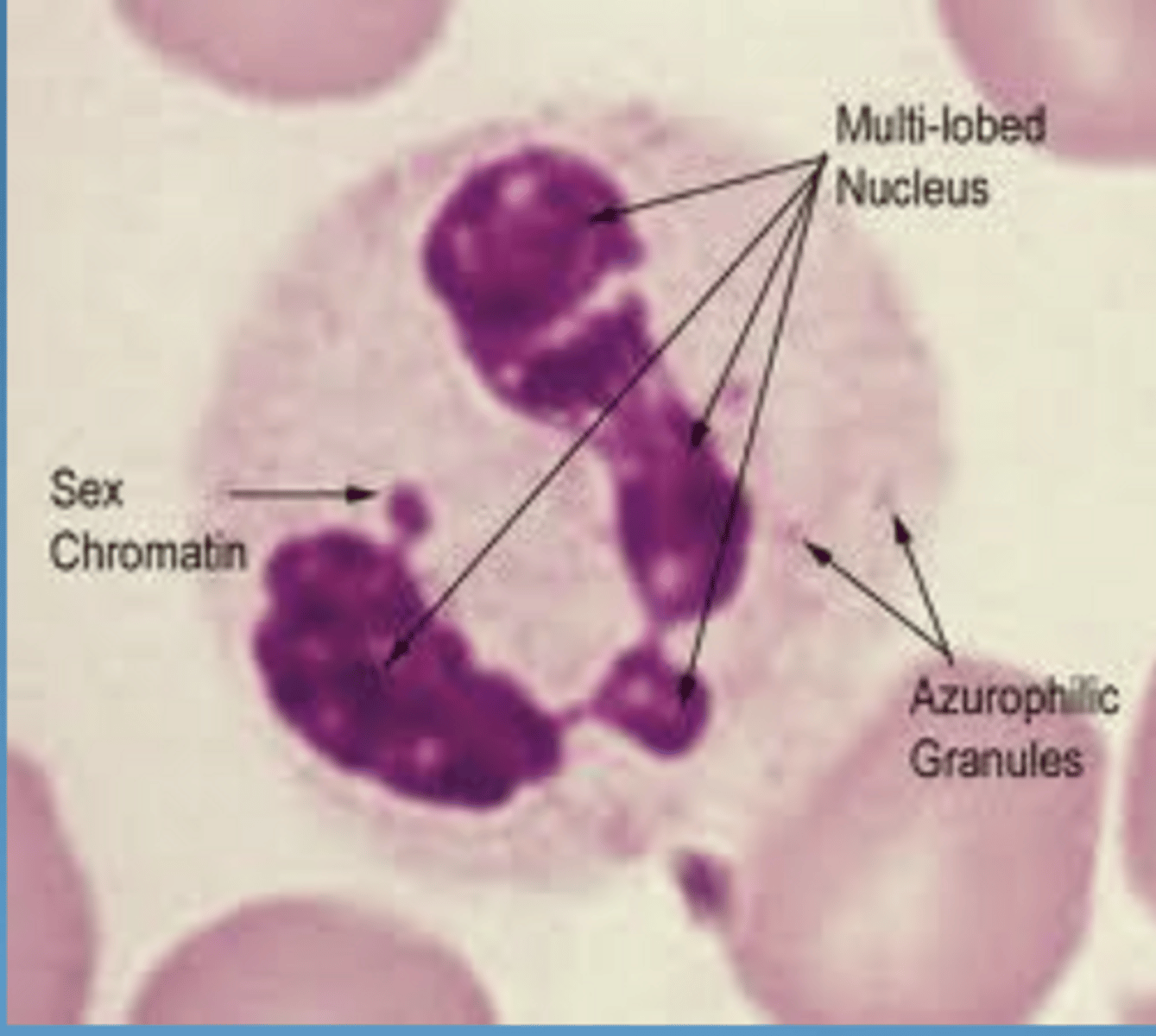
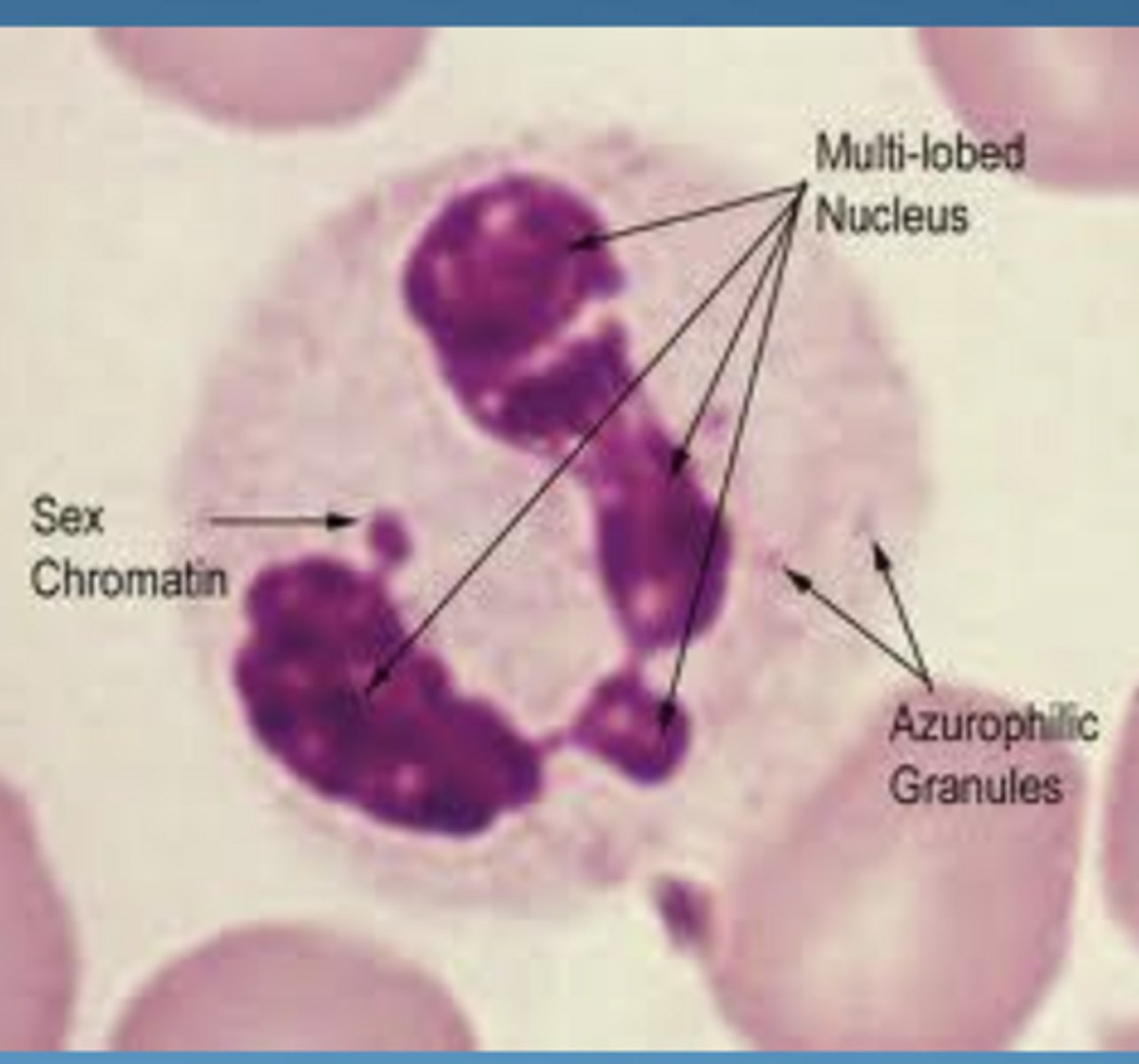
neutrophil granules and contents
azurophil (primary):
- large, dense
- lysosomal enzymes
- peroxidase ("myeloperoxidase")
- lysozyme (33%)
- cationic proteins
specific (secondary):
- smaller, less dense
- alkaline phosphatase
- lysozyme (67%)
- lactoferrin
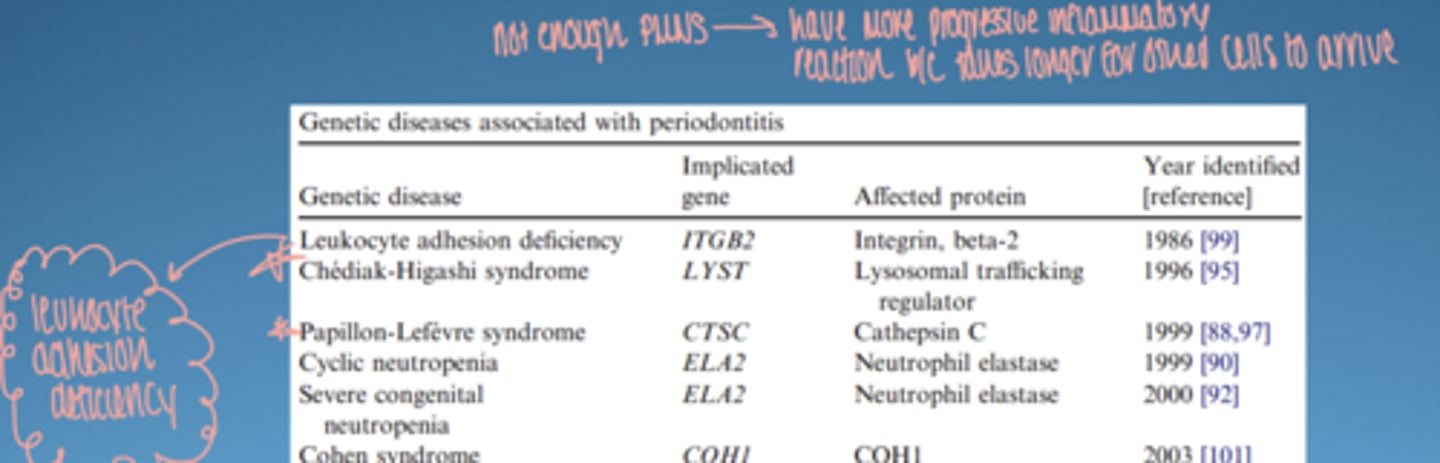
ex. of genetic disease affecting periodontitis
leukocyte adhesion deficiency:
-↓ neutrophil migration → severe disease
papillon-lefevre syndrome:
- causes severe, aggressive periodontitis resulting in early, rapid tooth loss
*not enough PMNS... have more progressive inflammatory reaction b/c takes longer for other cells to arrive*
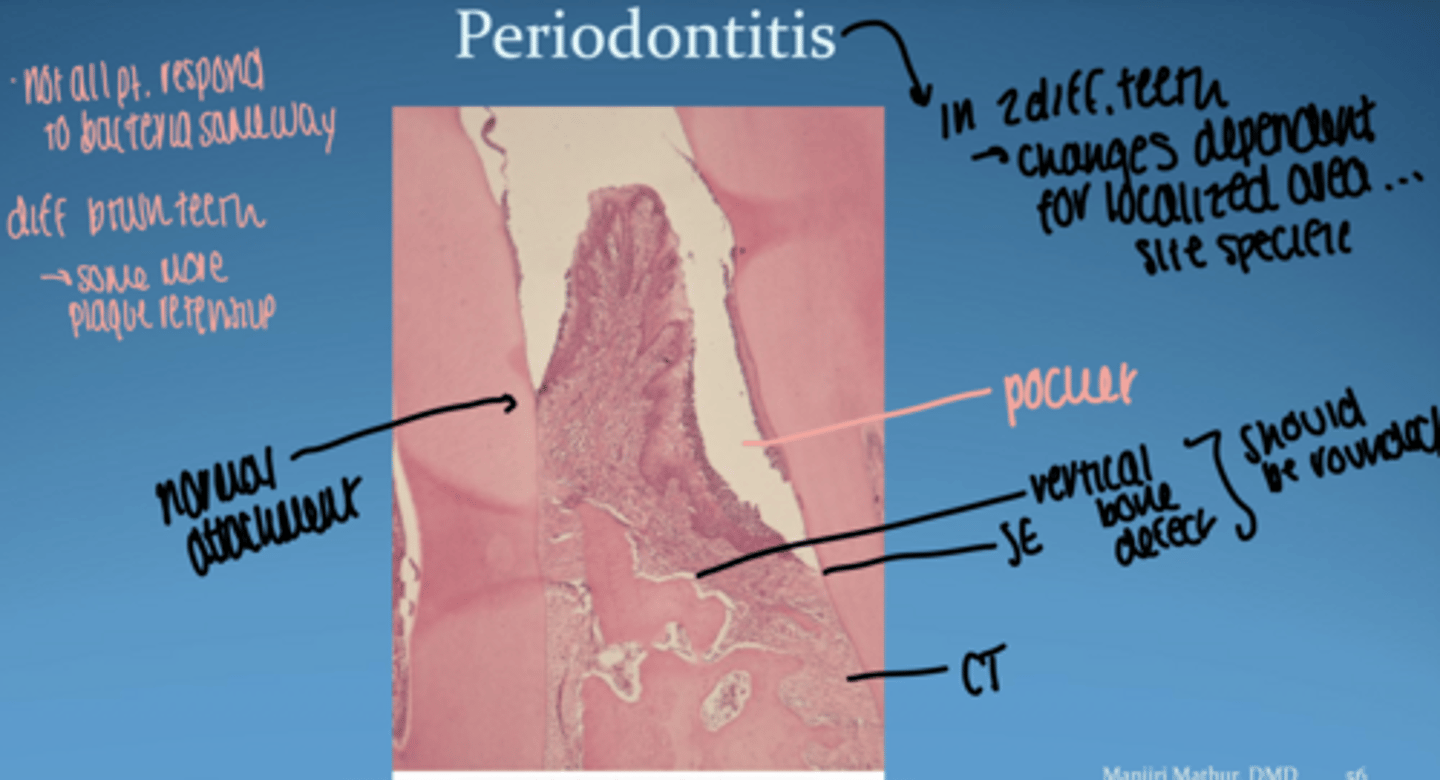
histopathology of gingival disease stages
stage 1 = initial lesion
stage 2 = early lesion
stage 3 = established lesion
stage 4 = advanced lesion
*know what cell present, inflammatory response, and how many days*
histopathology of gingival disease visual
initial lesion time frame
2-4 days
dominant cell in initial lesion
neutrophils
initial lesion
- clinically = looks healthy
- microroganisms in sulcus activate resident leukocytes
- migration of leukocytes into sulcus via chemotaxis
- increase in flow of gingival fluid into the sulcus
initial lesion vascular changes
- vasodilation of capillaries
- increased blood flow/permeability
- stimulation of endothelial cells
- GCF increase
- increased vascular permeability allows PMN and monocytes to migrate through CT to bacteria
initial lesion visual
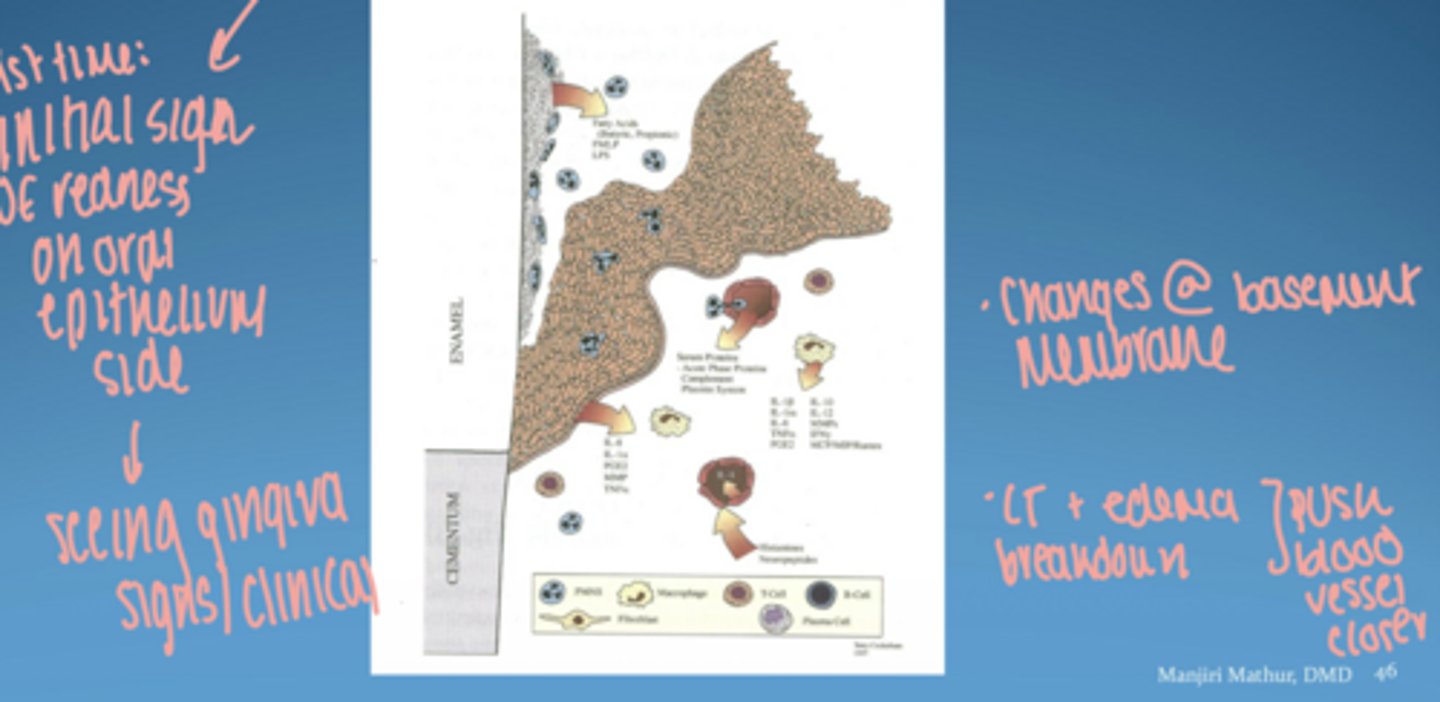
early lesion (4-7 days)
- changes in initial lesion continue to intensify
-proliferation of capillaries, formation of capillary loops
- increased migration of PMNs to epithelium
-phagocytosis of bacteria
- release of lysosomes
-collagen destruction as by product apical & lateral to junctional and sulcular epithelium
-proliferation of basal cells in order to maintain in tact barrier
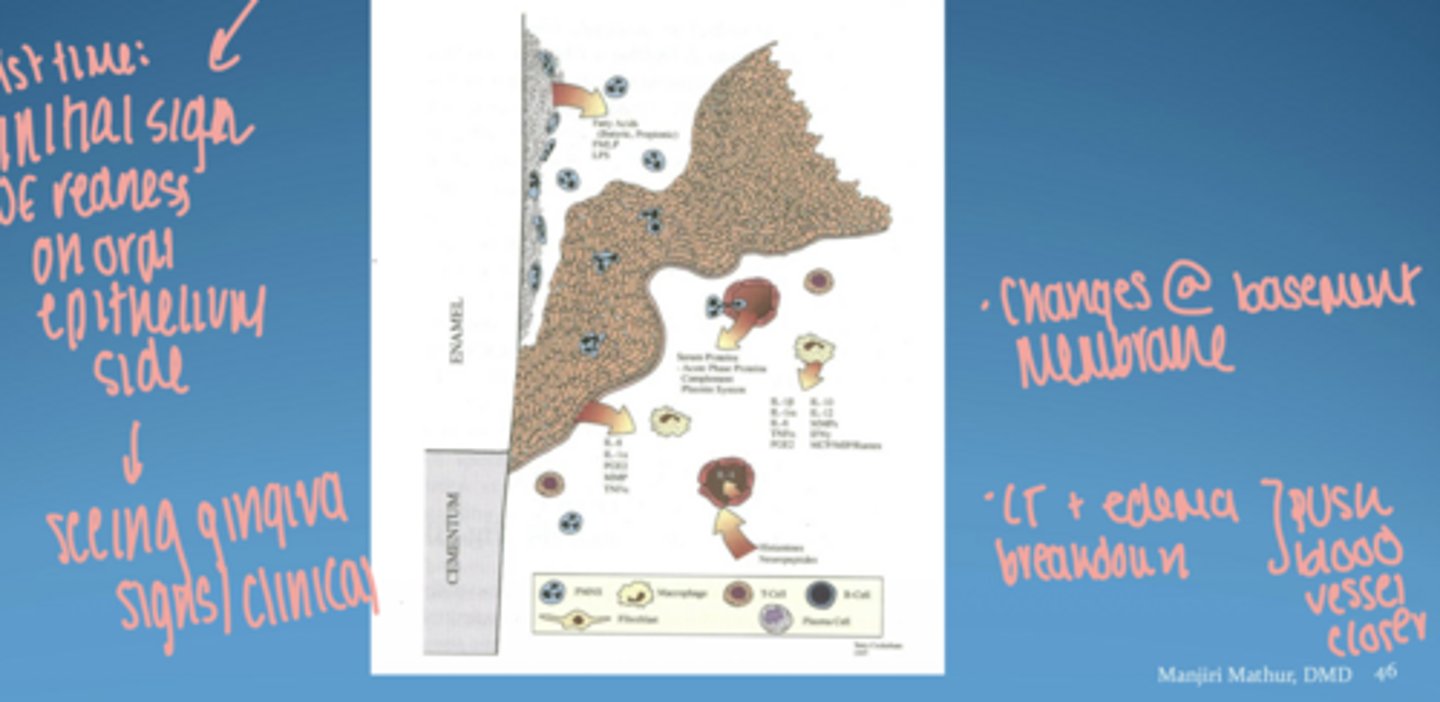
dominant cell in early lesion
T-lymphocytes
key features of early lesion
-collagen breakdown
-rete peg formation; epithelium proliferates into collagen depleted areas of CT
-capillary proliferation
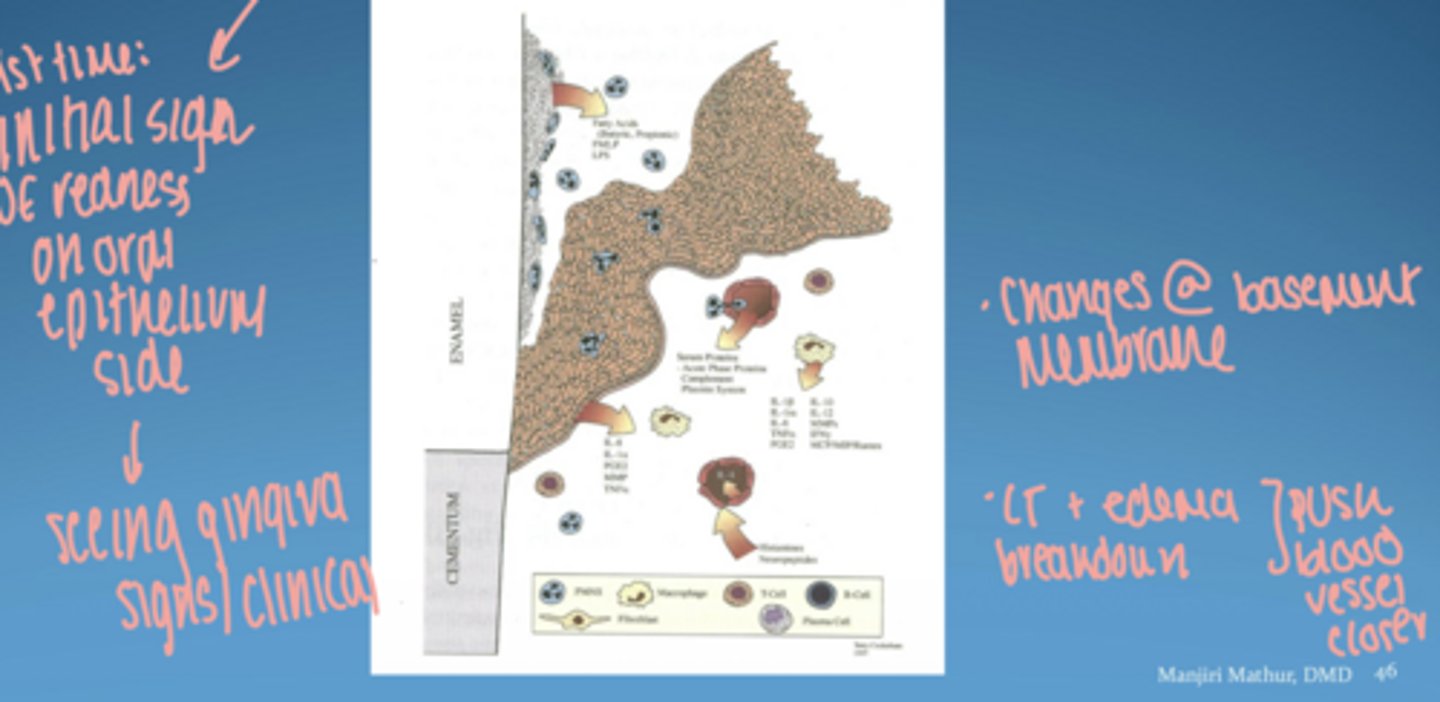
clinical signs -- early lesion
redness
first clinical signs of eryhtema, edema
bleeding on probing
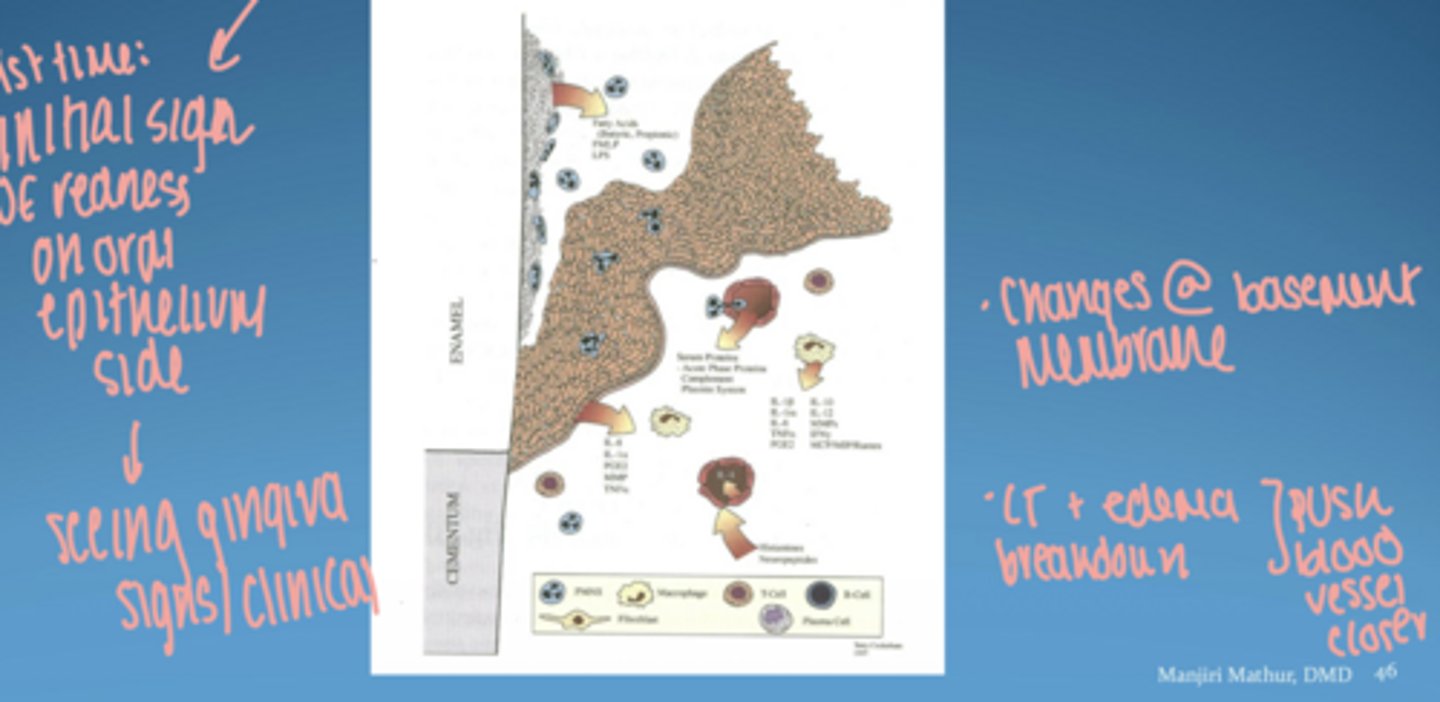
gingivitis (early)
no attachment loss
blood vessels forming capillary loops to get closer to epithelium
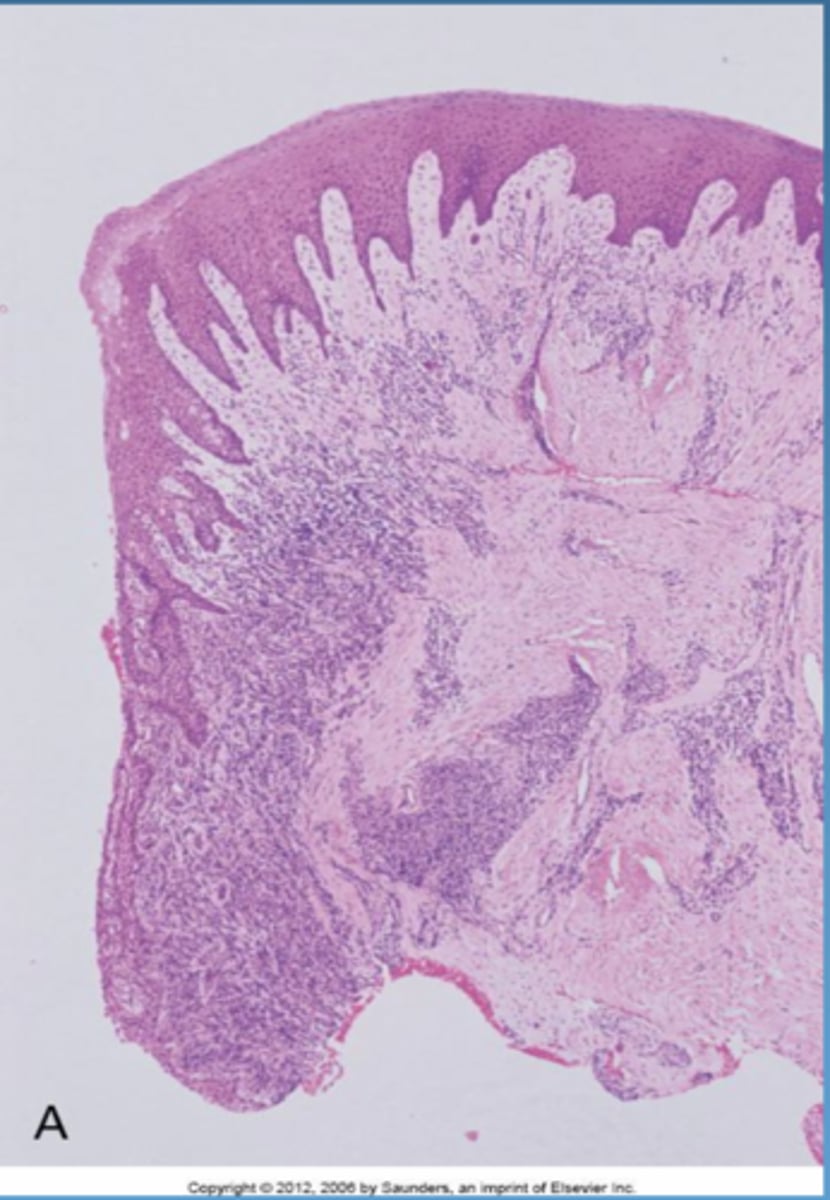
established lesion (14-21 days)
- changes in early lesion worsen
- continued breakdown of collagen, vascular prolfieration, developed/elongated formation of rete pegs
- moderately to severely inflamed gingiva
-changes in color, size, texture, consistency, contour of gingiva
-NO attachment loss
-gingival lesion is reversible
dominant cell in established lesion
plasma cells
is established lesion reversible
yes-- still in gingivitis stage
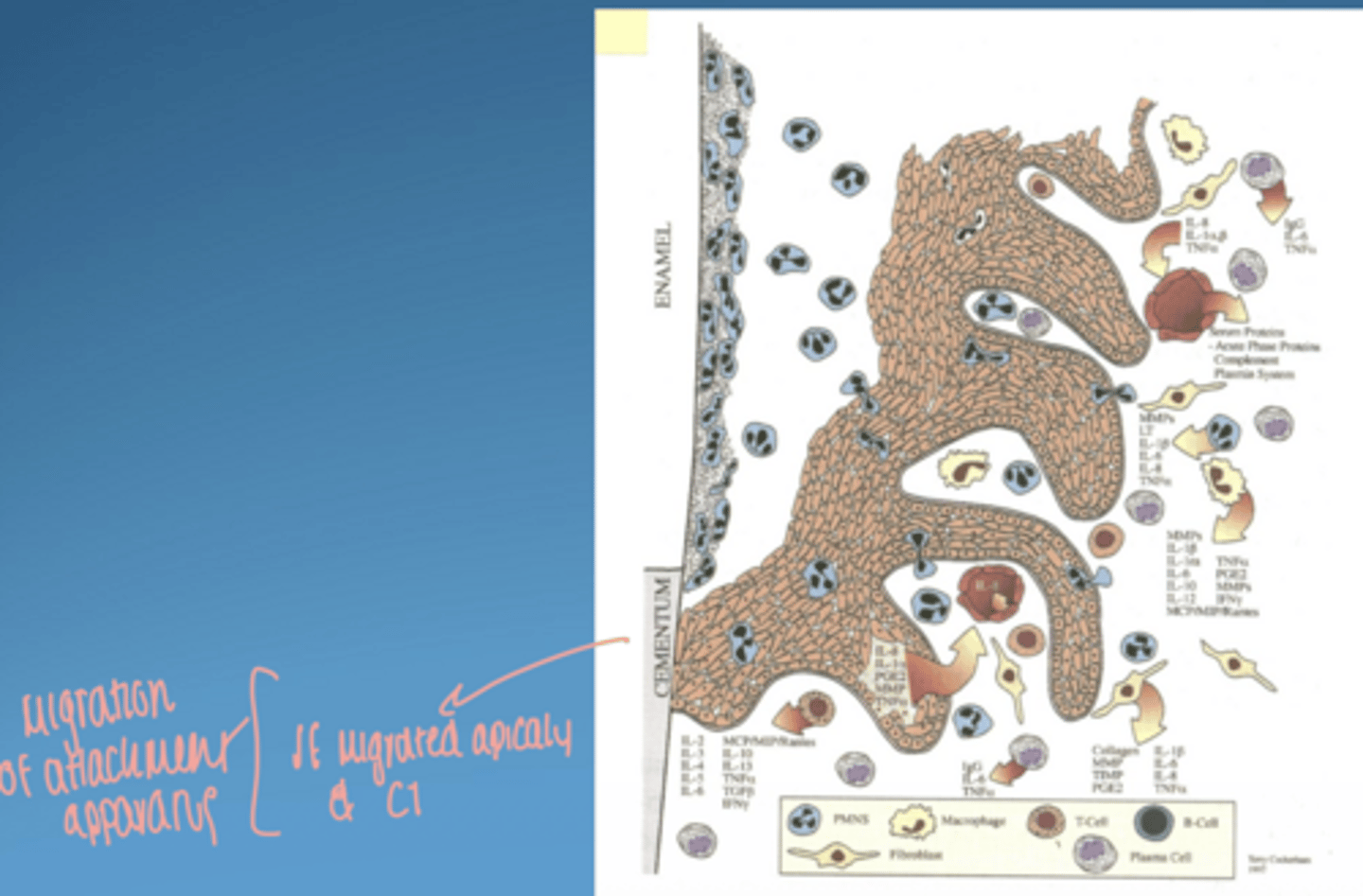
advanced lesion (periodontitis)
- extension of lesion into alveolar bone
- apical migration of junctional epithelium
bone loss:
- RANK/RANKL
-loss of attachment
-clinical signs of acute and/or chronic sings of inflammation may be present
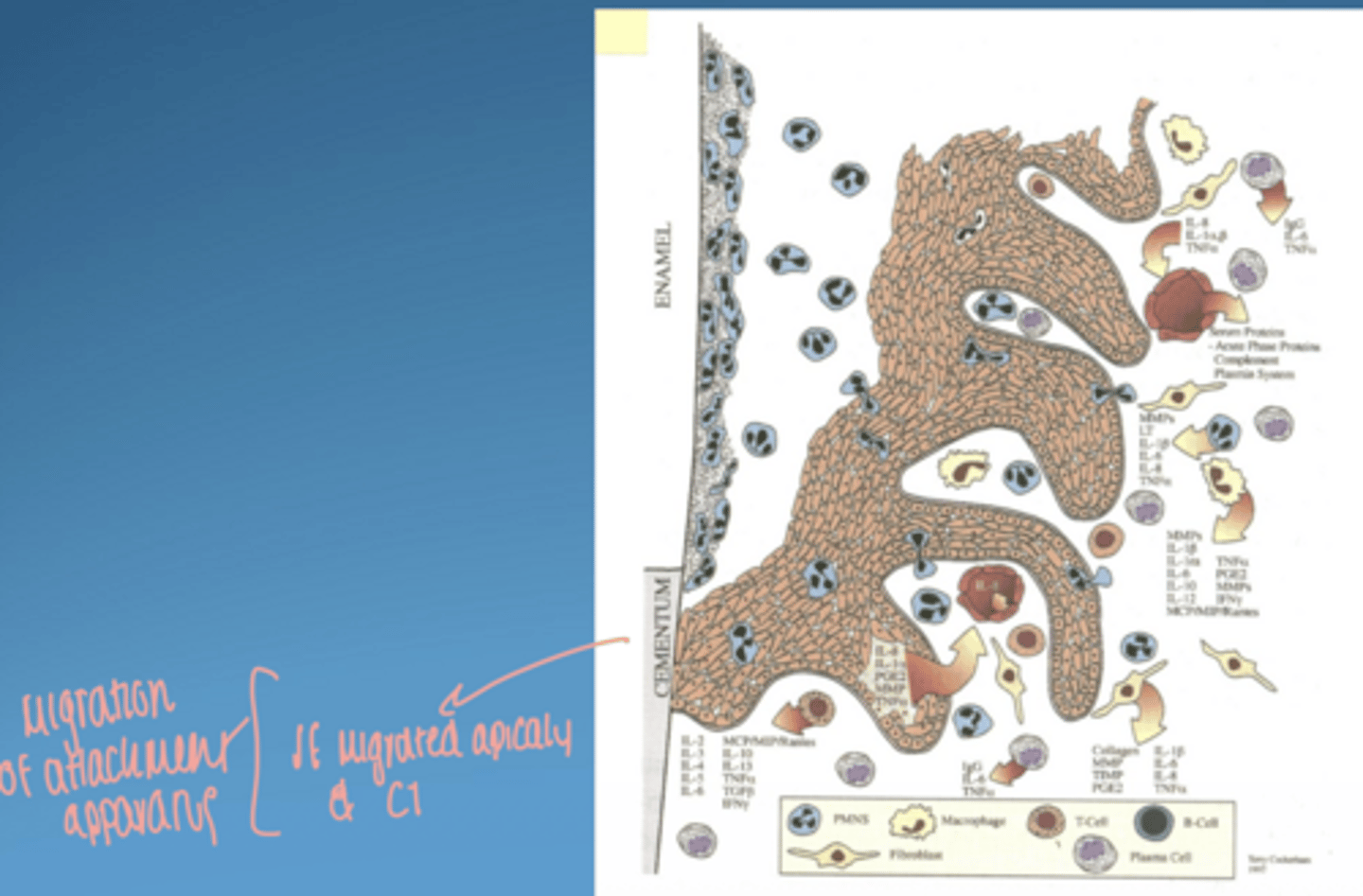
periodontitis visual
-- in 2 diff teeth.. changes are dependent for localized area (site specific)
-- not all pt respond to bacteria same way
microbial virulence factors.
LPS
bacterial enzymes
microbial invasion
inhibition of antimicrobial peptides/ IL-8
- initiate inflammatory response
- contribute directly to tissue damage by release of noxious substances
LPS
- outer membrane of gram -
- aka endotoxin
- elicits strong immune response
- host recognizes LPS via toll-like receptors (TLRs)
what does TLRs recognizing LPS result in
- increased production of inflammatory mediators (cytokines)
- differentiation & recruitment of immune cells
- increased vasodilation & vascular permeability
bacterial enzymes and waste products
- ammonia
- hydrogen sulfide
- butyric acid-- apoptosis of T, B, fibroblasts, & epithelial cells
- proteases
proteases
breakdown of CT components (ex. collagen, elastin, and fibronectin)
gingipains produced by P. gingivalis reduce [ ] of cytokines and inactivate TNF-a
microbial invasion
- bacteria invade intercellular spaces of epithelium & CT (Aa, P.g, F. nuc)
- bacteria invade epithelial cells & allow other bacteria to enter tissue
- bacteria in tissues may act as a "reservoir" for reinfection
*important to maintain treatment b/c bacteria present always & periodontitis is chronic*
cytokines
-key mediators in periodontal disease
-proteins that act as messengers to transmit signals from once cell to another
- bind to receptors on target cells, initiate intracellular signaling, results in gene expression (alteration of cell behavior OR secretion of more cytokines)
- produced by many cells: neutrophils, macrophages, lymphocytes, fibroblasts, epithelial cells
*ex. induce fibroblasts & osteoclasts to produce enzymes to break down CT and bone*
main pro-inflammatory cytokins
IL-1β
TNF-α
IL-6
innate & adaptive
IL-1β functions
↑ prostaglandins
↑ inflammation
↑ bone resorption
- stimualtes synthesis of prostaglandin E2 (PGE2)- vasodilation, bone resportion via RANKL
- increase blood flow to site of infection
- facilitates migration of neutrophils from blood vessel
-increase GCF [ ] in gingivitis/periodontitis
-regulates development of antigen-presenting T cells
what produces IL-1β
monocytes
macrophages
neutrophils
fibroblasts
keratinocytes
epithelial cells
B cells
osteocytes
TNF-α funcitons
↑ neutrophil activity
↑ MMPs (mediates cell & tissue turnover)
↑ osteoclasts & limits tissue repair by osteoblasts
↓ tissue repair
facilitates neutrophil recruitment, IL-1β production, secretion of PGE2
*shares many actions of IL-1β*
*antagonists of IL-1β and TNF-α = ~80% reduction in inflammation and 60% reduction in bone loss*
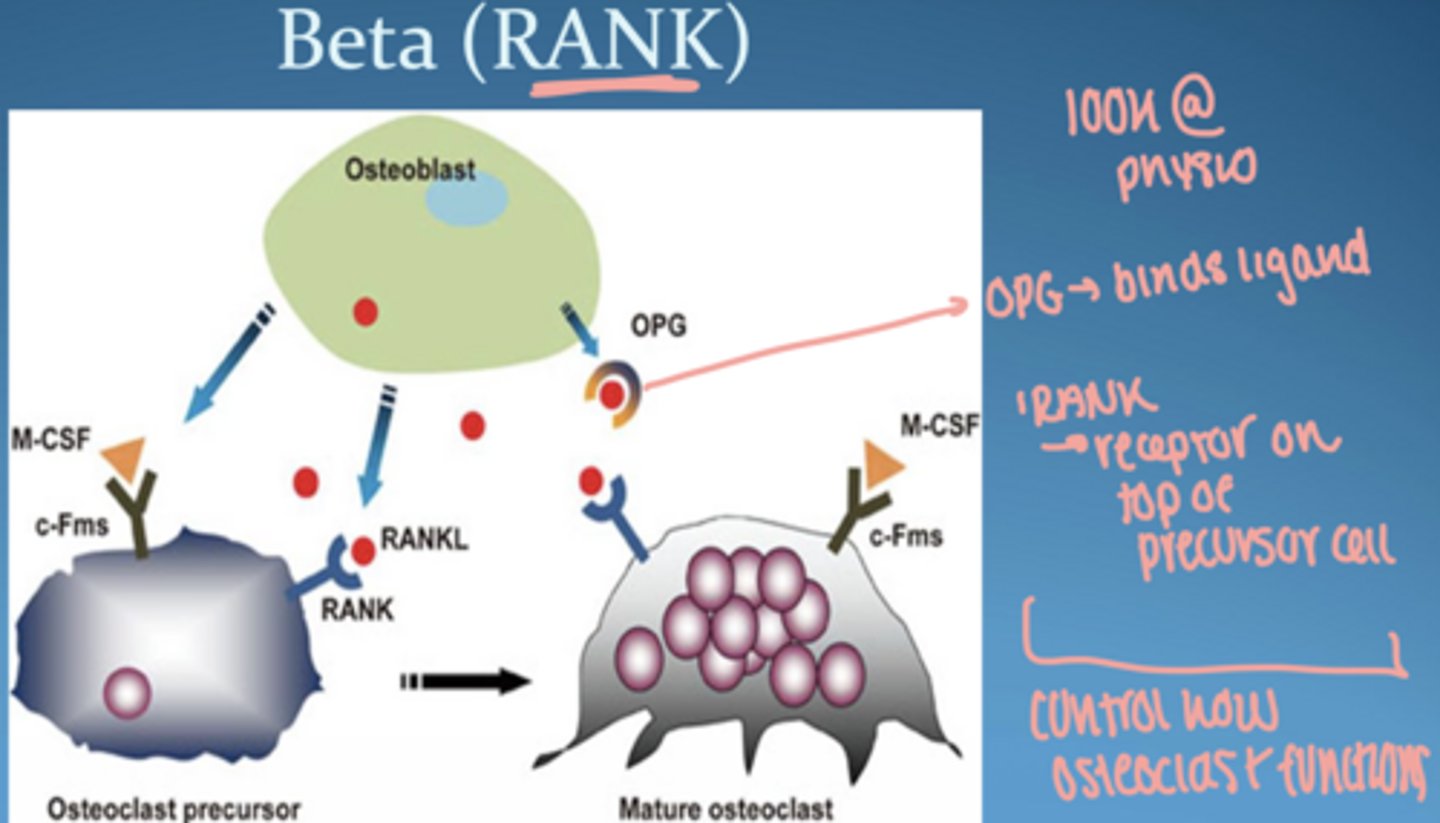
what is RANK (receptor activator of nuclear factor-Kappa beta)
receptor expressed by osteoclast progenitor cells
RANKL
cytokine produced by osteoblast and bone marrow stromal cells
activates osteoclasts → bone resorption
RANKL + RANK = regulate osteoclast differentiation and activation
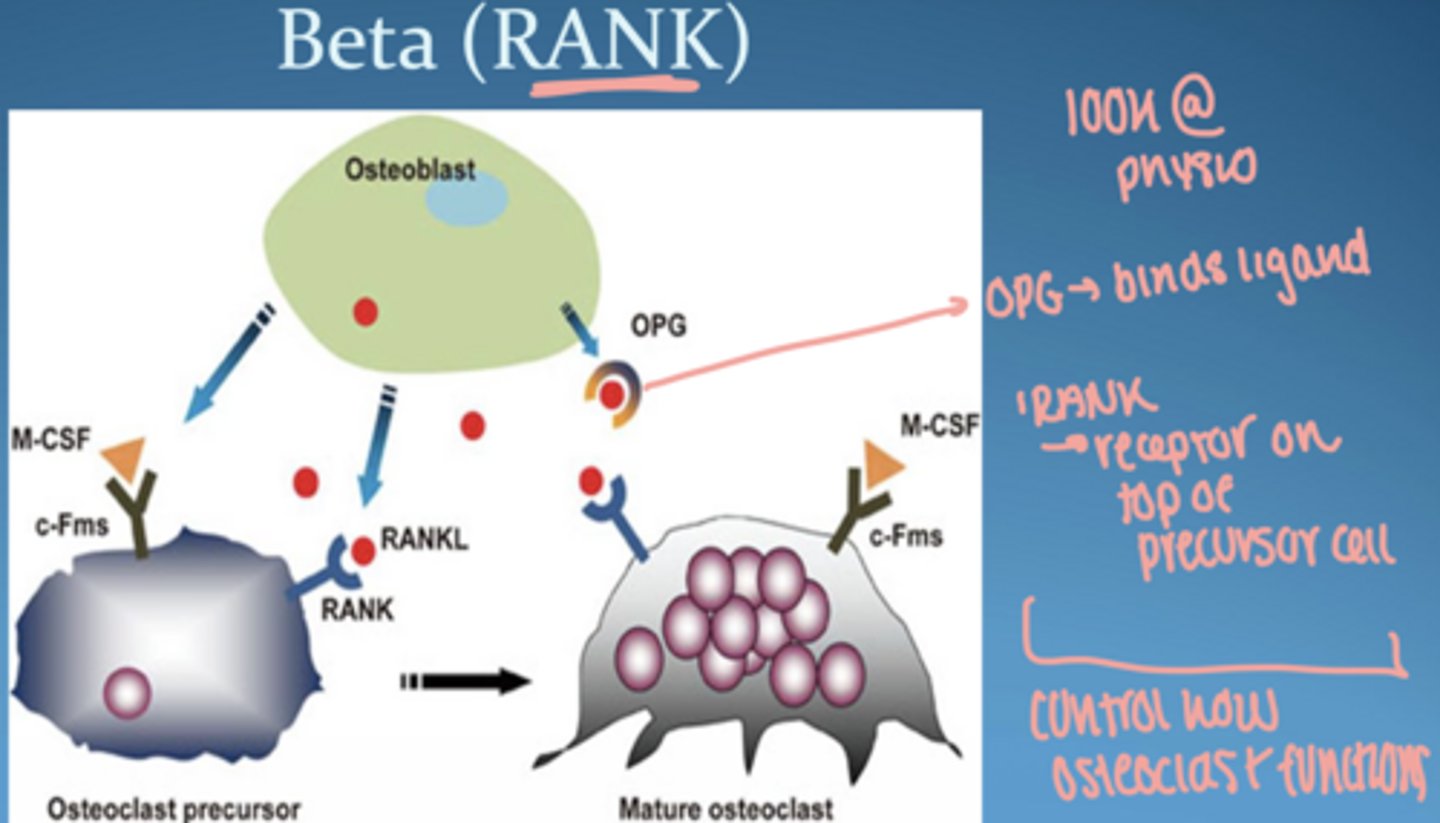
______ is a membrane-bound protein that binds to ____ receptors on osteoclast precursors to stimulate their differentiation, activation, and survival
RANKL, RANK
osteoprotegrin (OPG)
decoy receptor, secreted by osteoblasts
opposes RANKL to downregulate osteoclast activation
blocks RANKL = protects bone
IL-1 & TNF-α regulate the balance between RANKL and OPG
interleukin-6
stimulated by IL-1 β, TNF- α
secreted by many immune cells & osteoblasts to stimulate bone resorption and development of osteoclasts
matrix metalloproteinases (MMPs)
in health, MMP1 (collagenase) secreted by fibroblasts to maintain CT homeostasis
MMP production upregulated by IL-1 β, TNF- α and also produced by bacteria
in disease = MMPs secreted in excessive quantities and cause collagen and CT breakdown

when does adaptive immunity start
initiated in early lesion → dominant in established lesion (plasma cell)
if innate immunity fail to eliminate infection, then cells of adaptive immune are activated BUT innate doesn't turn off
adaptive immunity involves...
interactions between APC and T/B cells
non-progressing peridontal lesion
dominated by T cells
progressing periodontal lesion
dominated by plasma cells
TH1 vs TH2
TH1:
- cell mediated (APC)
-macrophages- phagocytose baceria
- NK cells & CD8 cytotoxic cells kill infected host cells
TH2:
- antibody- mediated (humoral)
- B cells produce plasma cells
- plasma cells produce antibody
- release of pro-inflammatory cytokines that lead to tissue destruction (bone loss in advanced lesion)
APC
macrophage:
- phagocytic from myeloid lineage
- ingest particulate antigen
- express MHC class II molecules to induce costimualtory activity on T cells
- homeostasis and defense
does gingivitis always become periodontits
no (despite poor oral hygiene)
depends on host susceptibility

Sir Lankan study
test laborers who had no access to dental care
3 groups:
- rapid progression (8%)
- moderate (81%)
- no progression (11%), only gingivitis but no periodontitis
same plaque, different outcomes → HOST RESPONSE matters
the major determinant of susceptibility to disease i the nature of the ____-_____ response
immune-inflammatory
*not everyone has same response to plaque bacteria*
host susceptibility
-patients with periodontal inflammation have a high concentrations of
TNF-α, IL-1β, RANKL and MMP-13
- immune responses to bacterial challenge take place in the context of other host and environmental factors that influence these responses and determine progression of disease
- more susceptible = excessive, or dysregulated, immune-inflammatory breakdown compared to those with a normal response
- bacterial threshold btwn stable and active disease that varies person to person
_____ ____ increase susceptibility to periodontal disease
risk factors
ex. smoking, diabetes, stress, genetics
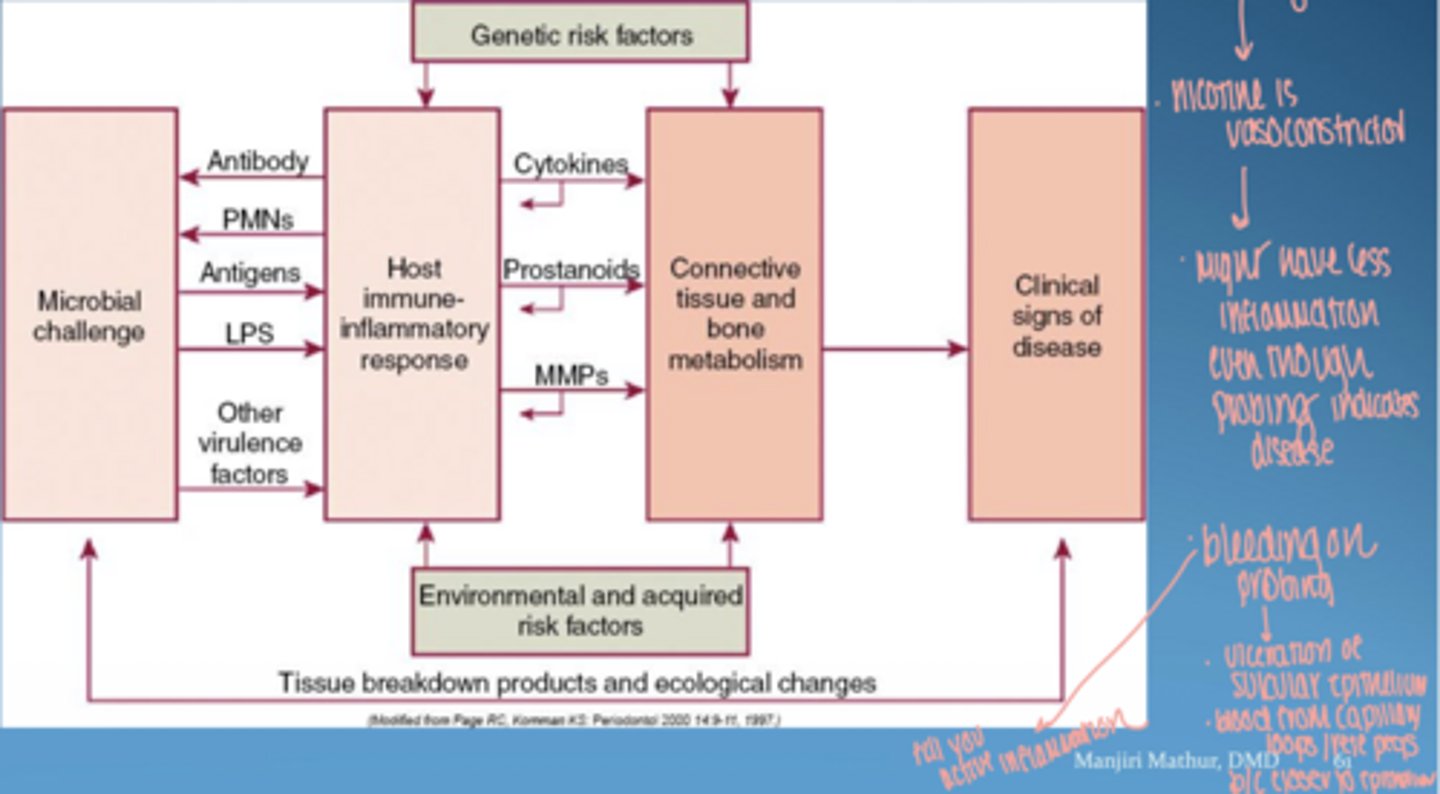
why is smoking tricky
vasoconstriction → ↓ bleeding
disease may be present but masked
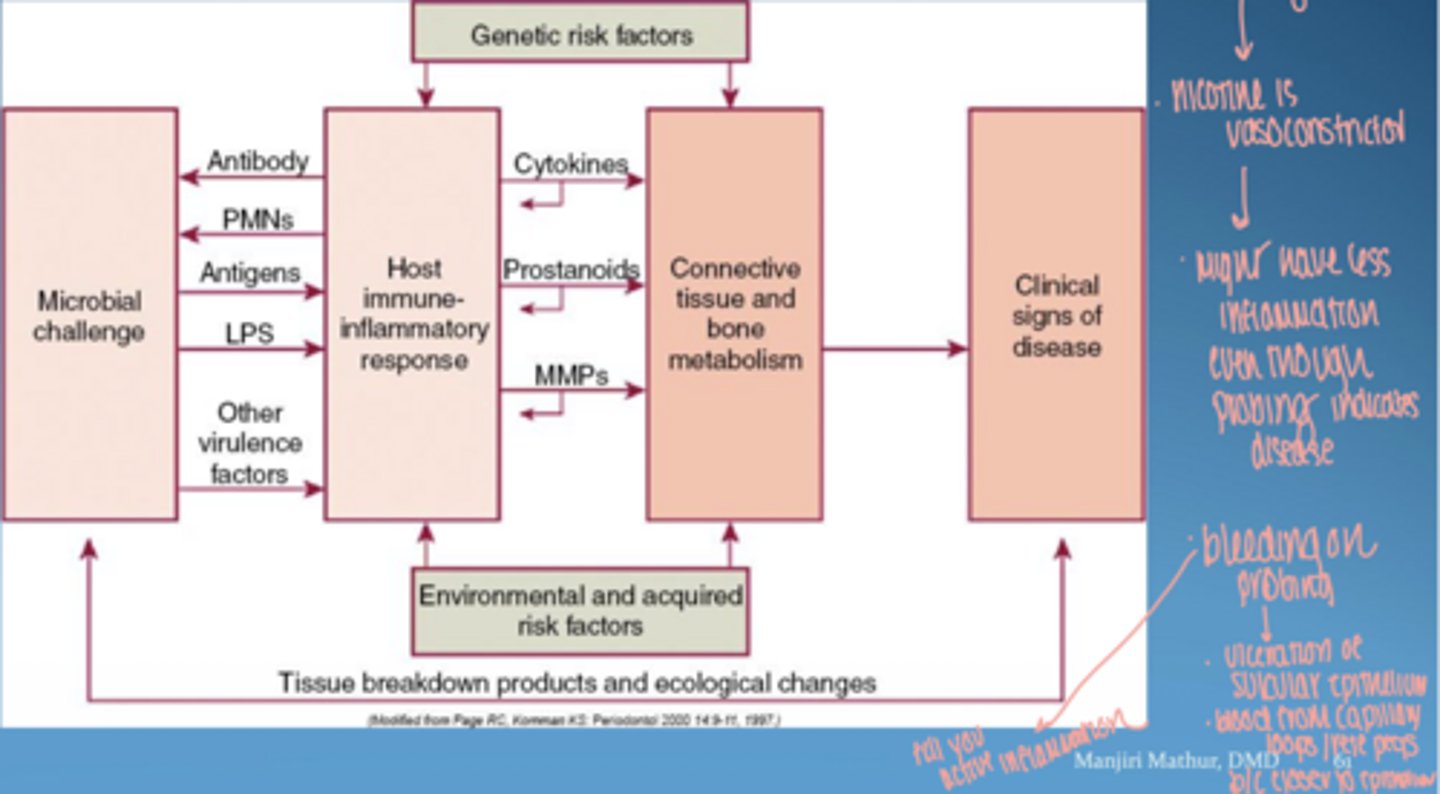
______ _____ may result in hyperinflammatory traits, increasing susceptibility (same bacterial challenge elicits a greater inflammatory response, increased tissue breakdown)
genetic polymorphisms
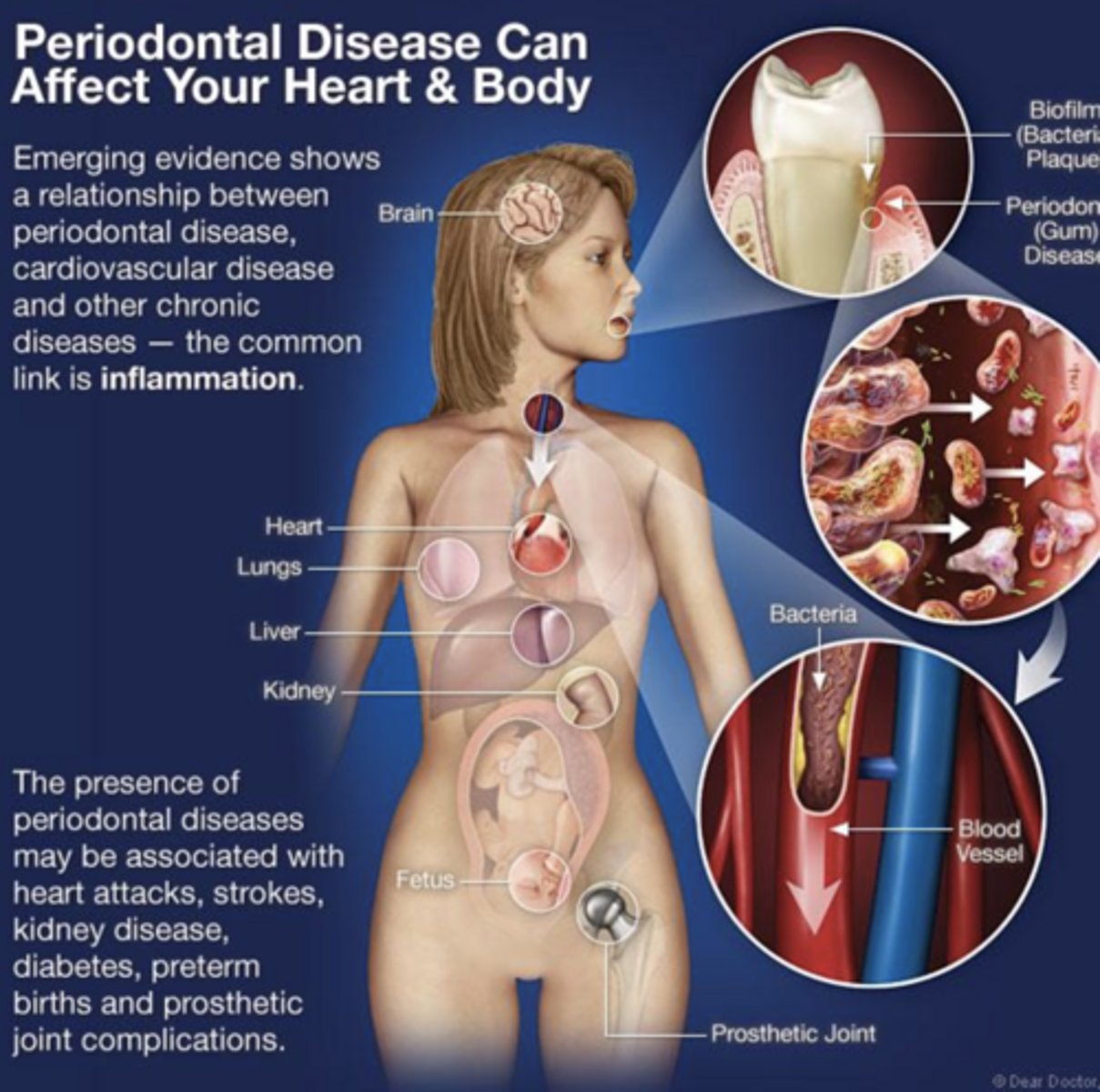
how gum disease affects body
1. alzheimers
2. heart disease
3. diabetes
4. pregnancy, health of fetus
5. overall inflammation

periodontal disease & heart
presence of periodontal disease may be associated with heart attacks, strokes, kidney disease, diabetes etc.
goal of periodontal therapy
reduce inflammation & biofilms
methods:
- plaque control
- mouthwash
- antibiotics
-scaling/ root planing
-surgery
