Antimicrobials targetting cell wall structure
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Give examples of β-lactams and glycopeptides
β-lactams - penicillins, cephalosporins, carbapenems, monobactams, clavulanic acid, sulbactam, tazobactam
Glycopeptides - vancomycin, teicoplanin
Cycloserine - targets peptidoglycan
Isoniazid - targets mycolic acids
Ethambutol - targets arabinogalactan
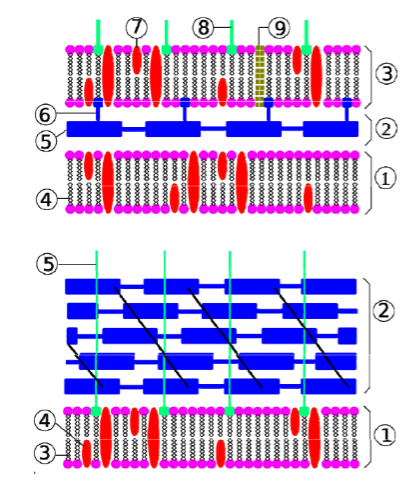
What are the differences in structure between gram- and gram+ bacteria?
Gram Negative
1-inner membrane
2-periplasmic space
3-impermeable outer membrane
4-phospolipid
5-peptidoglycan
6-lipoprotein
7-protein
8-Lipopolysaccharide
9 porins
Gram Positive
1-cytoplasmic membrane
2-peptidoglycan
3-phospholipid
4-protein
5-lipoteichoic acid
Why are gram- bacteria harder to treat and more resistant?
Impermeable outer membrane acting as a barrier, specialised efflux pumps that expel drugs, and high genetic plasticity.
Limits entry of many antibiotics, making them difficult to treat
What is the benefits and downsides to bacteria having peptidoglycan?
Benefits - strength, survive in hostile environment as unicellular organisms
Downsides - easier for us to treat as we don’t have it to get damaged by antibiotics
What do β-lactams and glycopeptides do?
Inhibit the enzymes for the synthesis of peptidoglycan
What is gram- peptidoglycan made out of?
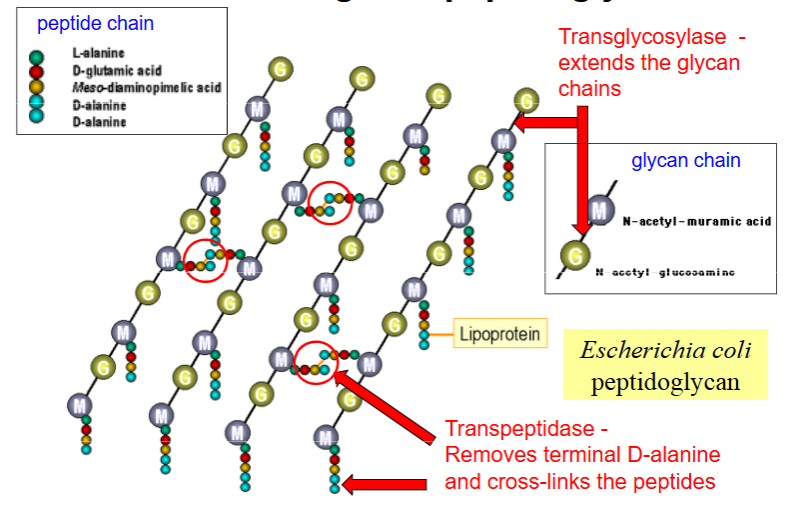
What is gram+ peptidoglycan made out of?
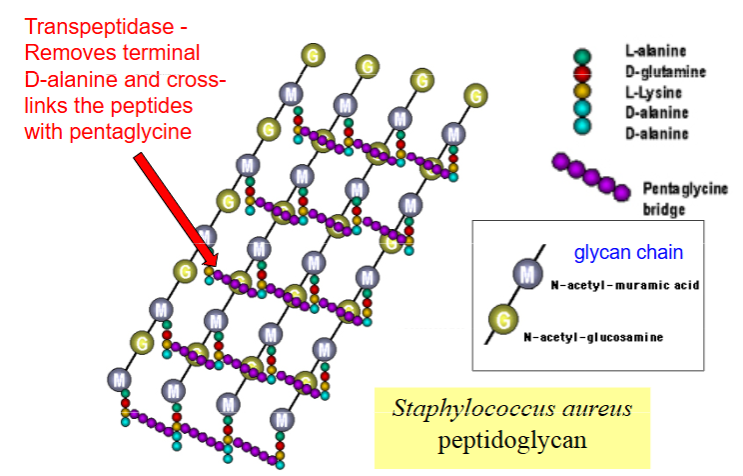
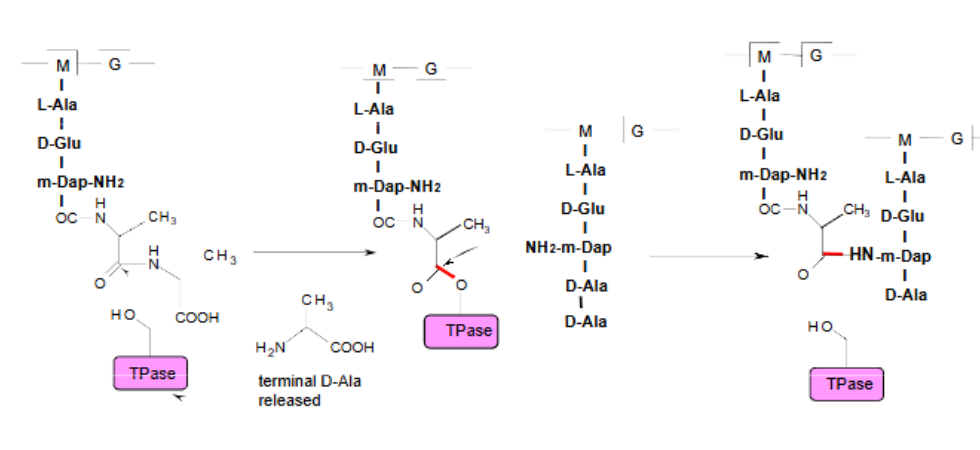
How are cross links formed in peptidoglycan?
Transpeptidation (4–3 crosslinks) - DD-transpeptidases (PBP) form a bond between the 4th residue (D-alanine) of one chain and the 3rd residue (meso-diaminopimelic acid or L-lysine) of another.
Alternative pathway (3–3 crosslinks) - LD-transpeptidases (LDTs) create 3-3 crosslinks, connecting the 3rd residue to the 3rd residue of neighboring chains.
Energy generation - reaction involves the cleavage of a terminal D-Ala-D-Ala linkage, which provides the energy needed to form a new peptide bond.
Bridge structures - In some bacteria, such as Staphylococcus aureus, the cross-link is not direct but involves a short interpeptide bridge, such as a pentaglycine chain, connected to the D-Ala of one stem and the L-lysine of another.
Amide → ester → amide
What is the structure of β-lactams and what is penicillins’ mechanism of action to inhibit petidoglycan synthesis of gram+ bacteria?
Its β-lactam ring irreversibly binds to PBPS as it has a similar structure to D-Ala-D-Ala
Inactivates penicillin-binding proteins, specifically DD-transpeptidase
Blocking transpeptidation prevents the cross-linking of peptidoglycan chains.
This weakens and unstabilises the cell wall, → to osmotic lysis and cell death
May also activate bacterial autolysins, enzymes that degrade the cell wall, accelerating breakdown of cell wall, contributing to bactericidal activity
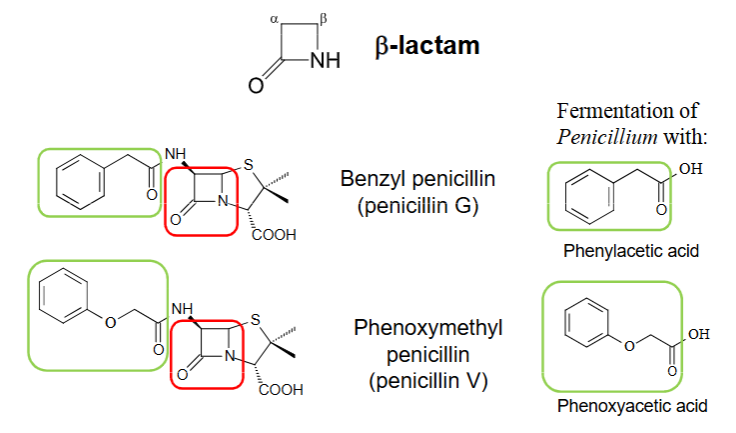
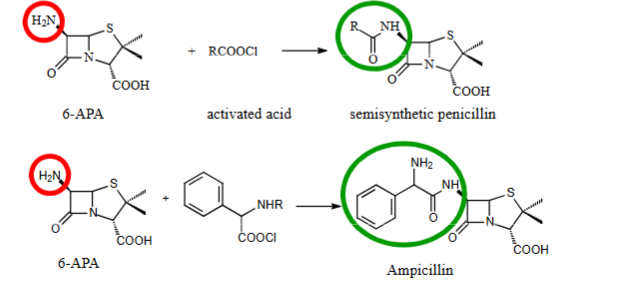
How are semi-synthetic penicillins made?
When starved of phenylacetic acid, Penicillium chrysogenum produces the penicillin nucleus, 6-aminopenicillanic acid (6-APA).
6-APA has little intrinsic activity
6-APA can be converted to an active penicillin by reaction with an activated acid (e.g. acyl chloride)
Can add side chains to make new antibiotics, that have better properties (Amp is stable in GI, large doses ok, slow metabolism, absorbs well across fat)
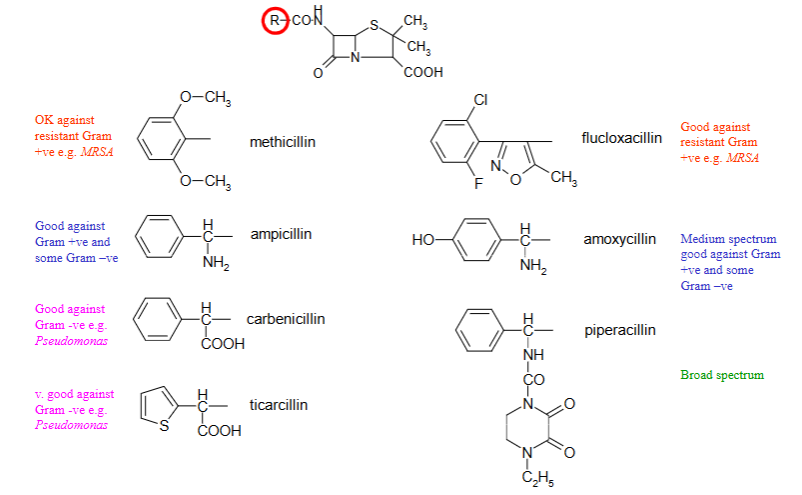
Structures of semi-synthetic penicillins
Carbenicillin - good for gram-
Methicillin - good for resistant (mrsa) and gram+
Ampicillin - good for gram+, some gram- and β‑lactamases inhibitor
Ticarcillin - good for gram-
Flucloxacillin - good for gram+
Amoxicillin - broad spec and β‑lactamases inhibitor
Piperacillin - 2nd line as its broad spec antibiotic, good for resistant bacteria and β‑lactamases inhibitor
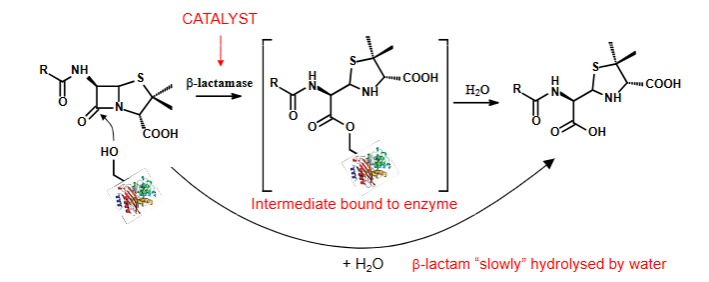
How do bacteria have resistance to penicillin?
May produce β‑lactamases that hydrolyse the β‑lactam ring.
Mutations in PBPs can reduce penicillin binding.
Overuse accelerates the emergence of resistant strains
What is the mechanism of action of beta lactamases?
β‑lactamases catalyse hydrolysis of the amide bond in the β‑lactam ring.
This opens the ring, destroying the structural mimicry of D‑Ala‑D‑Ala needed for binding to PBPs.
Once the ring is opened, the antibiotic cannot inhibit transpeptidases, so cell‑wall synthesis continues normally
What are the 2 types of β‑lactamases?
Serine β‑lactamases (Classes A, C, D) - active‑site serine to attack the β‑lactam ring.
Metallo β‑lactamases (Class B) - Zn²⁺ ions to activate water for hydrolysis.
What are the β‑lactamases inhibitors 2 mechanisms of action?
They resemble β‑lactam antibiotics closely enough that β‑lactamases bind them over the actual antibiotic.
Suicide - clavulanic acid, tazobactam, and sulbactam
Inhibitor enters the β‑lactamase active site.
The enzyme hydrolyses it → a covalent intermediate.
Inhibitor irreversibly inactivates the enzyme, destroying its catalytic function, no antibiotic hydrolysis
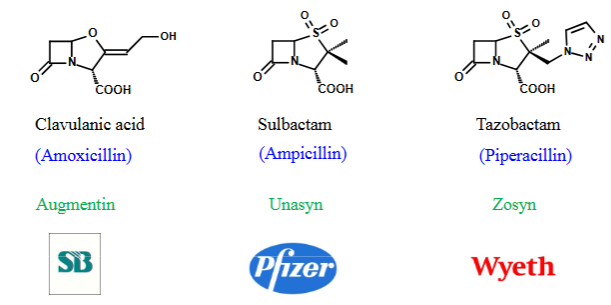
How does avibactam inhibit β‑lactamase?
Binds reversibly to the β‑lactamase active site.
Forms a stable, reversible covalent bond that prevents hydrolysis of the antibiotic.
After dissociation, avibactam can inhibit additional enzyme molecules, giving it broad and sustained activity
What are cephalosporins?
β-lactam antibiotic
For penicillin resistant bacteria
Treats pneumonia, meningitis, and skin infections
Limits clinical stability so semisynthetics (5 generations) are made
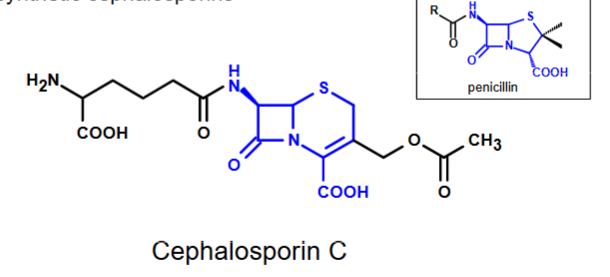
What are carbapenems?
β-lactam antibiotic - Thienamycin
Broad spec (G+ve and G-ve) and resistant to β-lactamase
Semi-synthetic carbapenems - Imipenem (sensitive to renal peptidase) Meropenem (resistant to renal peptidase)
Prescribed for multi-drug resistant in hospitals
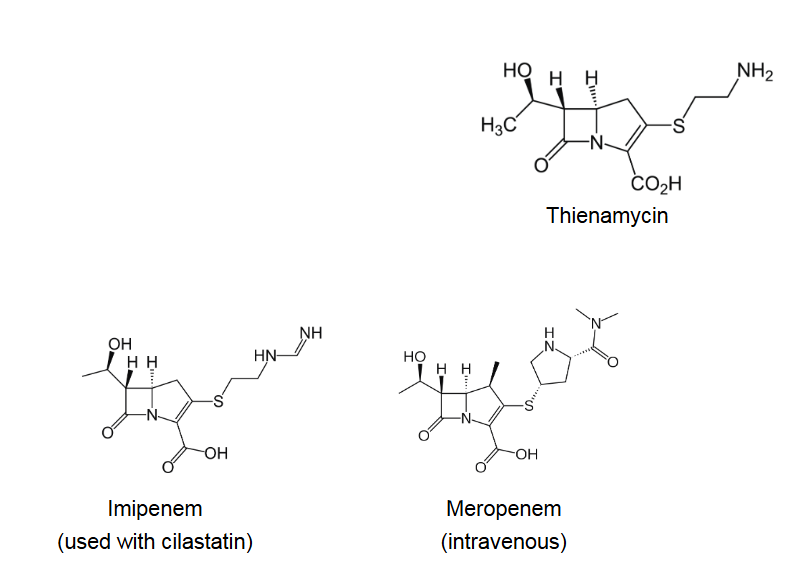
What are monobactams?
β-lactam antibiotic - Nocardicin A
Aztreonam is a synthetic monobactam, resistant to β-lactamases and is the only clinically used monobactam
Tolerated by patients who are hypersensitive to penicillins
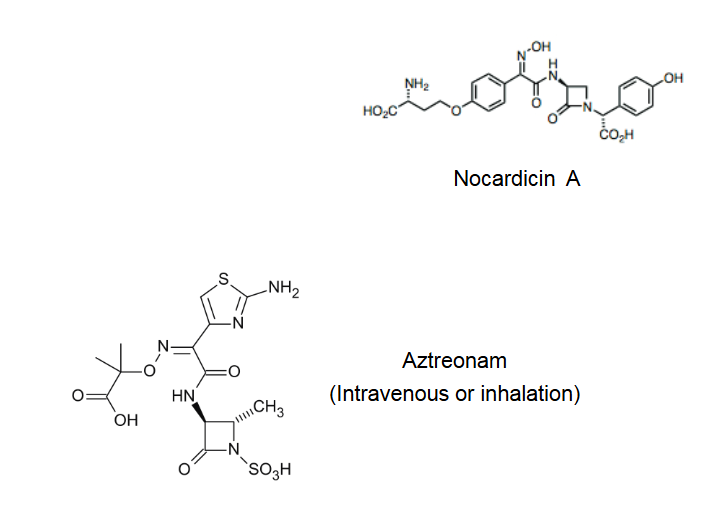
What are the ADRs for β-lactam antibiotics?
Type I (IgE) response believed to be due to “haptenization” of proteins
Diarrhoea, nausea, urticarial rash
Occurs in 1% of patients and severe anaphylaxis occurs in 0.01% of patients
Cross-sensitivity is low between classes
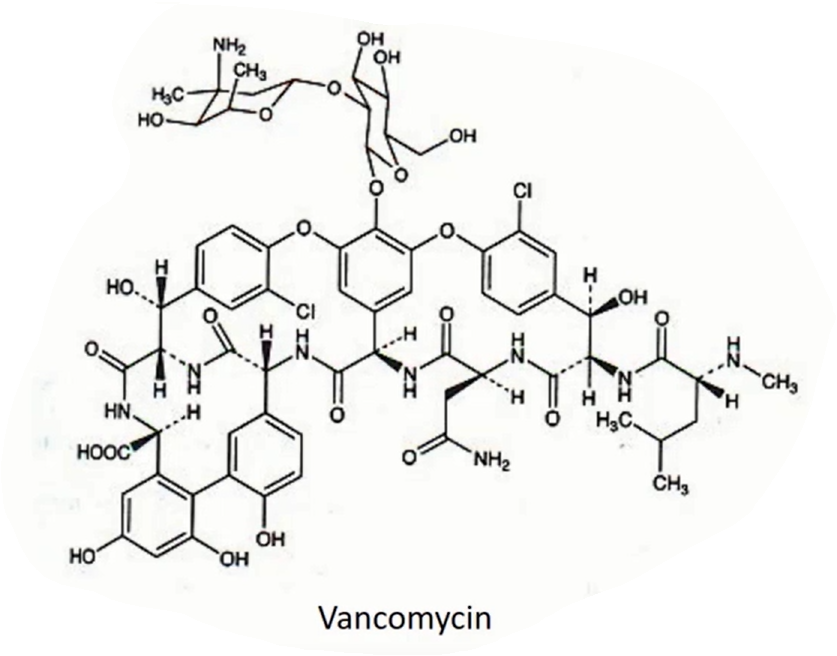
What are glycopeptide antibioitcs?
Vancomycin
Effective against gram+ bacteria as they’re big and too lipophilic
Natural but no produced by fermentation
Biosynthesis by unusual non-ribosomal peptide synthesis
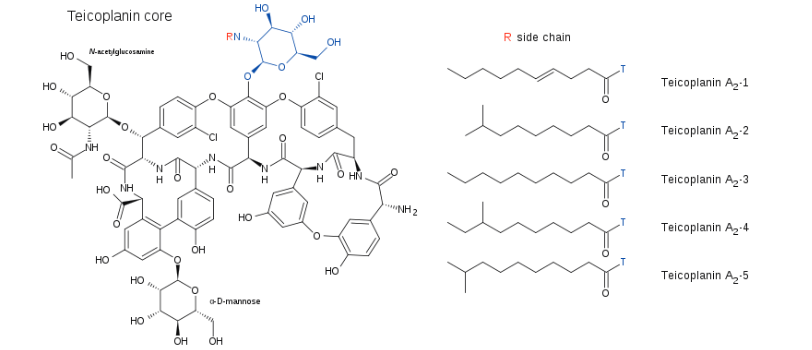
What are the problems with teicoplanin as an example of glycopeptide antibiotic?
Mixture of 5 compounds, various side chains
Different antibacterial activity
Potential for variable toxicity
Complex quality control
Differences in pharmacokinetics
Relative proportions of each component can vary between manufacturing batches
How do glycopeptides work on gram+ bacteria?
Preventing the formation of the linear glycan (NAG-NAM) strands by transglycosylase. Inhibiting peptide cross linking by transpeptidase
They do this by binding tightly to the terminal D-alanyl-D-alanine of the peptidoglycan by hydrogen bonding
Transglycosylases → cannot polymerise glycan chains
Transpeptidases (PBPs) → cannot cross‑link the chains
Cell wall cannot be extended or strengthened so cell lysis happens
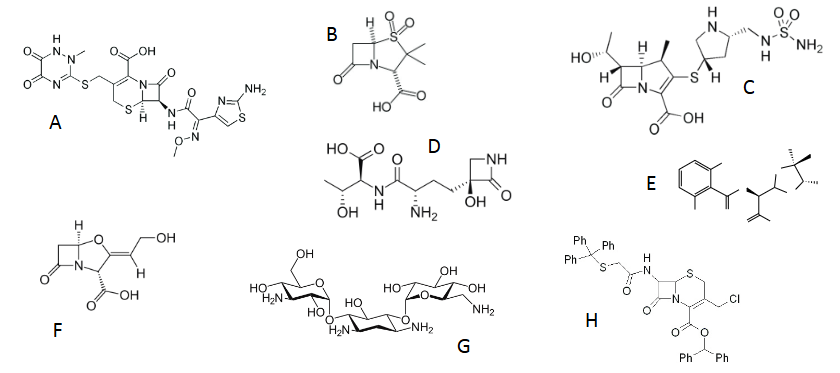
Which of these structures is a carbapenem?
Which of these antibiotics is not a β-lactam antibiotic?
Which of these structures is a cephalosporin?
Which of these structures is a β-lactamase inhibitor?
Which of these structures might be resistant β-lactamase activity?
Which of these structures could be a product of β-lactamase activity on a penicillin?
Which of these might be tolerated by a patient with penicillin sensitivity?
C → carbon and not sulfur on 4,5 ring
B, F and E, G -. they don’t have b-lactam
A, H → 4 membered ring fused to 6
B, F → 4,5 ring system but no side chain, has oxygen or modified sulfur
D,E,G and A,C → they’re late gen penicillins
E → b-lactam ring is open
D,G