WK 4: Energy and Enzymes, Oxidation and Reduction
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
Distinguish between metabolism, a metabolic pathway and a metabolic reaction.
Metabolism= all of biochemical reactions in an organism
Metabolic Pathway= specific, linked sequence of the biochemical reactions, they are often cyclical or linear converting a precursor into a final product
Metabolic Reaction= a single enzyme catalyzed step within a pathway that converts a reactant to a product
Define exergonic and endergonic reactions
Exergonic (energy out)= a chemical reaction that release free energy to their surroundings
Endergonic (energy in)= a chemical reaction that requires an input of energy to proceed
What is activation energy? What is a catalyst?
Activation Energy= the energy “hill” that reactant molecules must overcome to break existing bonds and form new ones
Catalyst= a substance that speeds up a reaction without being consumed by lowering this activation energy (typically providing an alternative reaction pathway)
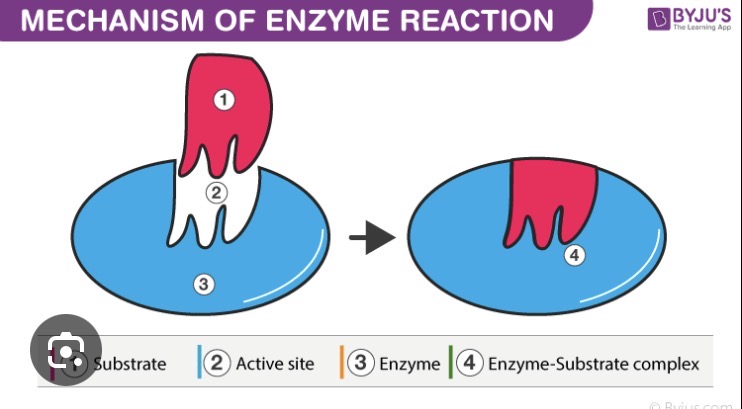
Sketch an enzyme, its substrates and products and illustrate the events that occur over the course of a reaction (binding, catalysis and dissociation). Describe each event.
Describe conformational change and how it occurs, as well as how it affects enzymatic reactions.
A conformational change is a structural shift in proteins. It occurs when an enzyme transitions from one three dimensional shape (conformation) to another caused by environmental factors or molecular binding.
It can affect enzymatic reactions because conformational change is fundamental to enzymatic efficiency. It allows enzymes to speed up reactions in many ways.
Design experiments to test the effect of each of the following variables on the rate of an enzyme catalyzed reaction: enzyme concentration, substrate concentration, affinity, temperature and pH. Be prepared to explain and graph expected results.
Describe how cofactors and coenzymes affect enzymes.
Cofactors and coenzymes are essential non-protein helpers that bind to enzymes, changes their shape or connect them with substrates to activate, increase the rate or enable catalysis.
Cofactors (inorganic metal ions)= stabilize structures
Coenzymes (organic vitamin derivatives)= act as transient carriers of chemical groups or electrons
Distinguish between allosteric inhibition and allosteric activation.
Allosteric Inhibition= slows down or stops the enzyme activity by altering the active site, making it unusable
Allosteric Activation= increases enzyme activity, stabilizing the active site to better fit substrate’s
Contrast allosteric regulation and competitive inhibition.
Allosteric Regulation= binds to an Allosteric site (site away from the active site), causes conformational change that increase (activation) or decrease (inhibition) substrate affinity, cannot be overcome by adding more substrate, and frequently involves complex, multi subunit enzymes such as feedback inhibition.
Competitive Inhibition= binds directly to the active site, obstructs the active site preventing substrate binding, can overcome by adding more substrate which than outcompetes the inhibitor, is often drug based and common with drug designs.
Given a metabolic pathway, be able to predict what would happen to all substrates, products, and intermediates if any part of the pathway were blocked.
A blocked metabolic pathway will cause a buildup of the substrate immediately preceding the block, a reduction in products downstream, and various regulatory shifts.
Explain what is meant by oxidation and reduction, and how it relates to hydrogen in biology.
Oxidation= is the loss of electrons (or hydrogen)
Reduction= is the gain of electrons (or hydrogen)
Oxidation and Reduction (redox reaction) relates to hydrogen in biology because these paired electrons drive energy transfer
Describe the role of NAD+ and FADH in cellular respiration.
In cellular respiration they act as the essential electron carriers in cellular respiration, high energy electrons from glycolysis and the citric acid cycle to the electron transport chain.
Act as a coenzyme, once reduced, provide the electrons needed to establish a proton gradient that drives ATP synthesis
Given a redox pair, be able to determine which is the oxidized form and which is the reduced.
How to Determine:
Compare their oxidation numbers (states) Because oxidation involves the loss of electrons (increase in oxidation number), while Reduction involves the gain of electrons (decrease in oxidation number)
Explain the relationship between redox and energy.
Relationship Between Redox and Energy:
Redox (reduction oxidation)= reactions drive energy transfer by shifting electrons from lower electronegativity atoms (high potential energy) to higher electronegativity atoms (low potential energy)
.