ETC and ATP Synthase
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
What is complex 1?
NADH Dehydrogenase
NADH DH is __ __ in the inner membrane
partially embedded
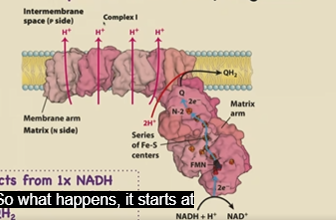
Describe the flow of e- up NADH Dehydrogenase (Complex 1)
2 e- passed into Complex 1 from NADH
Travels first to FMN, then a series of Fe-S clusters, with different redox potentials
2 e- (and 2 H+) passed to CoQ, making CoQH2
The energy from passing the 2e- to redox centers of higher redox potential provides energy for a physical conformation change that pumps protons into the P side (4 total H+ for the 2 e-)
Products: 4 H+, 1 QH2
What is Complex II called?
Succinate dehydrogenase
Describe the flow of e- through Complex 2
Succinate deposits 2 e- into Complex 2
FAD is converted to FADH2
2 e- passed to a series of Fe-S clusters of increasing redox potential, FAD regenerated
2 e- passed to another CoQ
NO H+ pumped out!!
Products: 1 reduced CoQ (QH2)
What are the products of complex 3 reaction?
2 reduced Cyt C, 1 Q, 4 H+ pumped to P side
Complex 3 is a…
Ub/CytC Oxidoreductase dimer
How many electrons does Complex 3 transfer to each CytC?
Only ONE!!
What is stage 1 of the Q Cycle?
QH2 travels through the inner membrane and reaches Complex 3
The 2 e- go different ways
One e- goes to an Fe-S cluster, then to Cyt C1, then finally to a CytC
The two Hs from this original QH2, that got stripped of its e-, now releases 2 H+ into the P side. This regenerates Q.
The other e- goes to a new Q that just entered, reducing it to its semireduced form.
What is stage 2 of the Q Cycle?
**A second QH2 arrives
Same e- thing: one goes “up” and is put onto CytC. 2 H+ released form QH2 to P side
The other goes “down”, but THIS TIME, it’s transferred to that Q radical we made, to make fully reduced QH2. This requires 2 H+. The QH2 leaves Complex 3
What is the net reaction for the Q Cycle?
QH2 + 2 CytC (oxidized) + 2HN+ → Q + 2 Cyt C (reduced) + 4HP+
Stage 1 of the Q cycle uses __ QH2. Stage 2 uses __ QH2 and produces __ QH2.
1, 1, 1
How is Complex 3 different in its proton “pumping” compared to Complex I?
It’s not actually pumping H+ from the N to P, it’s releasing 2 H+ from each QH2 that enters
Rember, Cyt C has what essential prosthetic group?
Heme
How many e- do we need to reduce one O2?
4 e-
In the Complex IV reaction, what’s the dealio with the protons?
4 H+ picked up from matrix to make 2 water molecules
4 additional H+ pumped from N side to P side
Complex IV passes e- to…
O2
Describe the reaction occurring in complex IV
4 CytC passes 4 e- to complex
4 e- passed to O2 molecule
4 H+ come from matrix into complex to help make the 2 water molecules
4 H+ are also pumped completely from N to P side
Complex 4 has a ___ reaction center…
Bimetallic
Fe-Cu reaction center
Binds to the O2 molecule
Why do we need 4 e- to reduce O2 to 2 H2O?
Because of the reduction of metals in the bimetallic reaction center of Complex IV
Where do the 10 pumped H+s come from if you start with ONE NADH?
4 from Complex I → missing if you start with succinate
4 from Complex III
2 from Complex IV → (from two Cyt C)
The other 2 H+ from Complex 4 require the other two Cyt C from the second QH2 that entered
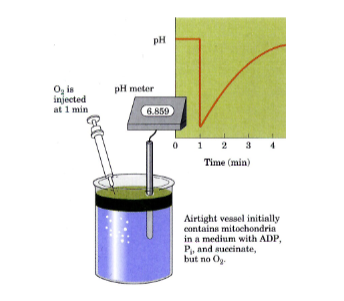
What experiment showed that e- transfer causes H+ flux
Put a bunch of mito, succinate, ATP and ADP in beaker. pH is constant. Add in O2, there’s a sudden drop in pH (pumping of H+), and then gradual rise in pH (H+ being consumed by ATP synthase)
What two small molecules can destroy the H+ gradient and thus block ATP synthesis?
What’s another molecule that can block ATP synthesis, and which complex does it block?
DNP and FCCP destroy the H+ gradient, by picking up H+s and depositing them into the matrix. They are proton shuttlers and thus destroy the gradient.
They’re able to do this because can pass through the inner membrane
Can also use cyanide → blocks complex IV. If e- are not passed to O2, the gradient formation can not continue.
Cyanide blocks…
e- transfer between complex IV and O2
Basically irreversible
What two molecules stall ATP synthase? What else happens?
Oligomycin, venturicidin
O2 reduction will stop eventually →too much accumulation of H+ gradient in P side
Why does O2 consumed shoot up when DNP is added
High proton gradient is destroyed, but still no ATP is produced
Describe the artificial gradient experiment
If you put mito into a high [H+] (nothing else), ATP can be synthesized for a bit, but then H+ accumulates in the matrix, and now there’s too much. So, ATP synthesis will stop!
If you want to see if it really is the H+ gradient causing ATP synthesis, you’d need to shuffle K+ ions OUT (suing valinomycin) so that the gradient can remain. ATP synthesis should continue
ATP Synthase has two parts…
Fo: integral membrane part, oligomycin sensitive
F1: “Fraction 1”, soluble, facing N side (matrix)
Subunit C function…
Connected to gamma blade
Rotates as H+ passes through from P to N side, as does gamma blade
Drives ATP synthesis
What subunits of ATP synthase are fixed in space?
A, B2, alpha beta
AKA everything besides c and gamma rod
ATP synthase dimer structure facilitates…
Mitochondrial cristae formation → maximizes surface area in the matrix, to maximize ATP synthase presence
When ATP Synthase folds, its 110o
The alpha and beta subunits form…
A hexomeric ring complex (distinct from C ring!!! C ring actually moves itself), because there are 3 alpha and 3 beta subunits
They alternate

B subunit binds…
ADP or ATP
There are 3 states for the B subunits of F1:
B empty
ADP bound
ATP being made
Each B must have a different state!!
Describe the Binding-change model of the B subunits of ATP synthase
Each of the 3 B subunits has a different form
The gamma blade rotates from the B subunit that just made an ATP to the one about to make ATP. Whatever B subunit that it’s pointed to, it kicks out the ATP from the subunit
Needs 3 H+ for every 120o turn
The ring rotates in what direction?
Counter clockwise
The ring has __ half channels. One faces down towards the __ side, the other faces the __ side.
2, P, N
Acts as entry point from P side
Remember, the C ring sits in the __ membrane
inner
Proton gradients can also drive __ movement
Flagella
alpha/ beta ring faces __ side. C ring (moves) faces __ side.
N (matrix)
P (intermembrane space)
Why do we need 4 H+ to make 1 ATP using ATP synthase?
Because the gamma rod uses 3 H+ per turn, and then 1 H+ is needed to come in with the Pi when making ATP

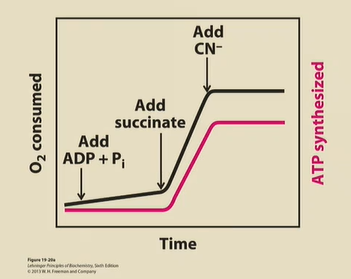
Draw how a graph of O2 consumed and ATP produced will look if you start with…
1) Just mitochondria, ADP and Pi
2) Add succinate
3) Add CN-
1) ATP not really produced → no NADH or FADH2
2) Krebs cycle starts churning, ETC starts pumping e-, so ATP synthesis can happen
3)CN- blocks complex IV, and so the H+ gradient is destroyed. O2 not consumed, ATP not produced.
What two molecules literally halt ATP synthase? What are the effects?
Oligomycin, venturicidin
O2 consumption will drop, ATP production will stop.
O2 drops because the H+ accumulates so much in the inter membrane space, and pumping won’t continue
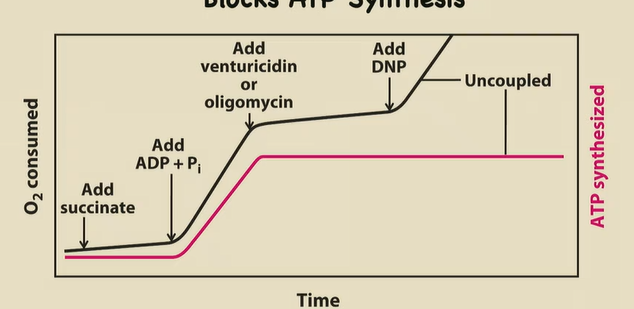
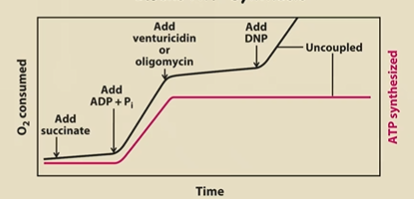
Describe what an O2 consumed and ATP produced graph will look like when…
1) Mito, succinate, ADP and Pi are together
2) Add venturicidin or oligomycin
3) Add DNP
1) Krebs churning, ERC working, ATP synthase working (O2 consumed and ATP shoot up)
2) These block ATP Synthase. So a proton gradient builds up, ATP production STOPS, and ETC works but very minimally (O2 consumption increasing but slowly)
3) This is a proton shuttler → levels out the gradient from being super congested in the P side. Allows O2 consumption to spike, but ATP is still not being produced.