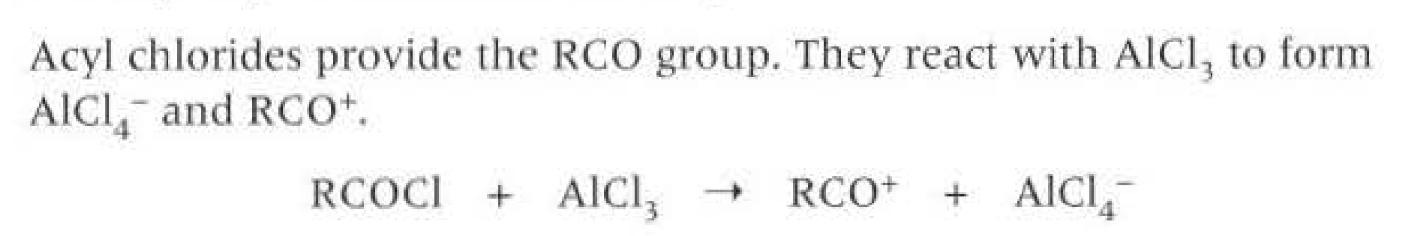Yr 2 CH27 Organic, Aromatic chemistry:
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
What are Arenes?
They are compounds that contain the Benzene ring (C6H6)
The structure behind Benzene and the evidence behind that:
In summary: The bond lengths, the enthalpy of hydration, the addition reactions
If carbon, double bond and single bond are different in length, the bonding between the carbon bonds in benzene are all the same length. So, the bonds between carbon have intermediate, between the length of a single bond and a double bond
Each carbon bond forms 3 bonds, 2 with carbon and one with hydrogen - but the fourth electron for each carbon atom is in its p-orbital, and forms region of delocaised electron ring above and below the benzene ring, making it stable.
The enthalpy of hydration for cyclohexene is -120kj/mol and the enthalpy of hydration for hypothetical benzene (with 3 carbon- carbon double bonds) is -360kj/mol, however for acutal benzene it is -208kj/mol [ARE NOT EXPECTED TO KNOW EXACT VALUES, BUT EXPLAIN THE IDEAS]
The reason being that the benzene is stable, so more energy required to break these bonds, so less exothermic reaction
Similarly, when reacting benzene with bromine water (should expect to go colourless readily), however remained orange, this is because, with the carbon forming a bond with bromine, this breaks the delocalised electron cloud, disrupting the stability
Idea behind the physical properties of Arenes (Benzene): MP and BP
Benzene has a higher MP and BP, because this is a flat molecule, they are able to fit together more easily so more energy is required to separate the molecules.
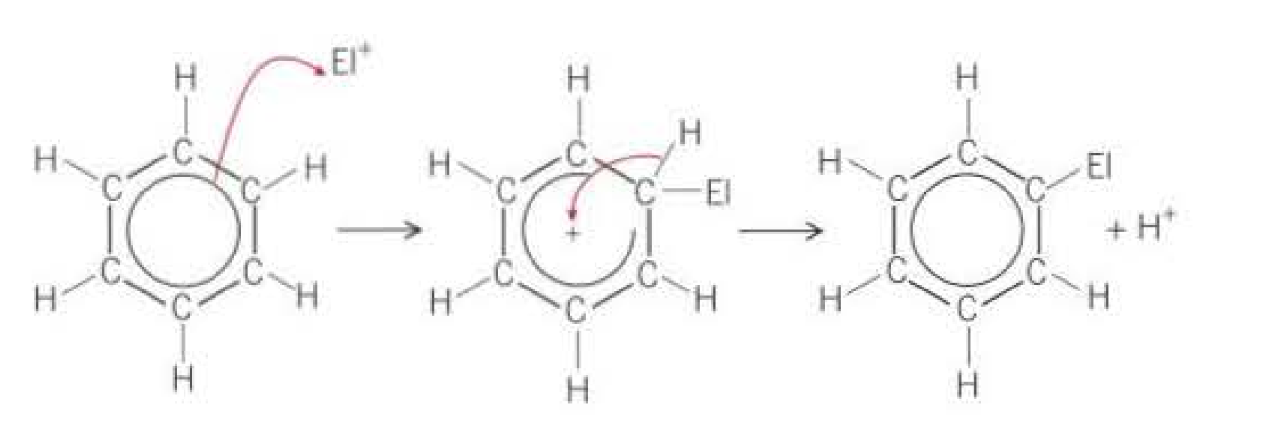
What is the most common reaction mechanism that molecules containing benzene contains?
Electrophillic substitution
Generating electrophiles: (nitrogen one)
Conditions: Concentrated sulfuric acid and Concentrated nitric acid
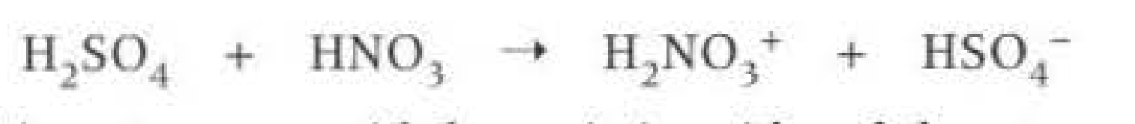

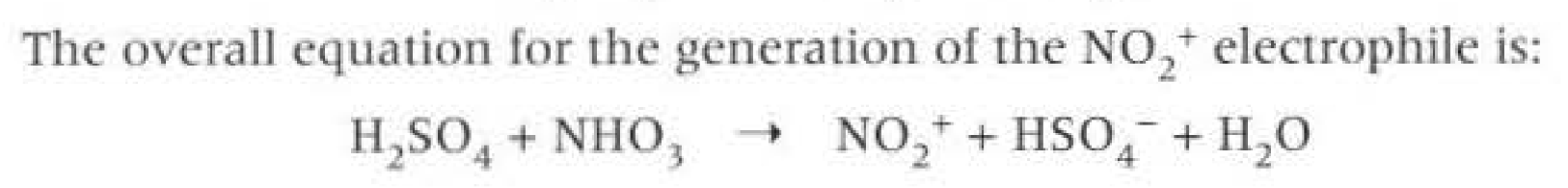
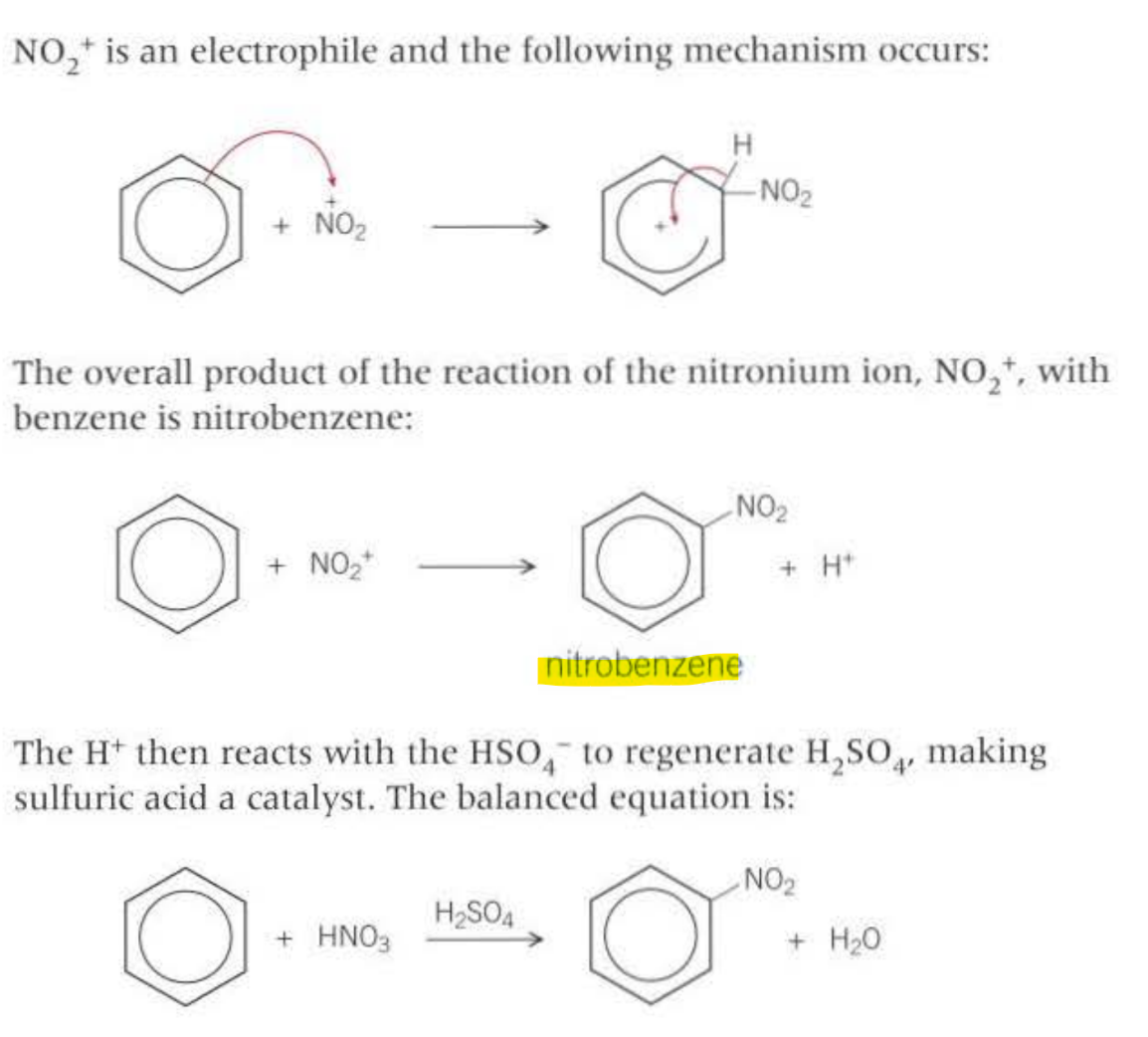
Generating electrophile (to form a ketone benzene)