Orgo 1 Reactions
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
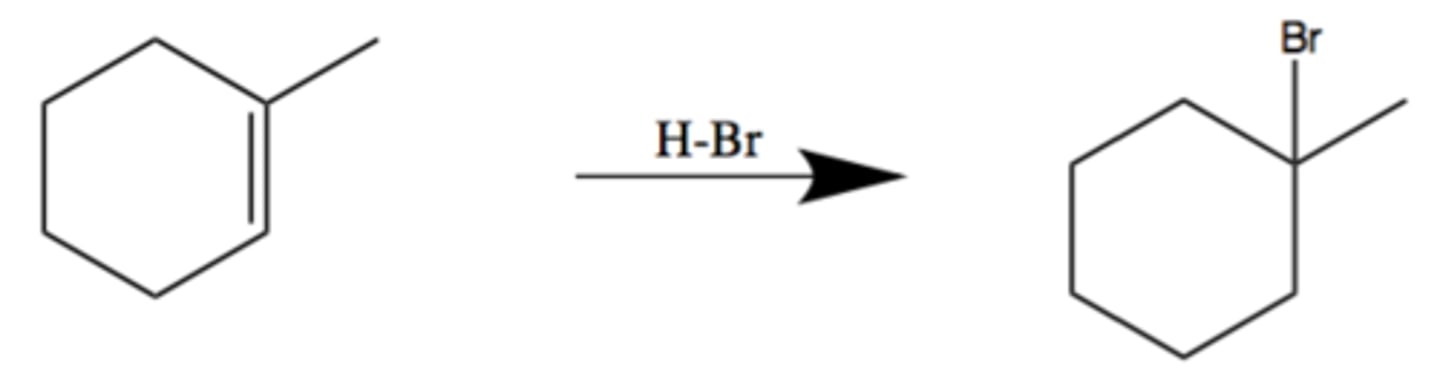
HBr
adds halide (Br) mark addition
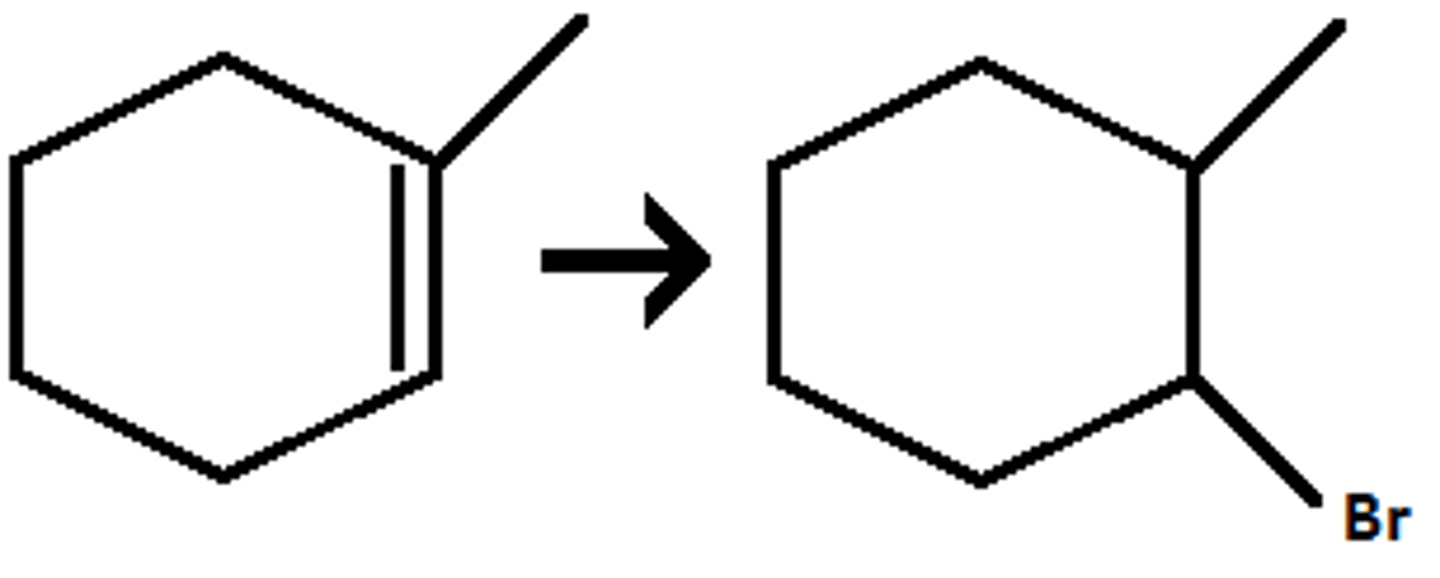
HBr, ROOR
Adds halide (Br) anti-mark
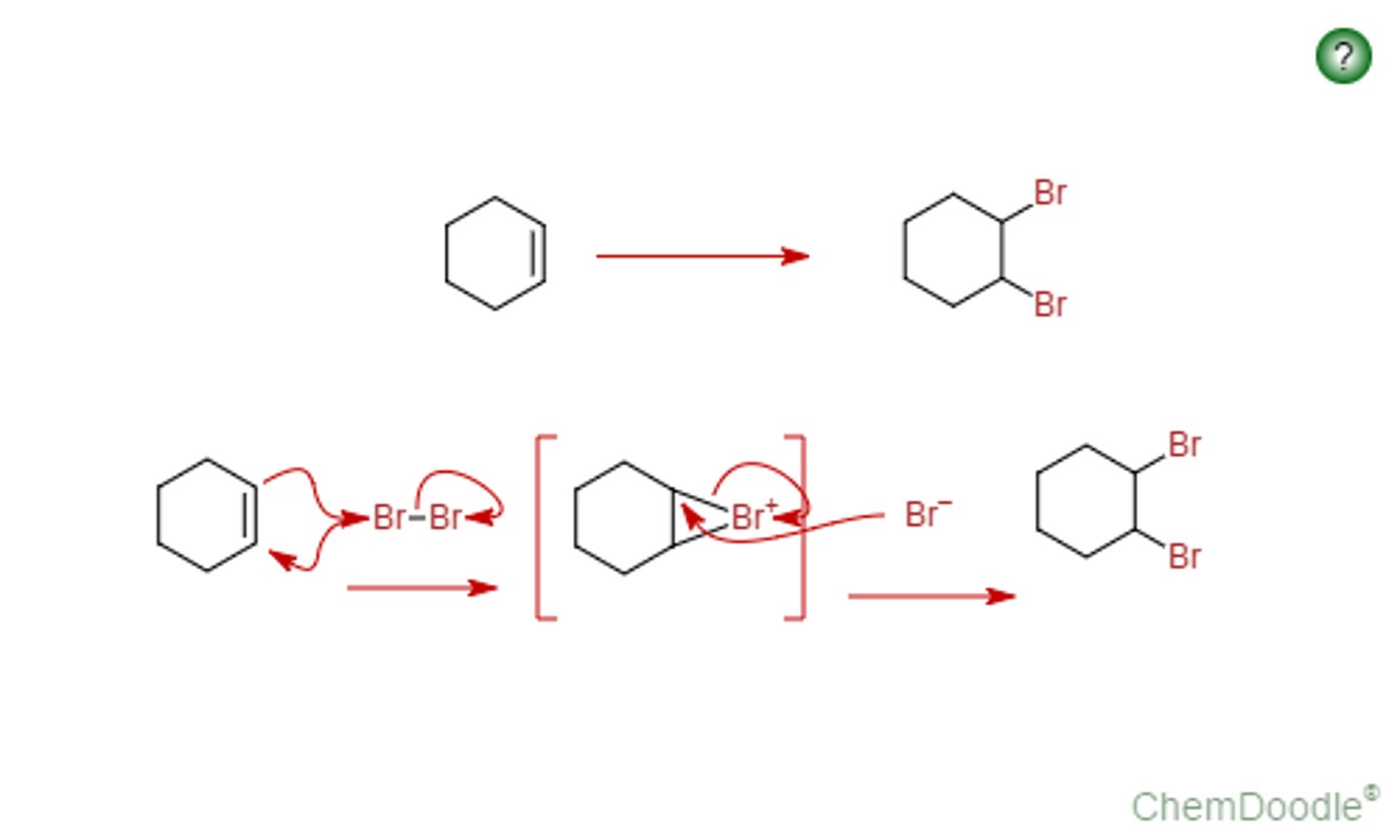
Br2, CH2CL2
Adds 2 Br's on both sides of double bond (anti)
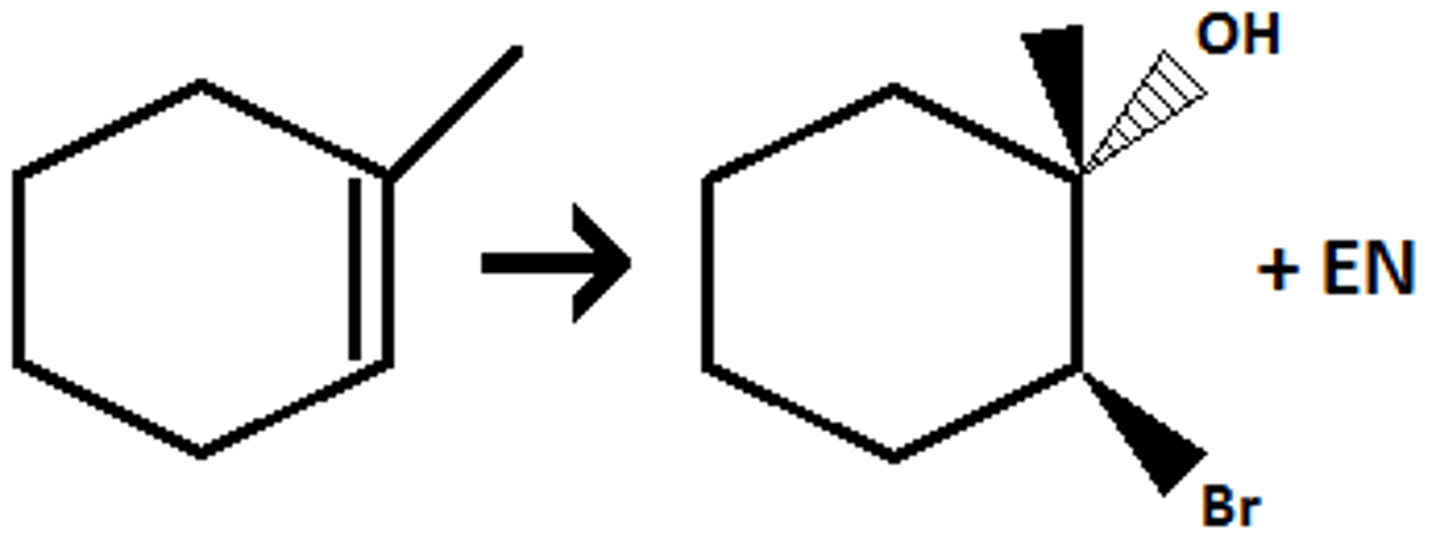
Br2/ H2O
Adds a Br and OH (Mark addition of OH, Br is the H)
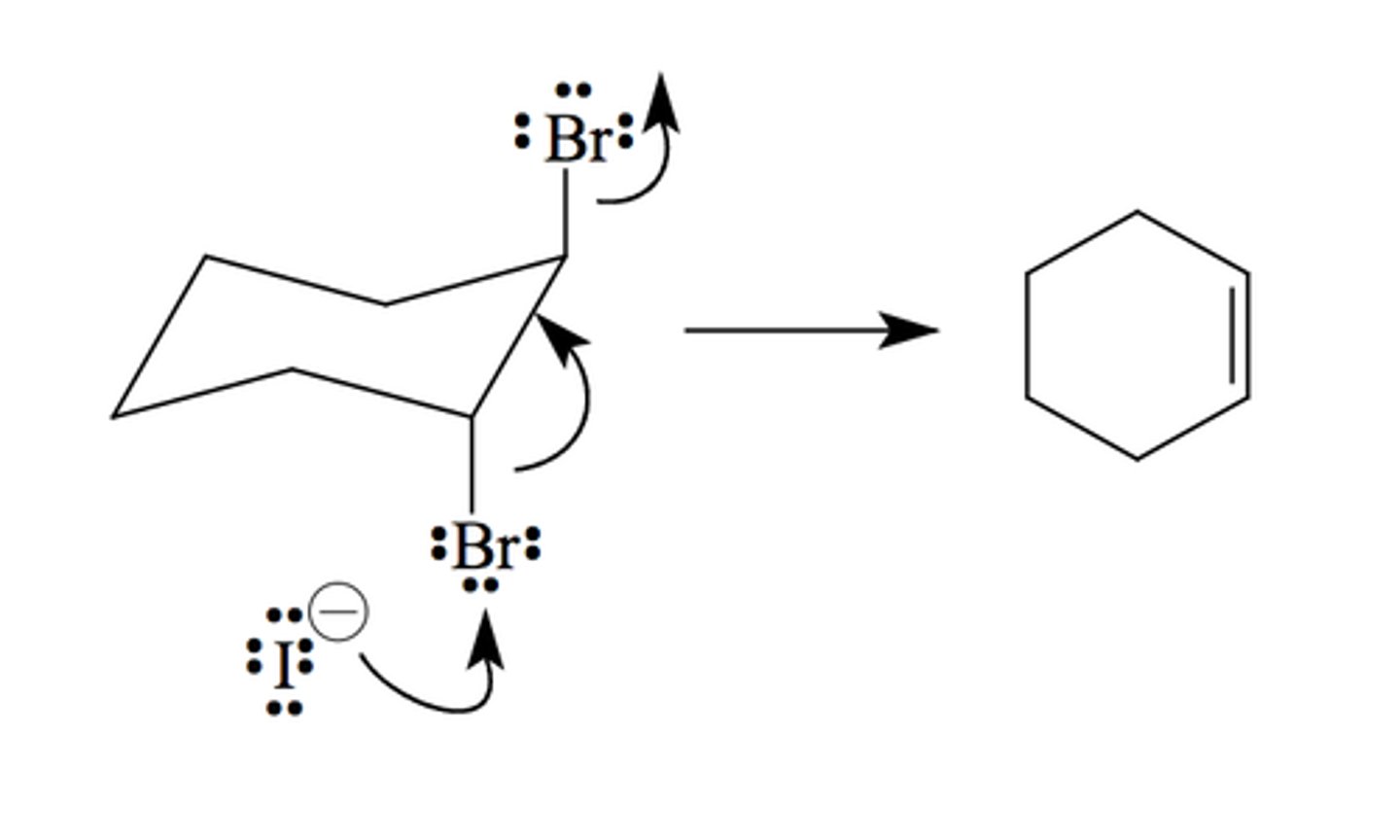
NaI or KI/ acetone
Gets rid of leaving group to form a double bond (alkene)
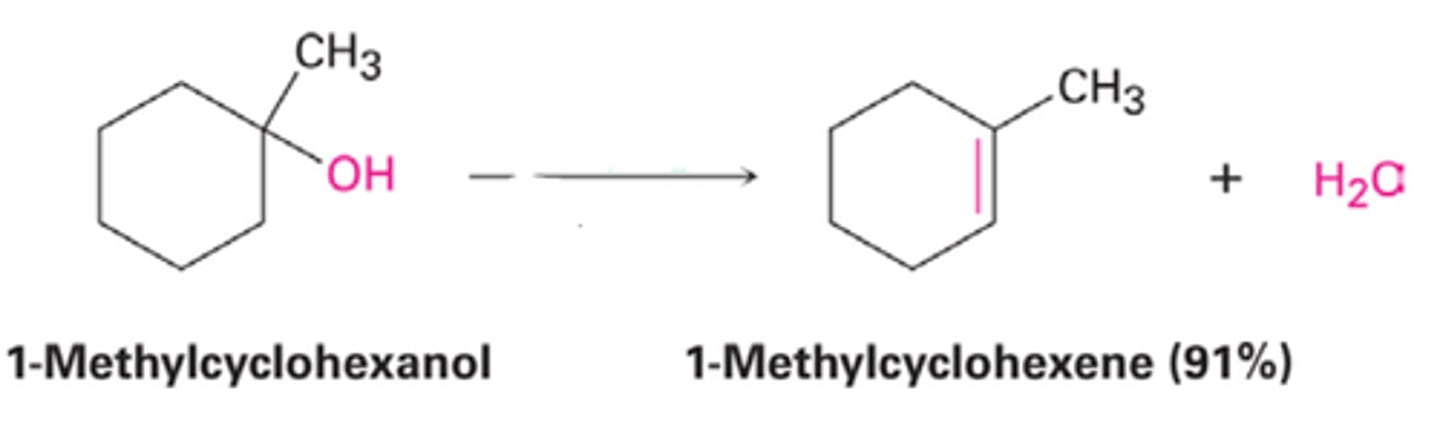
H2SO4/heat
removes OH, forms double bond (inside only, no terminal alkene)
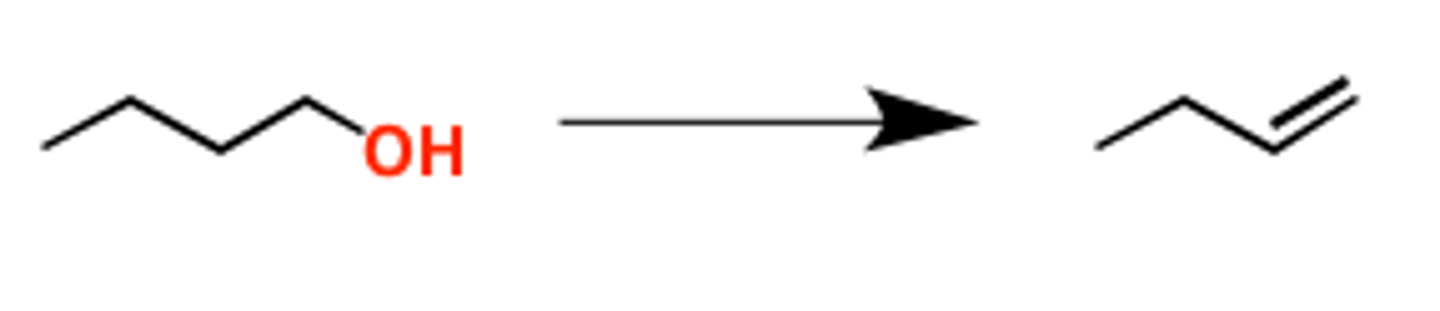
POCL3/heat
removes OH, forms double bond (terminal)
H3O+
adds OH, mark
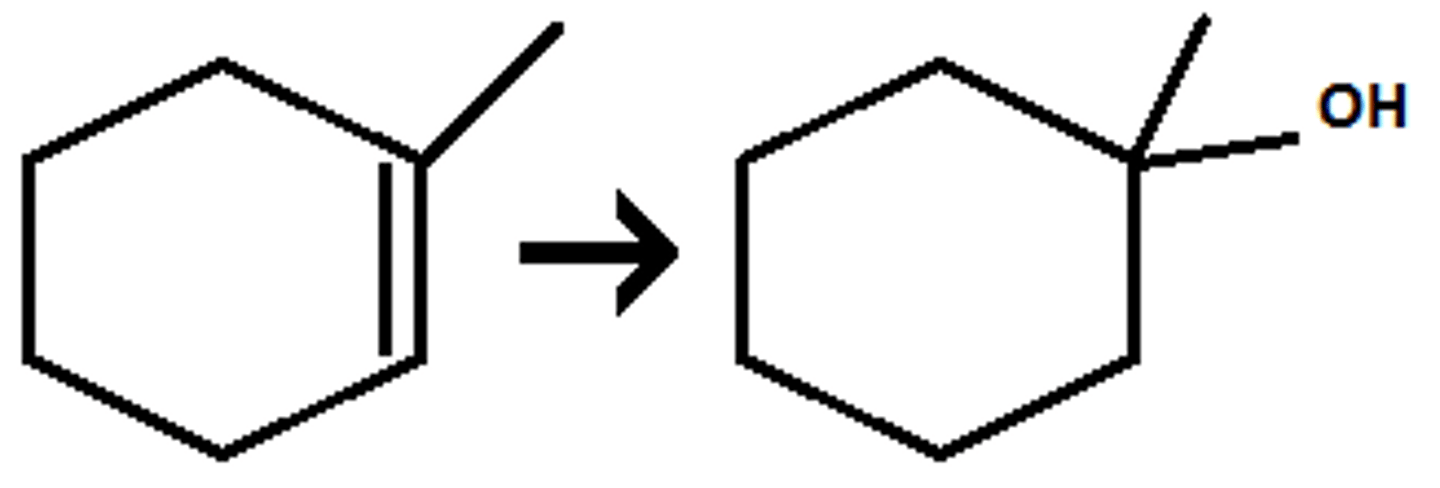
Hg(OAc)2, H2O, NaBH4
Mark addition of OH
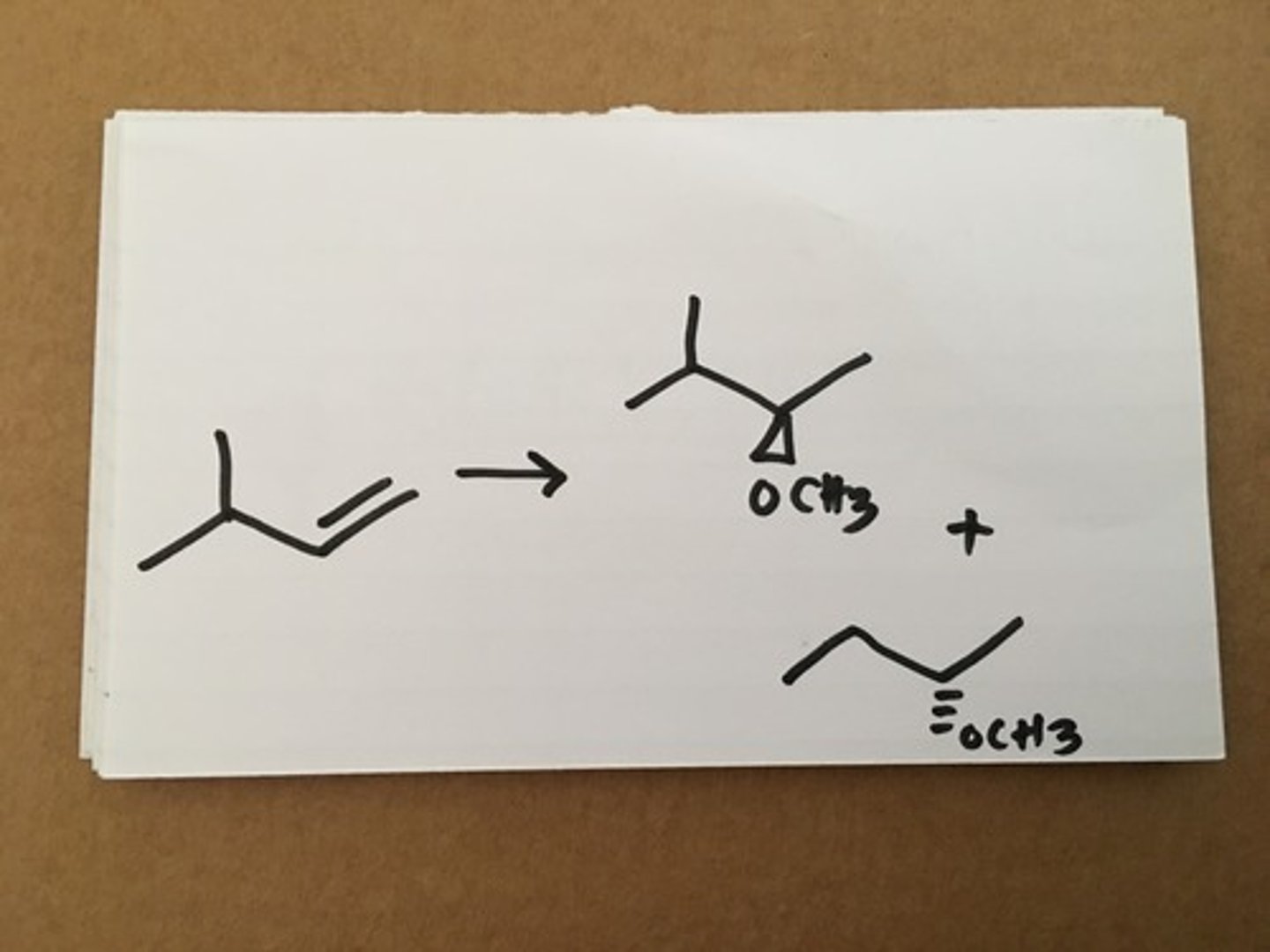
Hg(OAc)2, CH3OH, NaBH4
Mark addition of O-CH3
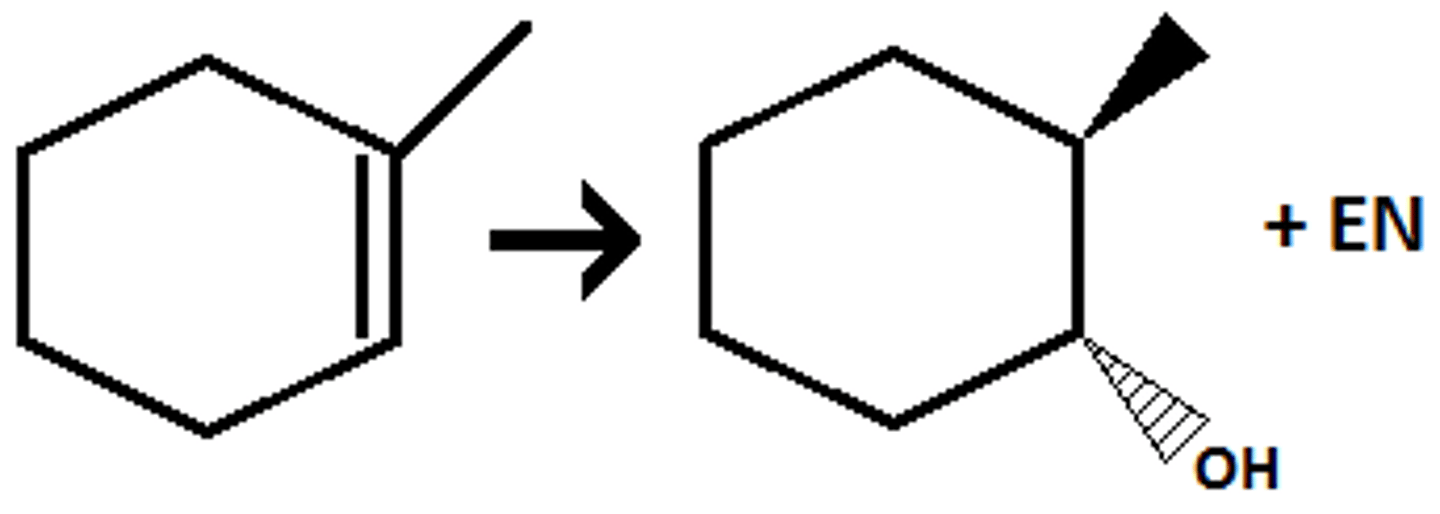
BH3/THF, H2O2/OH
anti mark addition of OH
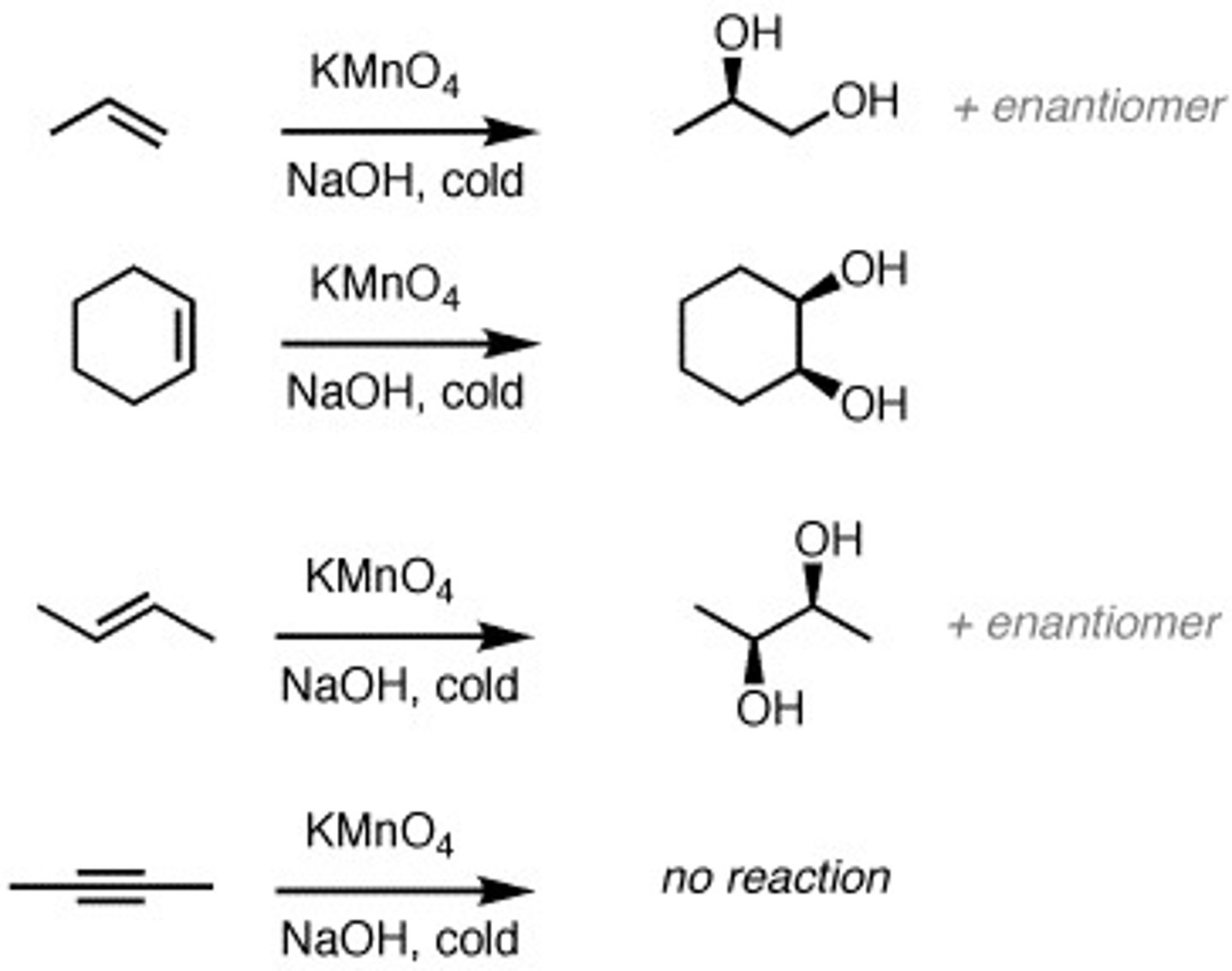
H2/ Pt, Pd, or Ni
turns double bond to single bond, syn addition of H
OsO4/ H2O2
Adds OH to both sides of double bond (syn)

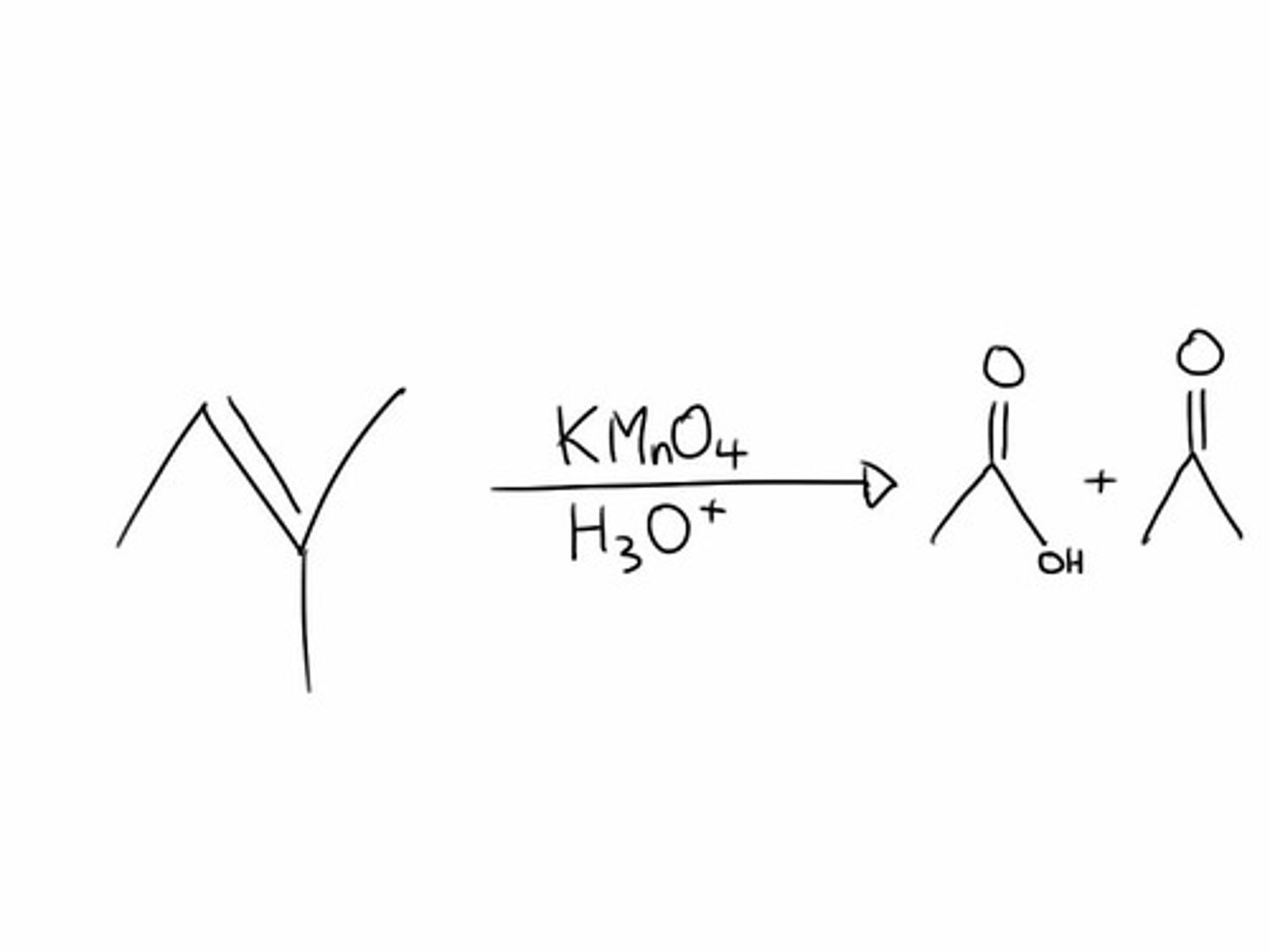
KMnO4/ cold
Adds OH to both sides of double bond (syn)
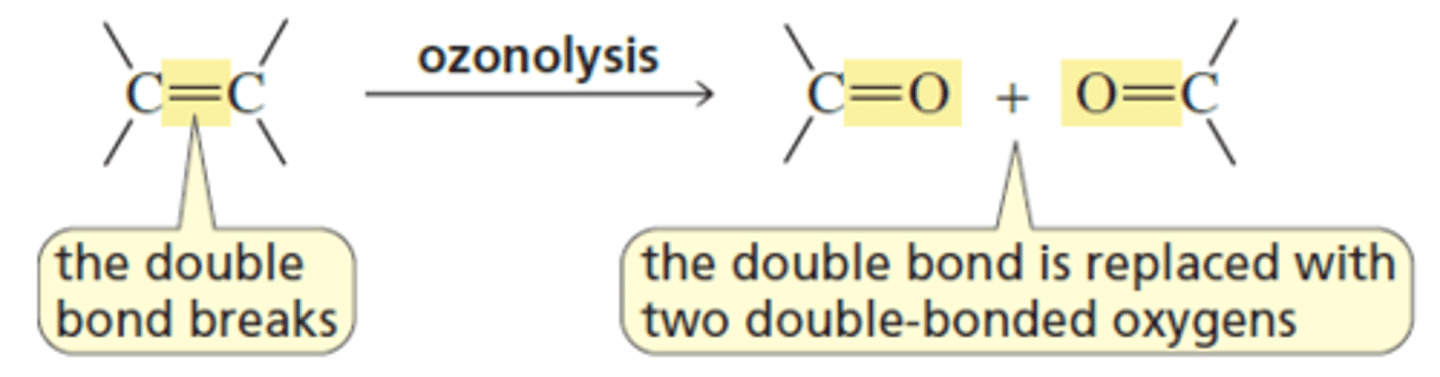
O3/ CH2CL2, (CH3)2S
Double bond cleavage
O=R-R
O=R-H
O=H-H
KMnO4, warm
Double bond cleavage
O=R-R
O=R-OH
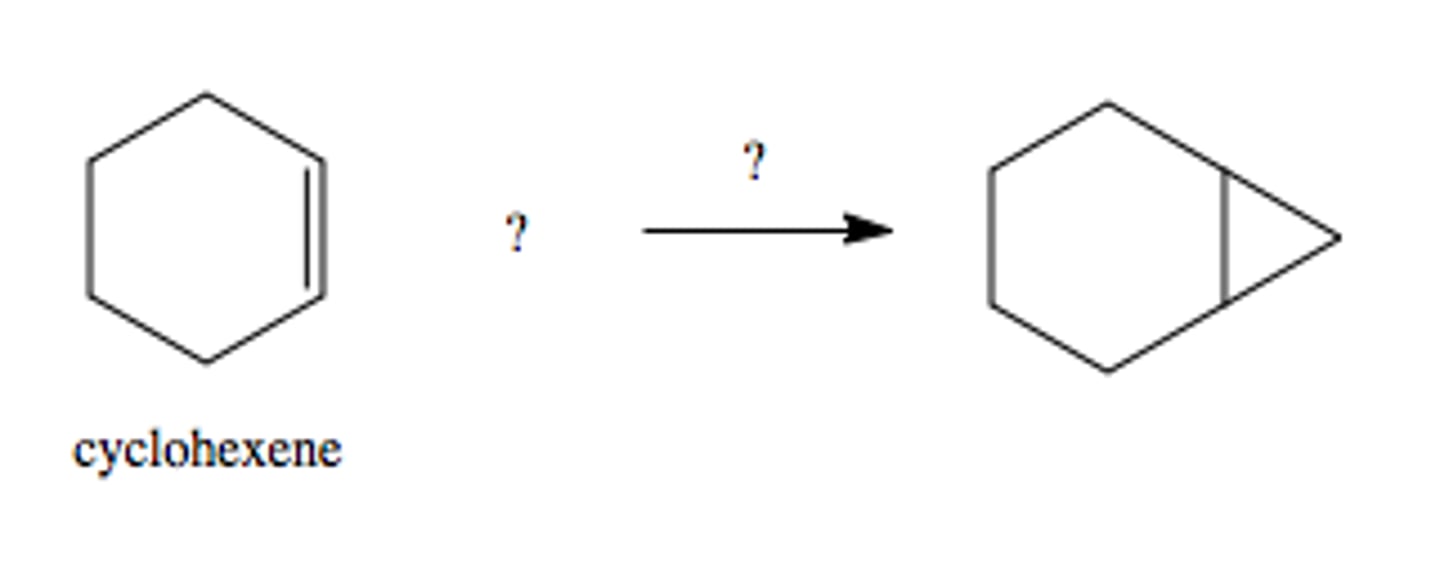
CH2N2, heat
forms cyclopropane from double bond
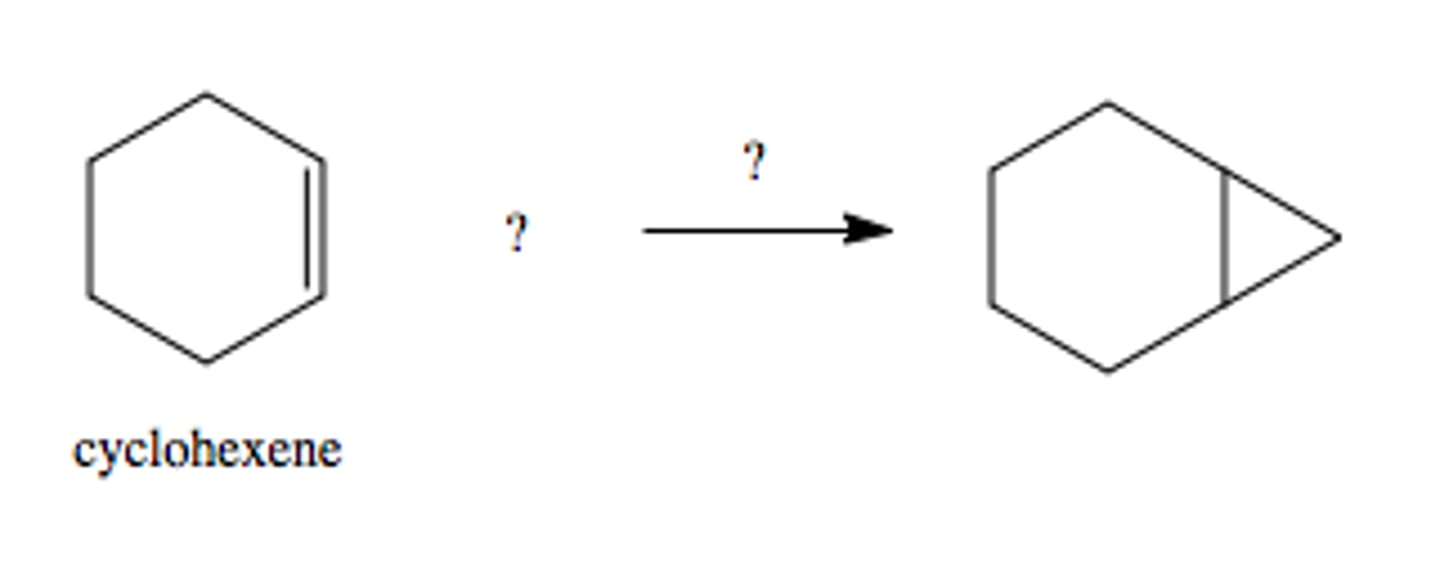
CH2I2, Zn(Cu)
forms cyclopropane from double bond
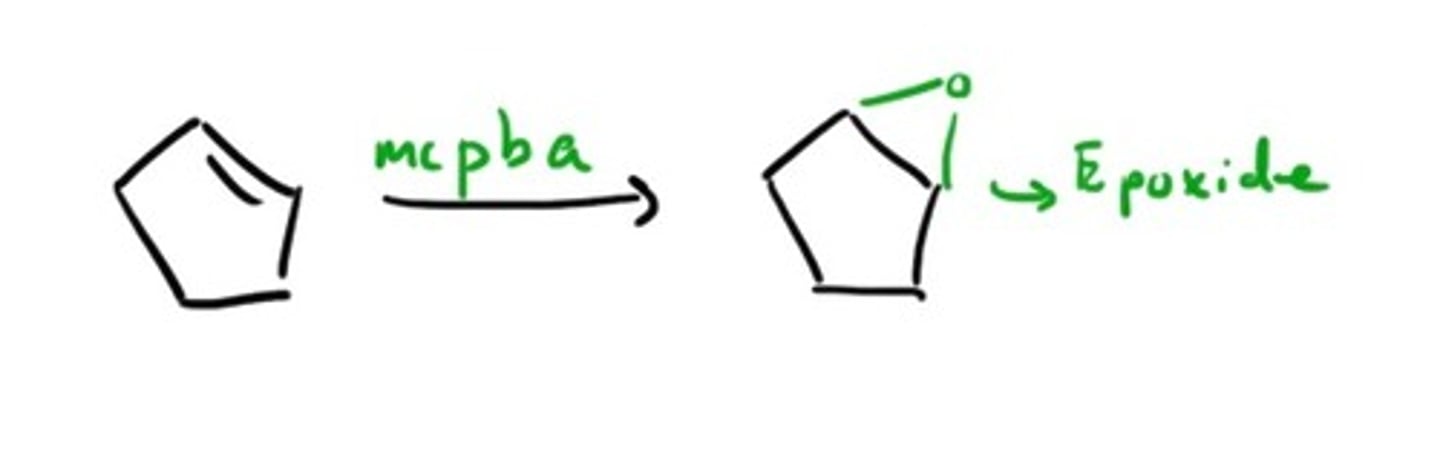
MCPBA
forms epoxides from double bond
H30+, H20
opens up epoxides to two OH groups (wedge and dash)
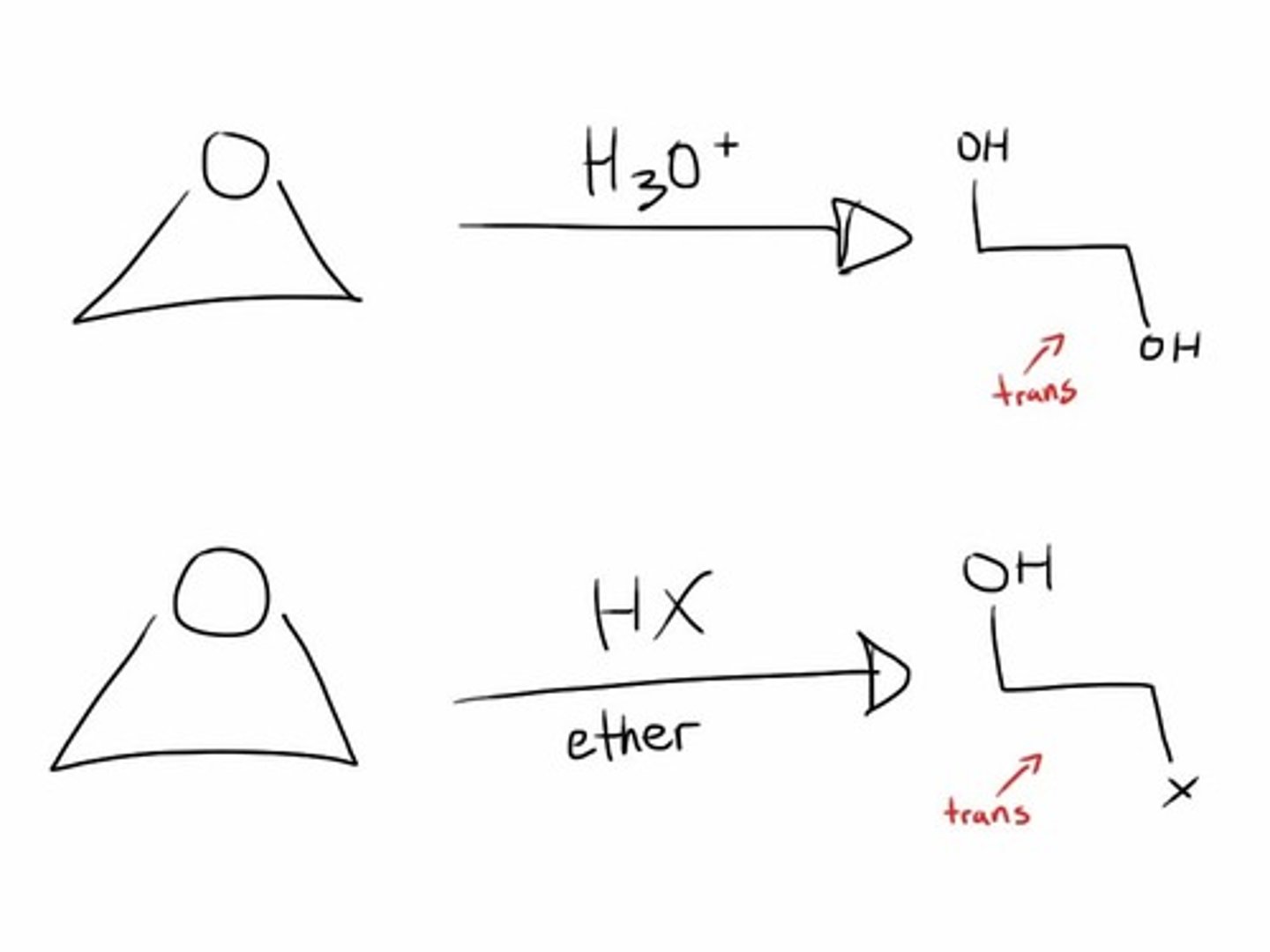
OH, H3O+
opens up epoxides to two OH groups (wedge and dash)
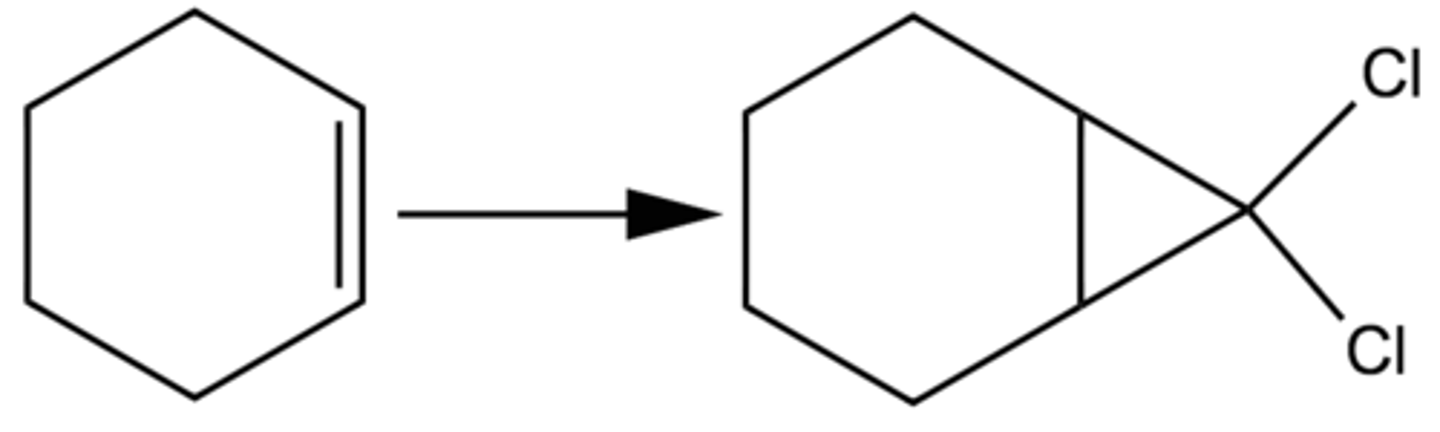
CHCL3, KOH
double bond forms triangle, two chlorines added to tip of it
CHBR3, KOH
double bond forms triangle, two bromines added to tip of it
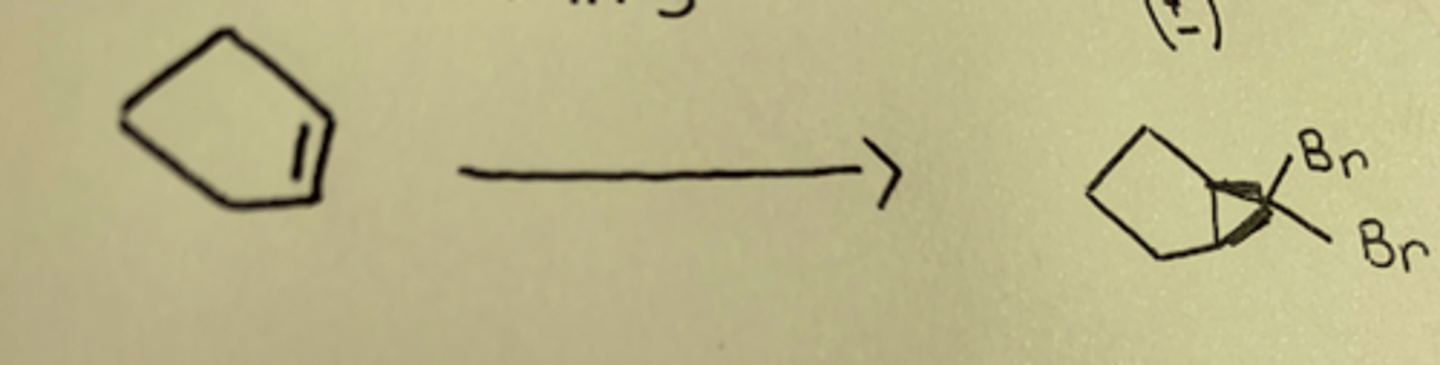
NaNH2
removes H, forms anion
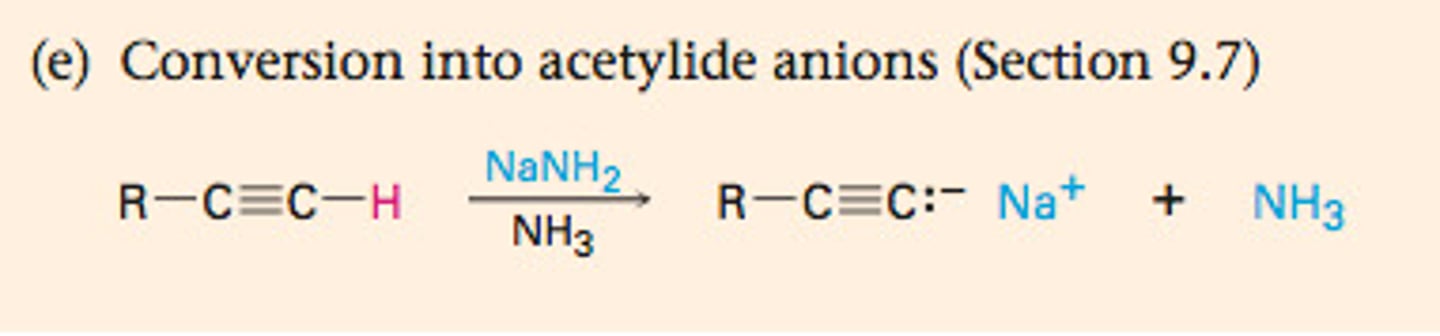
CH3Br
adds CH3 to anion
NaNH2/ H3O+
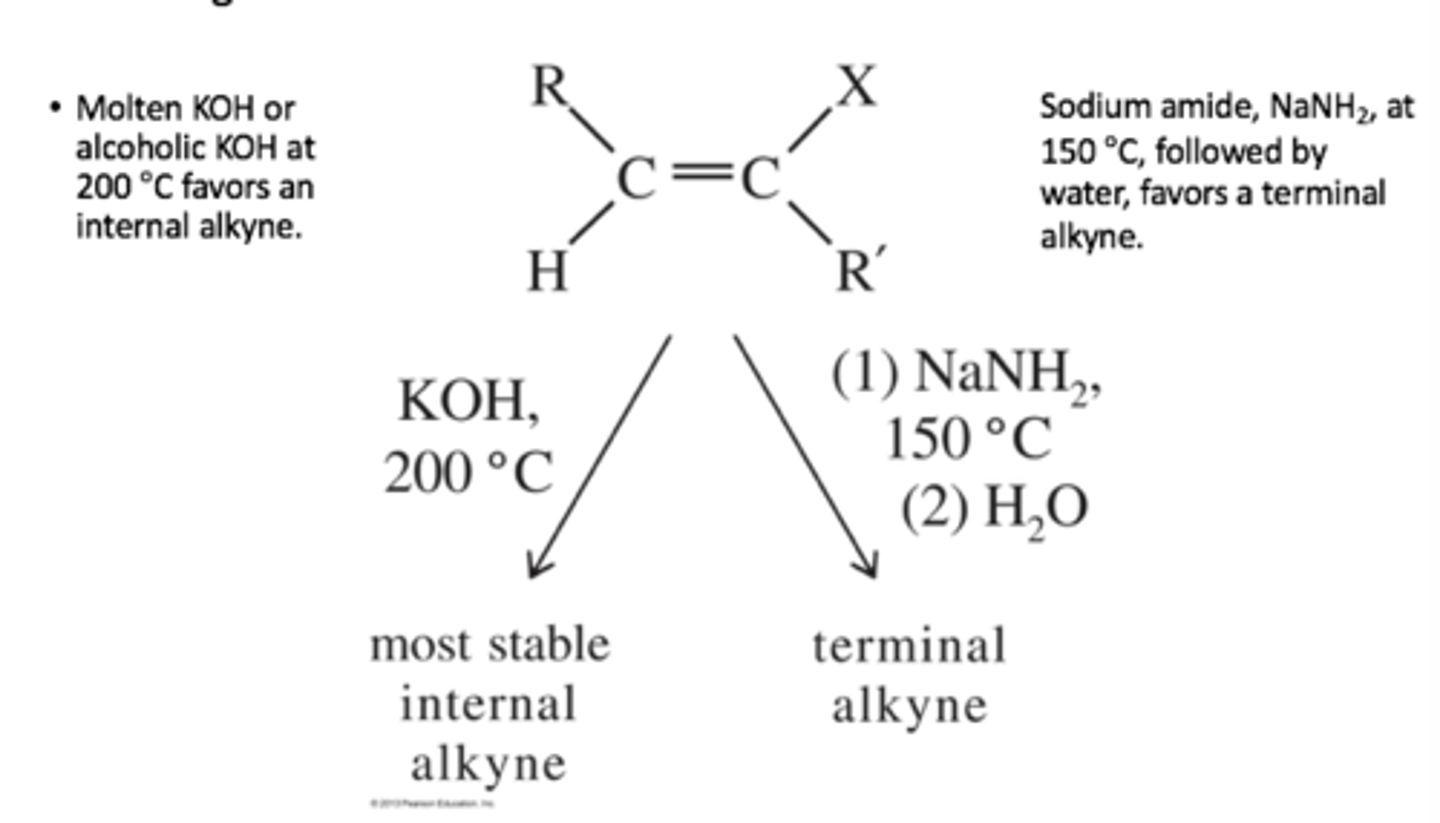
Removes Br (or other halide) groups to form a triple bond,
NaNH2 gives terminal alkyne
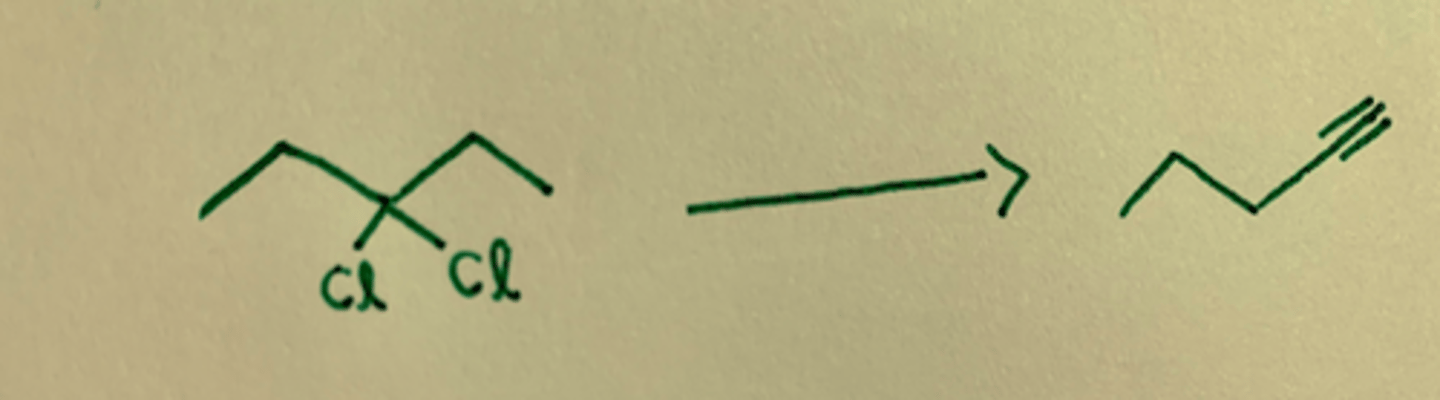
KOH/200 C
Removes Br (or other halide) groups to form a triple bond,
KOH gives internal alkyne
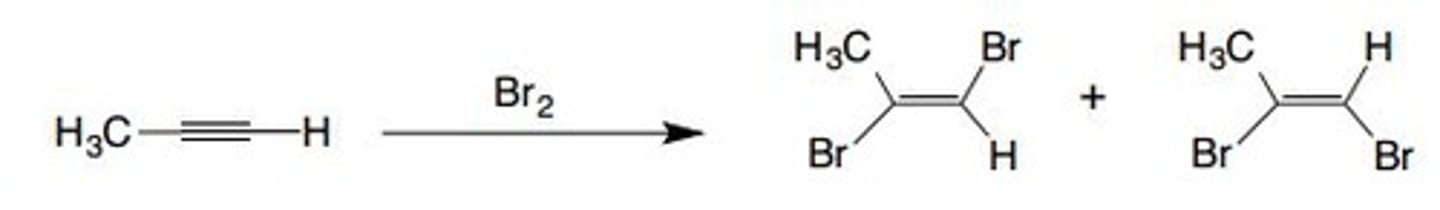
Br2 (1 eq)
adds two Br groups to triple bond, forms double bond, one cis one trans
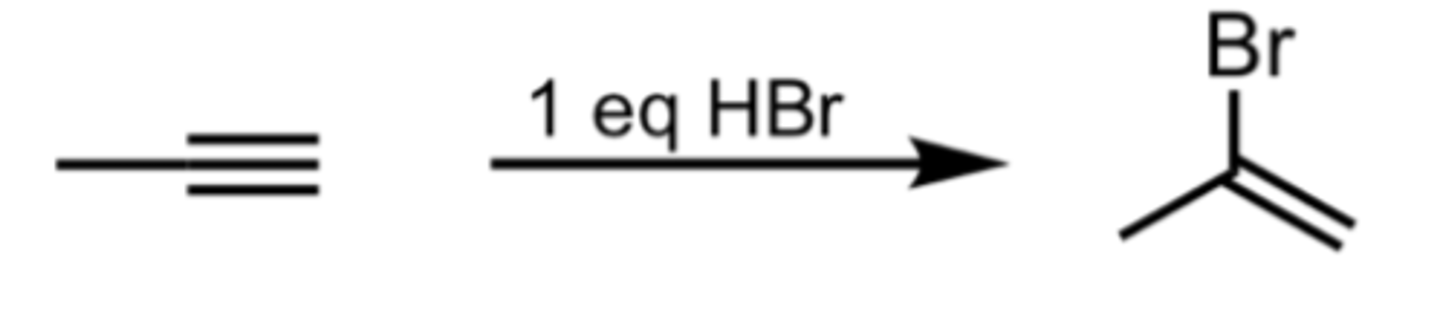
HBr (1 eq)
mark syn addition of Br to triple bond, forms double bond
HBr (2 eq)
mark syn addition of 2 Br
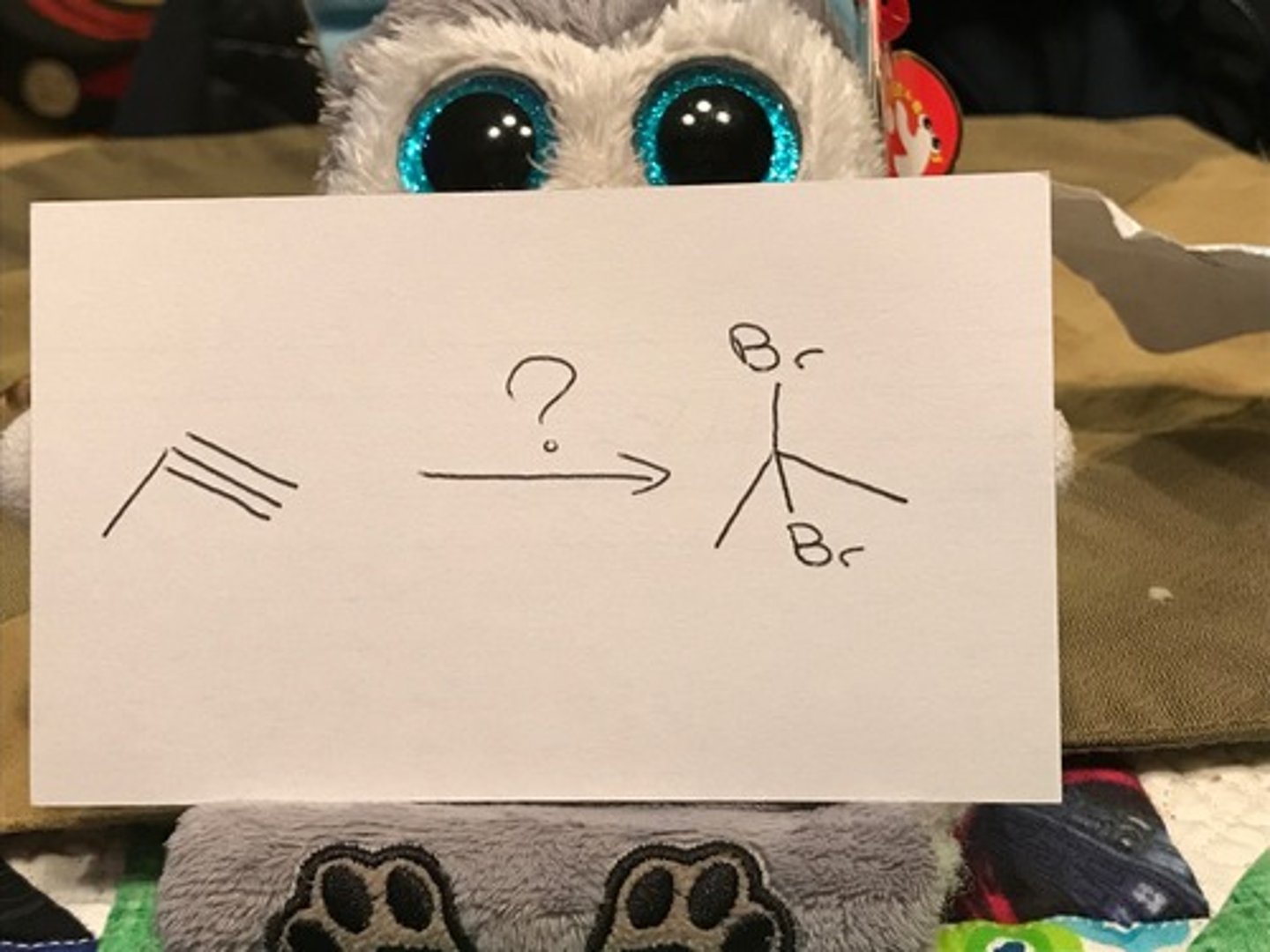
HBr, ROOR (alkynes)
anti mark syn addition of Br on triple bond, forms double bond
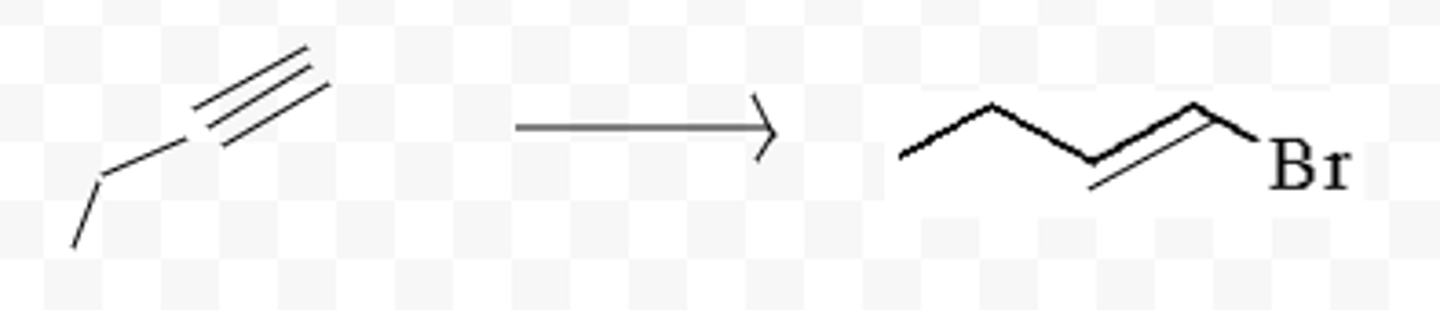
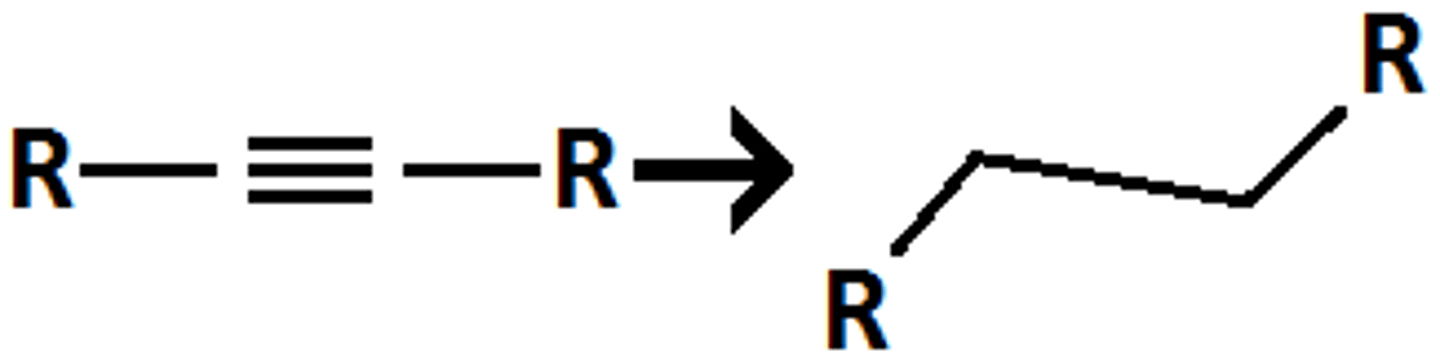
H2, Pt/Pd/ or Ni
turns triple bond into single bond
H2/Pd (BaSO4), quinoline
triple to double bond with syn addition of H
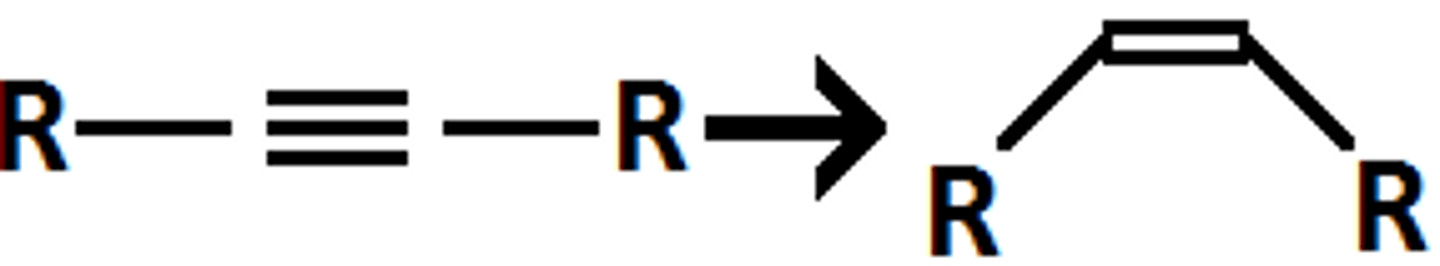
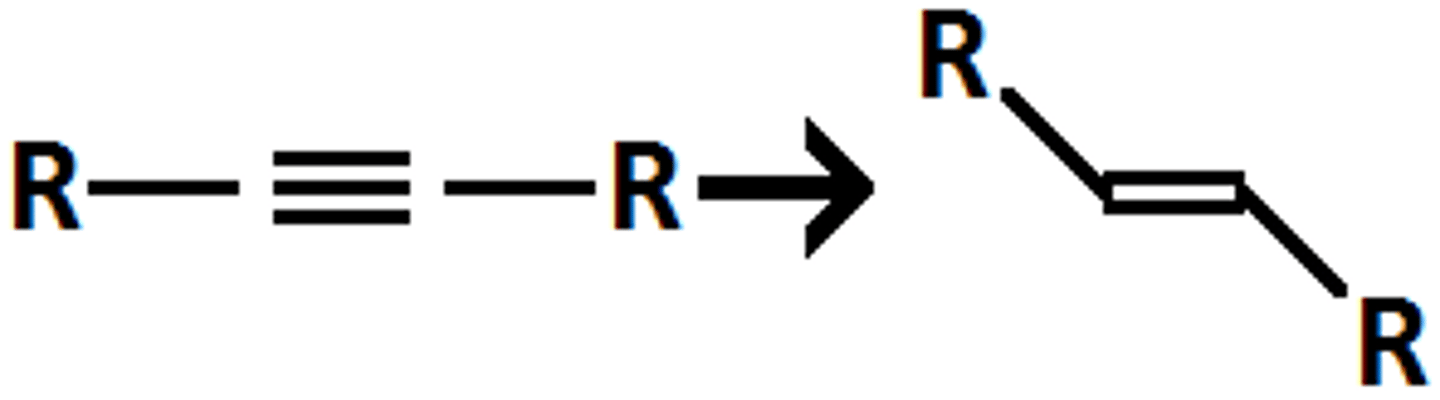
NaNH3
triple to double bond with anti addition of H
HgSO4/H20
addition of H-OH to triple bonds
formation of Ketone O=C-C
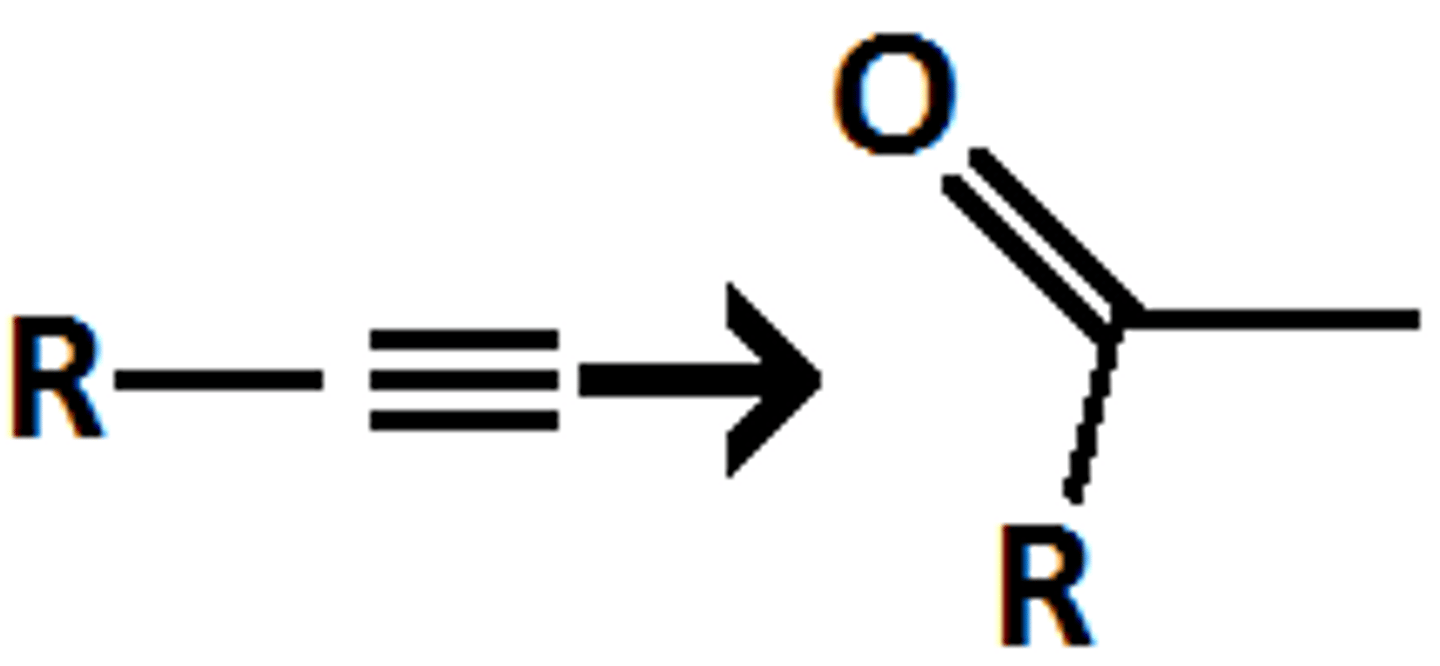
Sia2BH, H202/OH
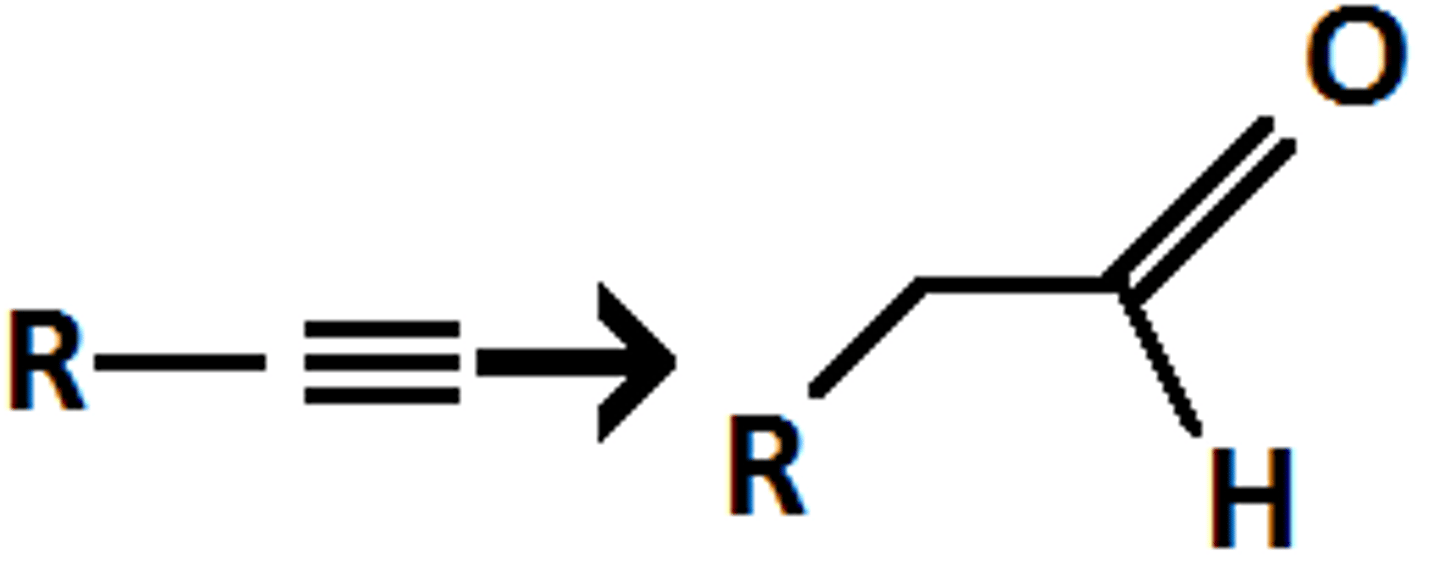
antimark addition of H-OH to triple bonds
formation of aldehyde O=C-H
KMnO4/ H2O
neutral, cold
oxidizes triple bonds to form carboxylic acid (O=) at triple bond
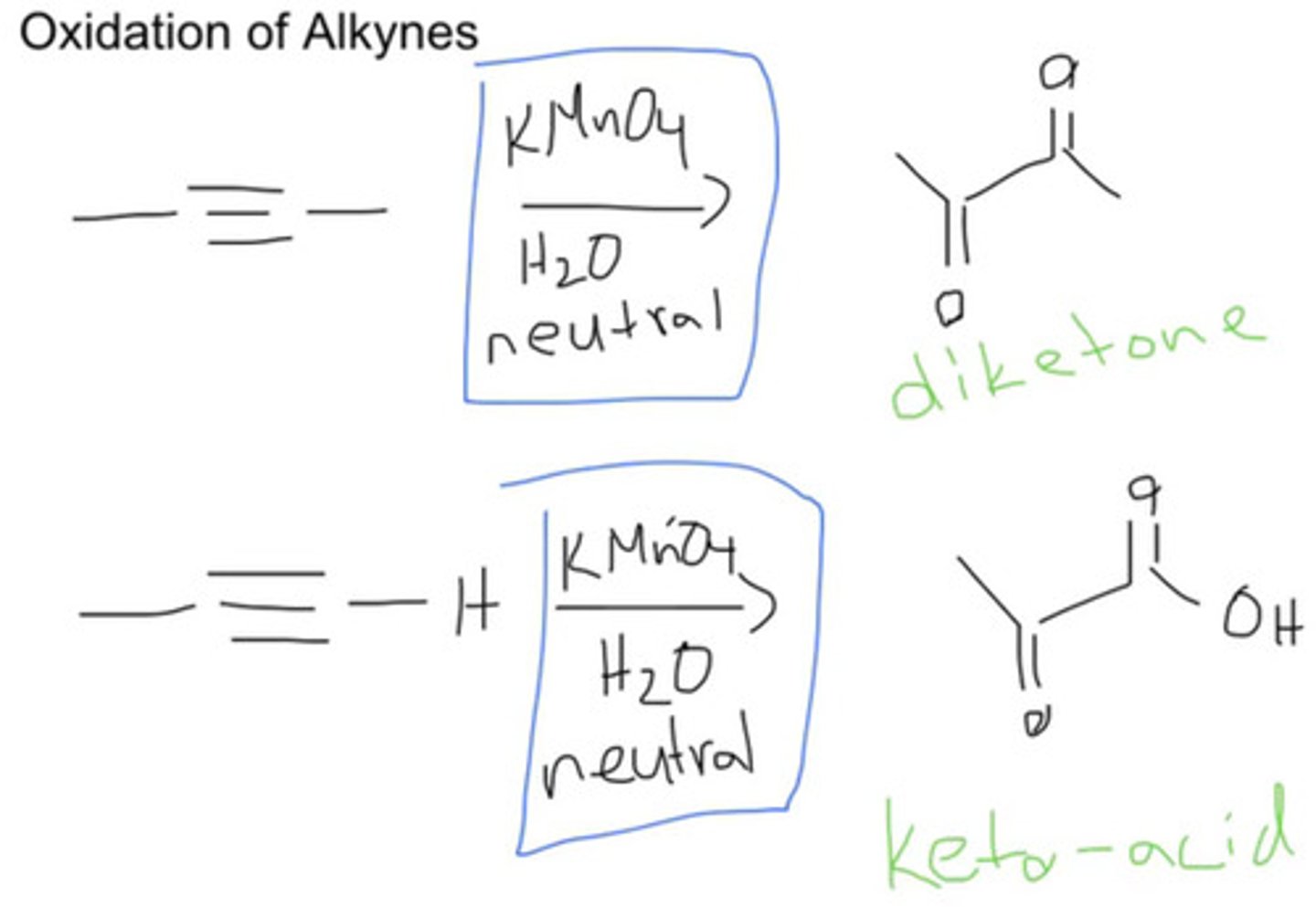
KMnO4/ H20, OH/heat
cleavage of triple bonds into O=C-OH
if triple bond is terminal, H2O and CO2 form
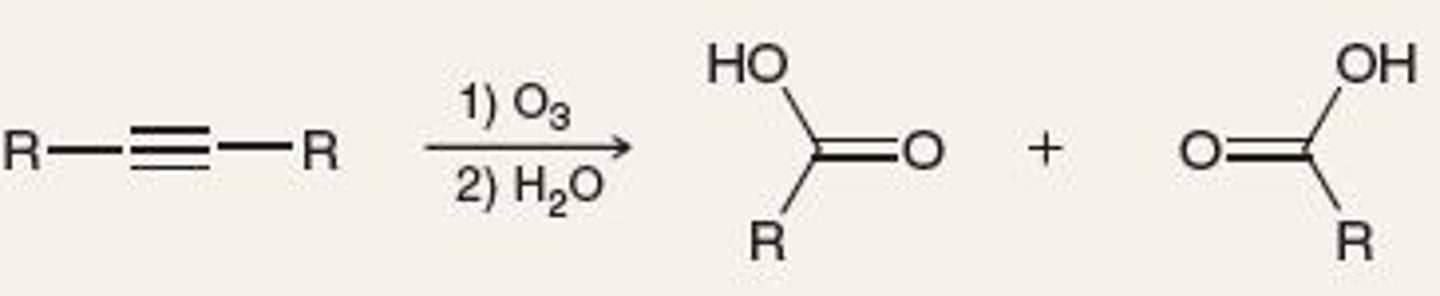
O3, H2O
cleavage of triple bond, into O=C-OH
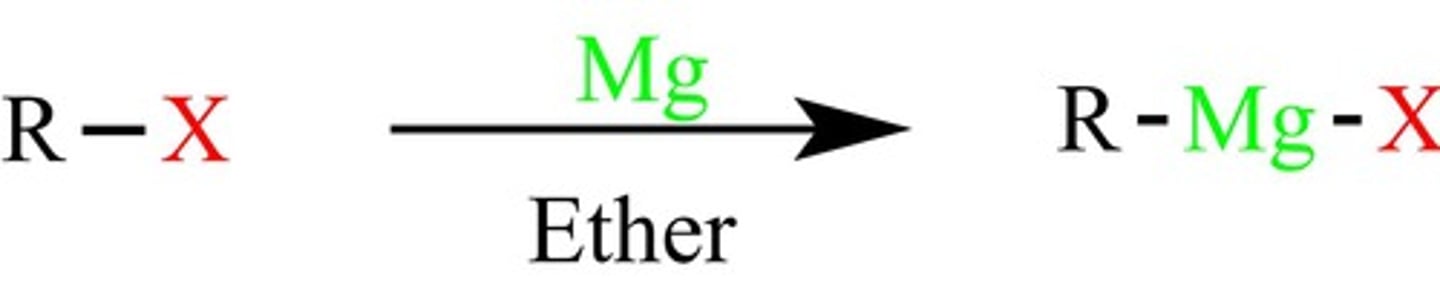
Mg, ether
Replaces Br with MgBr
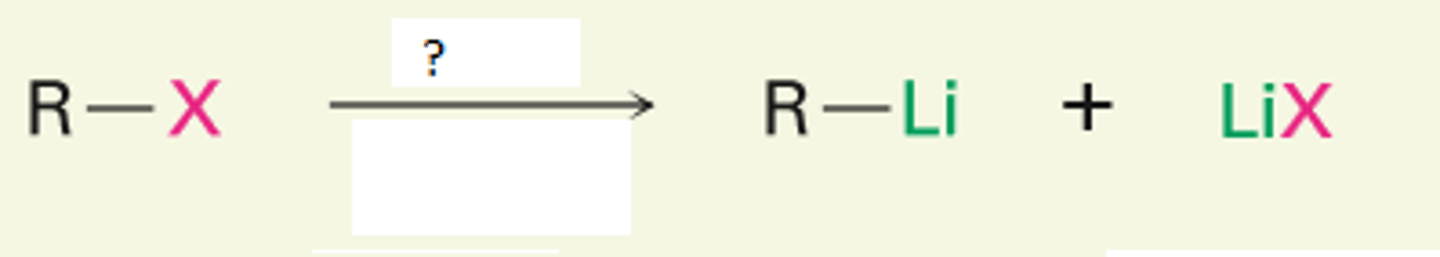
Li, pentane or hexane
replaces halide with Li
O=H-H, H3O+
makes alcohol (OH) from grignard (MgBr)
O=H-R, H3O+
makes alcohol (OH) from grignard (MgBr)
D2O
attaches Detuerium (D) to carbons (replaces MgBr)
NaBH4, EtOH
reduces carboyls (O=) to (-OH)

NaBH4, EtOH with chloride
no reaction
LiAlH4/ ether
reduces carbonyls (O=) to (-OH)
H2, Ra-Ni
reduces carbonyl (O=) to (-OH) and takes away double bonds, to make single bonds
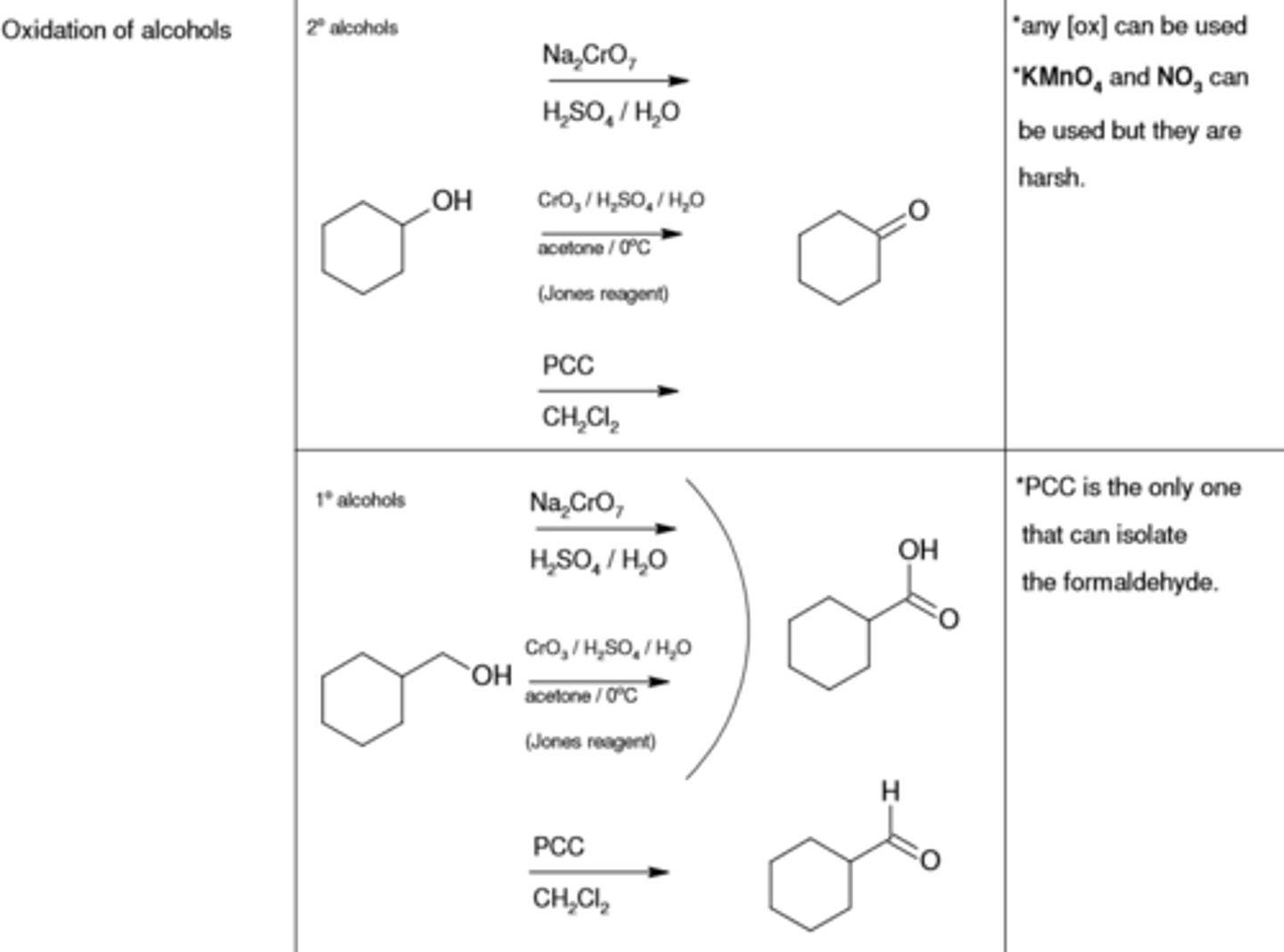
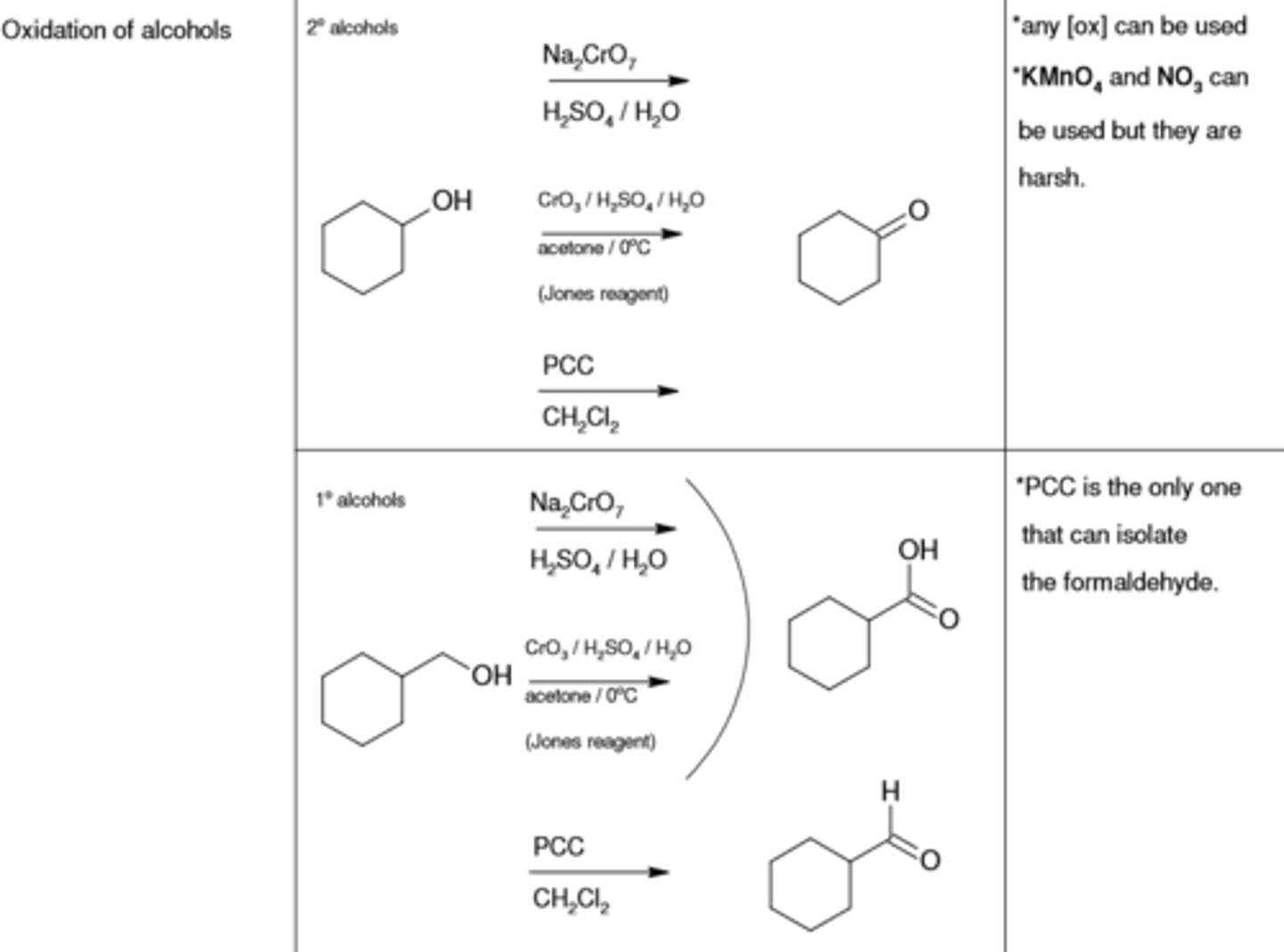
Na2CrO7, H2SO4/H2O
oxidizes alcohol (-OH) to (=O)
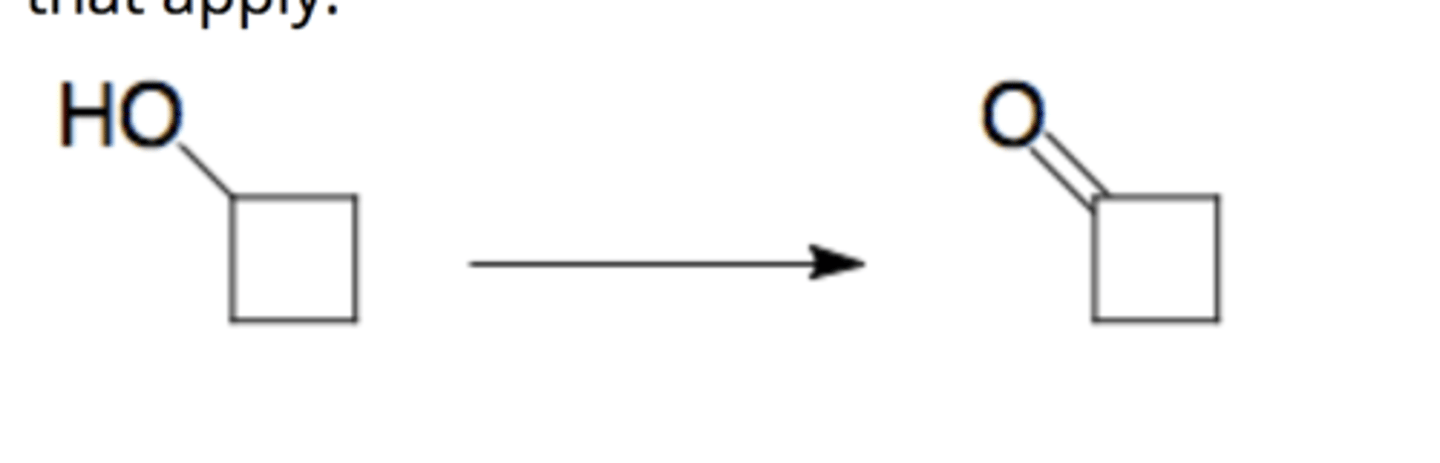
CrO3/ H2SO4/H2O, acetone
oxidizes alcohol (-OH) to (=O)
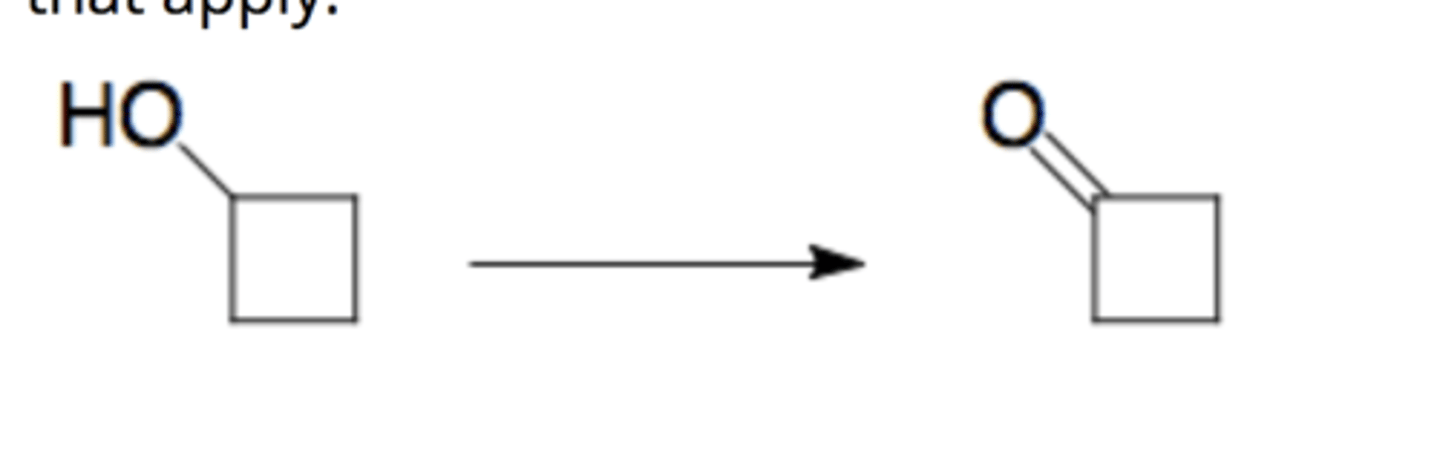
PCC, CH2Cl2
oxidizes alcohol (-OH) to (=O)
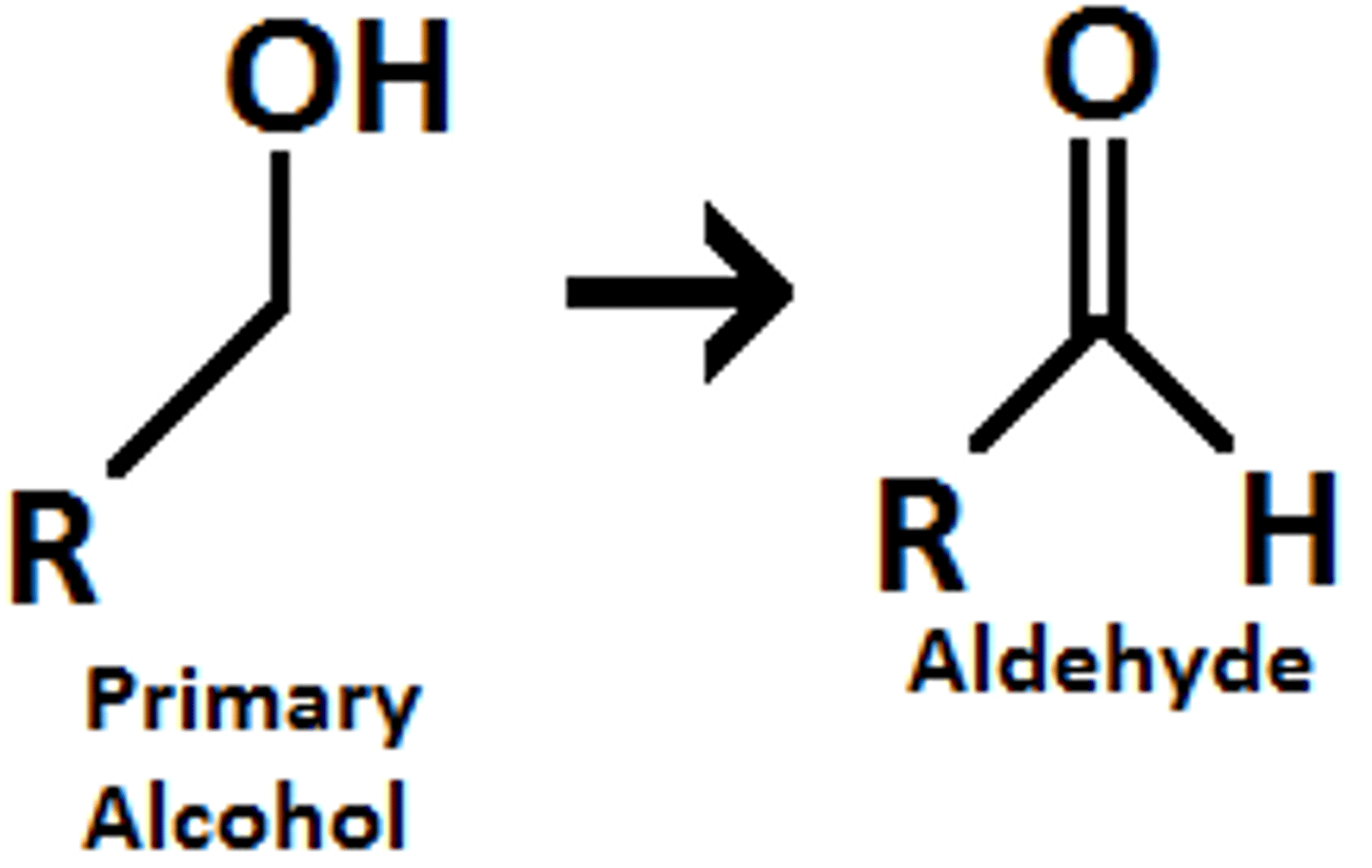
Na2CrO7, H2SO4/ H2O
CrO3/ H2SO4/H2O, acetone
turns primary alcohols (-OH) into carboxylic acids (OH=C=O)
TSCI
Forms (OTos) from (OH)
HCl/ ether, 0C
forms alykly halid (-Cl) from alcohols (-OH)
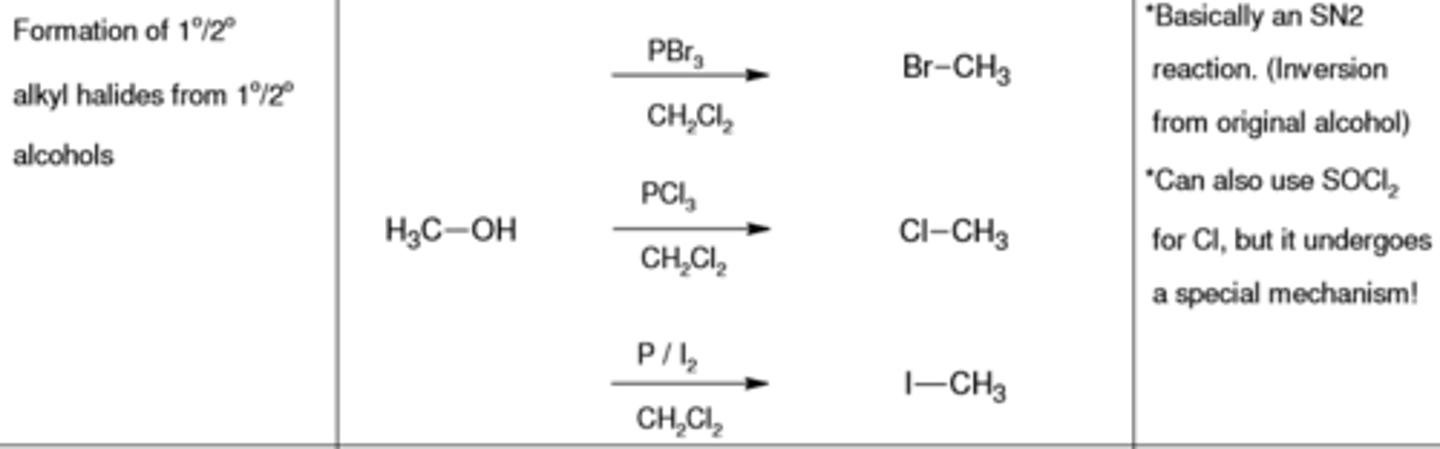
PBr3, CH2CL2
replaces (-OH) with (-Br)
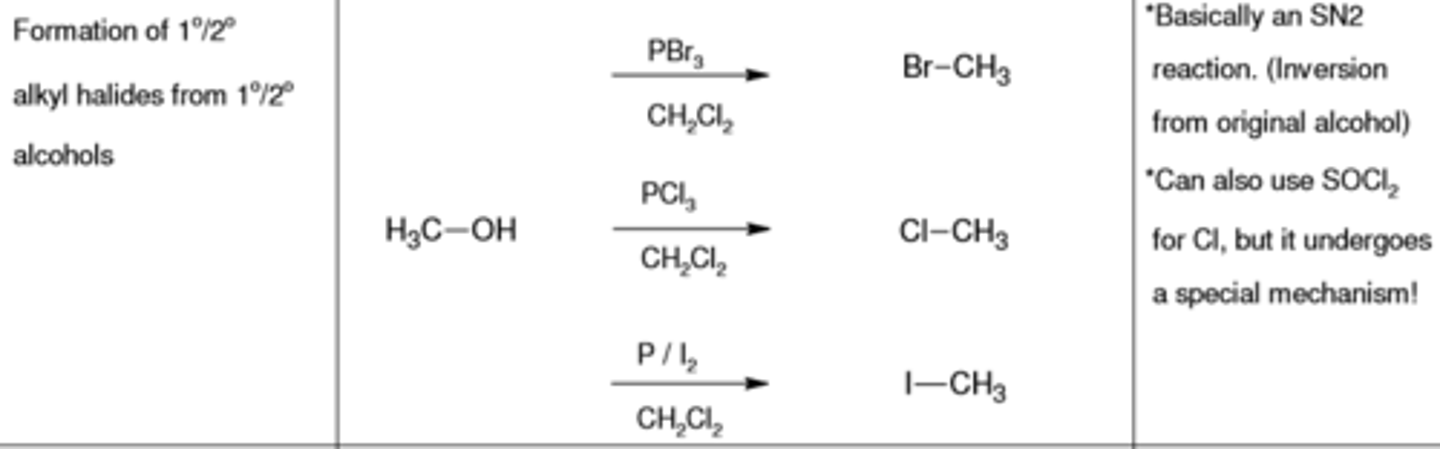
PCl3, CH2CL2
replaces (-OH) with (-Cl)
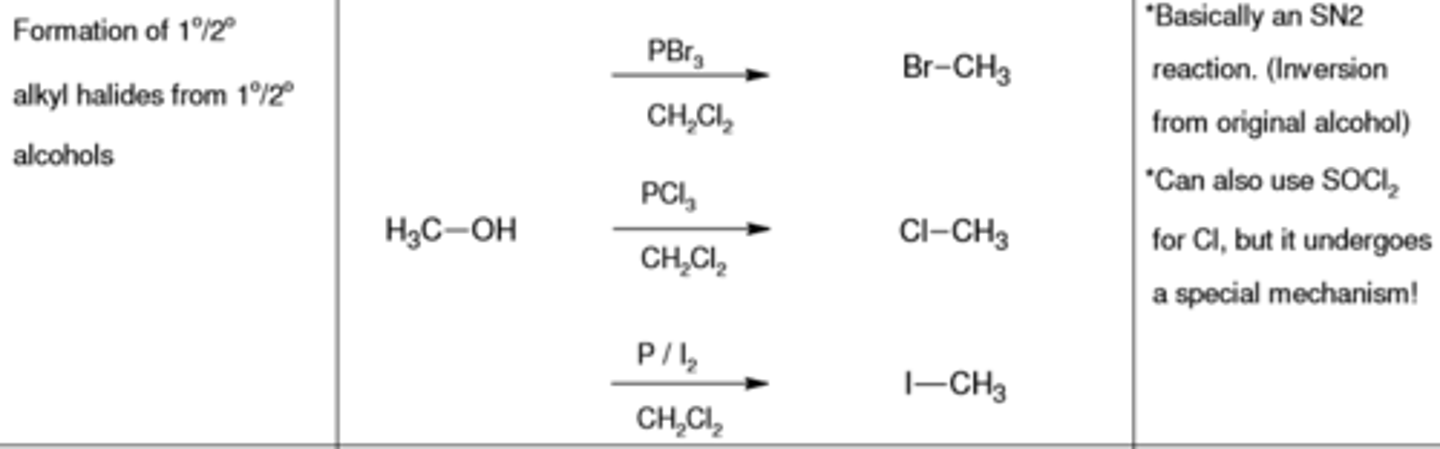
PI2, CH2CL2
replaces (-OH) with (-I)