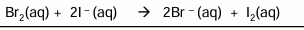Group 7-Halogens
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
What do group 7 elements exist as ?
Diatomic molecules
I.e 2 atoms sharing electrons in a covalent bond
What happens to atomic radius down group 7?
Increases
What colour and state is F2
Pale yellow gas
What colour and sate is Cl2
Green gas
What colour and sate is Br2
Brown/ Red liquid
What colour and sate is I2
Grey solid
What happens to electronegativity down G7 and why
Electronegativity decreases
Atomic radius increases/increased shielding
Weaker attraction between nucleus and shared electrons in covalent bond
What happens to BP down G7 and why
Increases
Halogen molecules get larger
VDWS forces between molecules increase
-Strength of VDWs influences BP and thus the state of the halogens at room temp
What is oxidising ability ?
Power to take electrons
As oxidising agents get reduced i.e accept electrons
What happens to the oxidising ability of the halogens as you go down G7 and why
Decreases
Atom radius increases /increased shielding
Electrons gained less easily
Best oxidising agent
Fluorine
What happens in a displacement reaction
A halogen that is a strong oxidising agent will displace a halogen that has a lower oxidising power from one of its compounds.
What happens when you add chlorine to NaBr give equations
Cl better oxidising agent than Br
Orange solution forms →As Bromide has been displaced/oxidised
2Br- → Br2 +2e-
Cl2 +2e- → 2Cl-
Full ionic equation Cl2 + 2Br - → 2Cl- + Br2
What happens when you add Cl2 to NaI and give equations
Chlorine better oxidising agent than iodine
Brown solution forms →As Iodide has been oxidised
2I- → I2 +2e-
Cl2 +2e-—>2Cl-
Full ionic equation; Cl2 +2I- →2Cl- + I2
What is bleach used in?
Water treatment
Cleaning
Textiles
How do you make NaClO ? i.e used in bleach
React Cl2 with cold dilute NaOH
Equation for producing NaClO
2NaOH + Cl2 →NaClO + NaCl + H2O
ClO- is what kills the bacteria
What type of reaction is the production of NaClO?
Disproportionation reaction
What happens in a disproportionation reaction
One substance is being oxidised and reduced simultaneously
What is being reduced and oxidised at the same time in the production of NaClO
Cl2
One chlorine atom is being oxidised the other reduced
Equation for Cl2 reacting with water and the significance of the product
ClO- produced which kills bacteria in water
Also prevents growth of algae →removing any bad tastes and smell
Is a disproportionation reaction

What’s the cons of reacting chlorine with water?
Chlorine gas can irritate respiratory system and as a liquid it can cause chemical burns
The benefits to health of water treatment by chlorine outweigh its toxic effects.
What happens to chlorine in sunlight ?
It decomposes
This must be prevented when we react it with water →Otherwise you’ll get a different reaction
What reaction occurs when you react chlorine with water in the presence of sunlight
Cl2 doesn’t undergo the disproportionation reaction
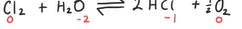
What is the reagent used to test for halides?
Acidified silver nitrate
Why is silver nitrate acidified using nitric acid when testing for halide ions? give equation
Nitric acid will react with impurities
Such as carbonate ions
As they will also react with Silver nitrate producing ppt
Impurities will be released as gas
2H+ + CO32- →CO2 +H2O (H+ comes from ntirc acid)
What happens when you add silver nitrate to test for fluoride ions ?
No visible change
What colour ppt do chloride ,bromide and iodide ions form ?
White = chloride
Cream =bromide
Yellow=iodide
What is the follow up test used to test for halide ions and why is it used
Ammonia solution is used
Colours of the ppt are difficult to differentiate
What ppt will dissolve in dilute ammonia solution
AgCl
What ppt will dissolve in conc ammonia solution
AgBr
What ppt doesn’t dissolve at all in ammonia solution
AgI
What is reducing power?
Ability to donate electrons
As reducing agents gets oxidises
What happens to reducing power of halide ions as you go down group 7 and why
Increases
Ions get bigger
Attraction between nucleus and outer electrons decreases due to increased shielding
Electrons lost more easily
What is produced when Halide salts react with concentrated sulfuric acid?
Sodium hydrogen sulfate/NaHSO4
Hydrogen halide e.g HCl or HBr or HI
(These reactions with sulfuric acid prove the pattern of reducing ability)
What is the equation of NaF reacting with conc H2SO4 and the observations
White steamy fumes of HF
F- is not a strong enough reducing agent to reduce S in H2SO4
So this is not a redox reaction
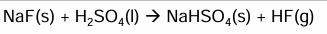
What is the equation of NaCl reacting with H2SO4 and observations
White steamy fumes of HCl
Cl- ion is not a strong enough reducing agent to reduce the S in H2SO4
So this not a redox reaction

What type of reaction occurs when NaF or NaCl reacts with H2SO4
Acid base reaction
H2SO4 acts as a proton donor
What equations occur when you react NaBr with H2SO4 -observations
2Br- → Br2 + 2e-
H2SO4 + 2H+ + 2e- → SO2 + 2H2O
Combine the 2 equations to get the full redox equation
-Orange fumes as 2Br- is oxidised into Br2
-SO2 is a choking gas
How do u test for SO2
Use filter paper soaked in K2Cr2O7
Will change from orange to green
What happens Iodide ions react with sulfuric acid
I- can reduce the sulfur to produce 3 different Sulfur containing compounds
Because I- is an extremely good reducing agent
What is the reaction for Iodide ion reducing S from H2SO4 into SO2
H2SO4 → SO2
2I- → I2
Combine to get full equation
I2 produced will either be a grey solid or purple gas
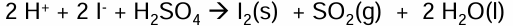
Reaction of I- reducing Sulfur in H2SO4 into S
H2SO4 → S
2I- → I2
Combine to get full equation
Sulfur produced is a yellow solid
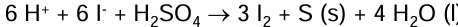
Reaction of I- reducing the S in H2SO4 into H2S
H2SO4 → H2S
2I- → I2
Combine to get full equation
H2S has a rotten egg smell
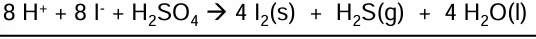
What happens when you add Br2 to NaI and give equations
Bromine will displace Iodide ions