Green Oxidation
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Whats the premise of Green Oxidation lab
Using a Cu-based reeaction to catalyze the oxidation of a primary alochol to a aldehyde
In what conditions did the Green oxidation lab take place
In Aerobic conditions (the presence of oxygen)
What ion Cu did we use
Copper (I)
Why did we choose Copper over OsO4
OsO4 is highly Toxic
Why did we choose Copper over CrO3
(Cr(IV))
Chromium is bad for the enviorment and hard to dispose it
What are some metals that we typically see oxidize reactions
OsO4, Cro3 in conc. H2SO4, KMnO4, and PCC
What does PCC stand for
Pyridinium chlorochromate
Why are the oxidative agents that I just noted bad
We need the same mol amount of reactant that needs to be oxidize (though they are catalyze we need alot)
It yeilds by inorganic salts as by products that requires an additional seperative step
They promote over oxidation, we just want an aldehyde not a carboxylic acid
What is the definition of a catalyst
An additive used in sub-stiochiometric amount that increase the rate of reaction without itsefl being consumed or undergoing a net chemical change
How does a catalyst increase the rate of reaction
lowers the activation energy process, mean there is a change of the energy at the transition state
What is the EXACT catalyst molecule (all)
Copper (I) Bromide
2,2-bipyridine (bpy)
N-methyl; imidazole (NMI)
TEMPO
Tell me the reaction of this lab
CuBr+NMPI+BPY form this big catalytic group which then associates with TEMPOH
this group gets oxdized by the use of ½ O2 and H2O to go from a red/brown color to a green color
In this oxidization reacction Copper 1 turns into copper 2 by adding an OH to it the Oxygen came from the ½ O2 and the H came from TEMPOH to become TEMPO
Then this green group oxidize a benzyl alcohol to an benzaldehyde and releases H2O to go back to it’s orginal red/brown color
Summary: What gets reduced
TEMPO and Cu(II) in the coordinated complex
Summary: What gets oxidized
TEMPOH and Cu(I) in the coordinated complex
Give me the stichiometric reaction of this lab
ArCH2OH + ½ O2 → ArCHO + H2O
What does TEMPO Stand for
2,2,6,6-Tetramehyl-1piperidinyloxyl
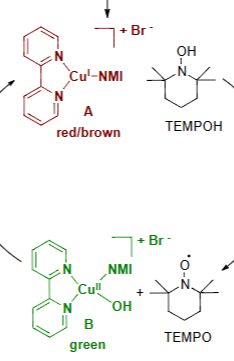
Attached are 4 molecules what gets reduced and what gets oxidized
The red/brown molecule gets oxdized
The Green molecule gets reduced
Is Copper Bromide dense
Yes!
Based on the American Chemical Society “Green
Chemistry Institute”, how does this reaction conform to the principles of green
chemistry?
Prevention of waste in compirson of other reagents
highly atom-econimical
Less hazard chemical substance in comparison to other metal catalyst
things to ask
What part of the reaction are we supposed to know (ask about the top of page 2)
What Hazards should we know
What part of the procedure should we know, will it ask why specific reagents
How did we know the reaction is complete
When an solution turned green and a color change was seen
Why did we use nitrogen to evaporate at the end
So iwe can remove our solvent (pentane) and it’s a safe way
In the lab we did a liquid-liquid extraction which layer is which
The top layer will be the organic layer because the pentane solvent is less dense than water.
What was the main hazard we needed to look out for and what was it’s hazard
Acetone and pentane, they are flammable