optical isomerism
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
what is optical isomerism?
a form of stereoisomerism
what is optical isomerism a result of?
it occurs as a result of chirality in molecules, limited to molecules with a single chiral centre
what do optical isomers contain?
a chiral centre: a single carbon atom is bonded to 4 different atoms or groups
outline what type of molecules optical isomers are
the molecule has no centre of symmetry, no plane of symmetry + no axis of symmetry
because the molecule has no symmetry, what does this result in?
an asymmetric carbon atom (a chiral centre)
what are optical isomers described as? outline why
as non-superimposable over its mirror image → this means it cannot be placed on top of its mirror image as they are different
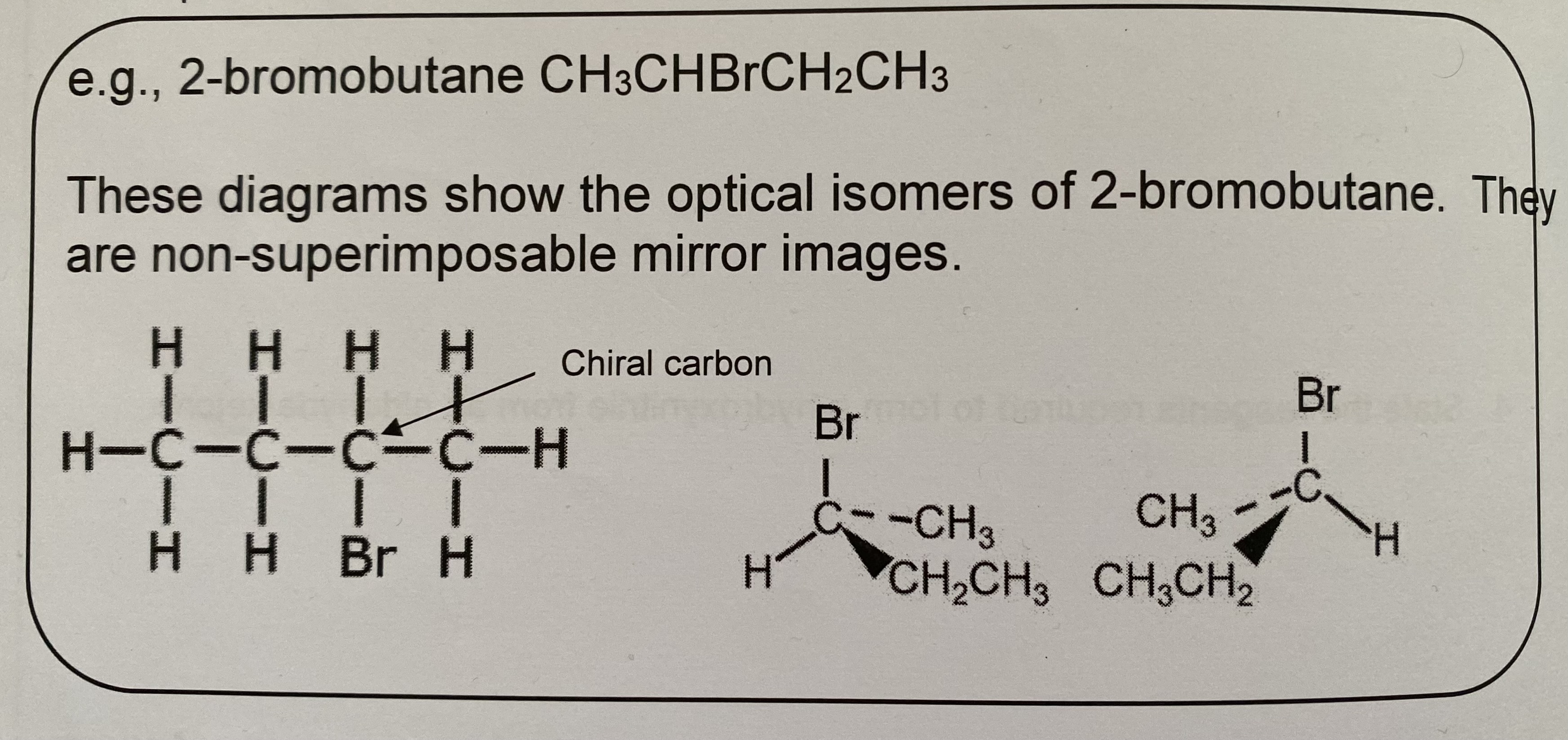
eg how many chiral carbons are in this molecule, CH₂(OH)CCl(CH₃)CH₂CH₂CHClBr
two
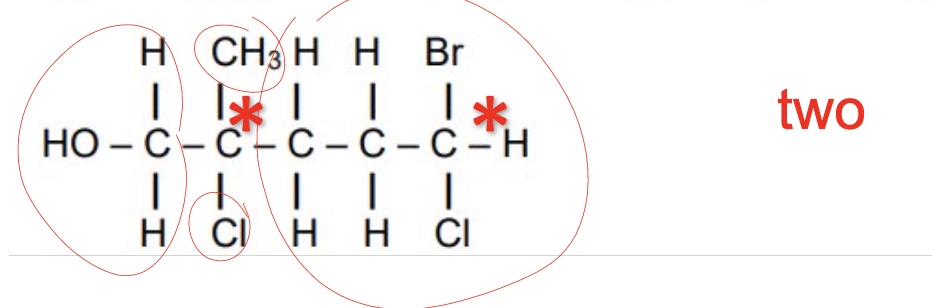
what can optical isomers also be known as?
enantiomers
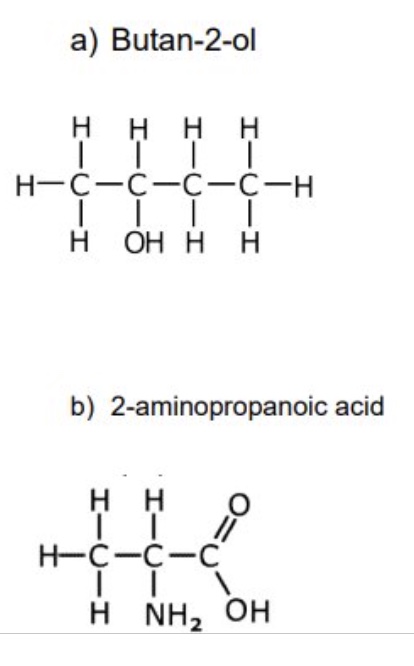
for these two examples, identify the chiral carbon + draw 3D representations to show the optical isomers (enantiomers)
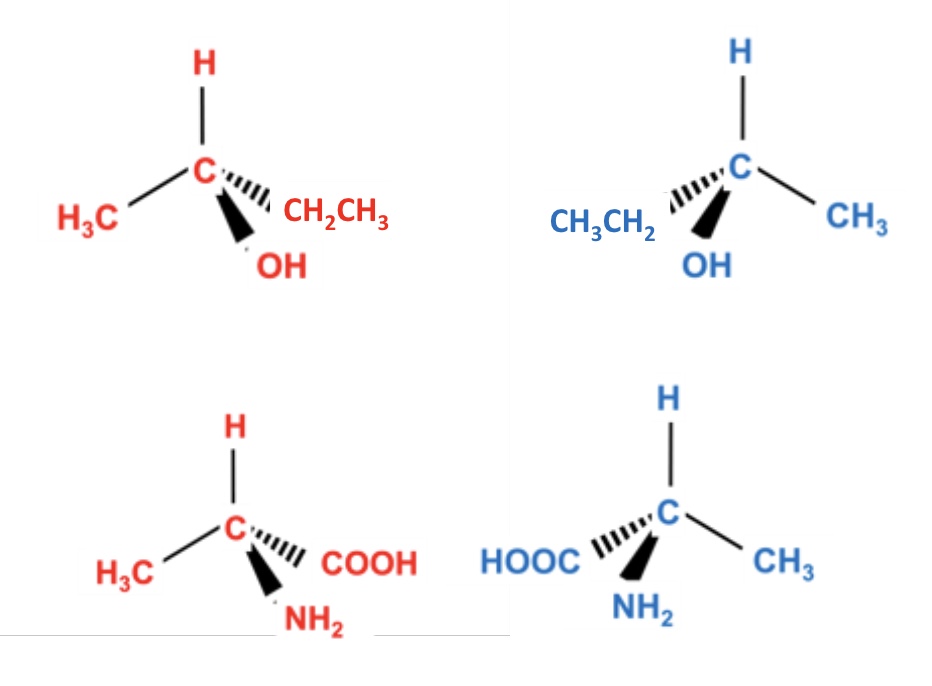
the pair of optical isomers (enantiomers) have the same… and…. apart from one importance difference.
what is this difference?
they have the same chemical properties + physical properties
they rotate the plane of polarised light in opposite directions
outline what ‘plane polarised light’ means
light that consists of vibrating electric + magnetic fields
waves that have vibrations occurring at right angles to the direction of motion
pass the light through a special filter, then we get all vibrations removed except one
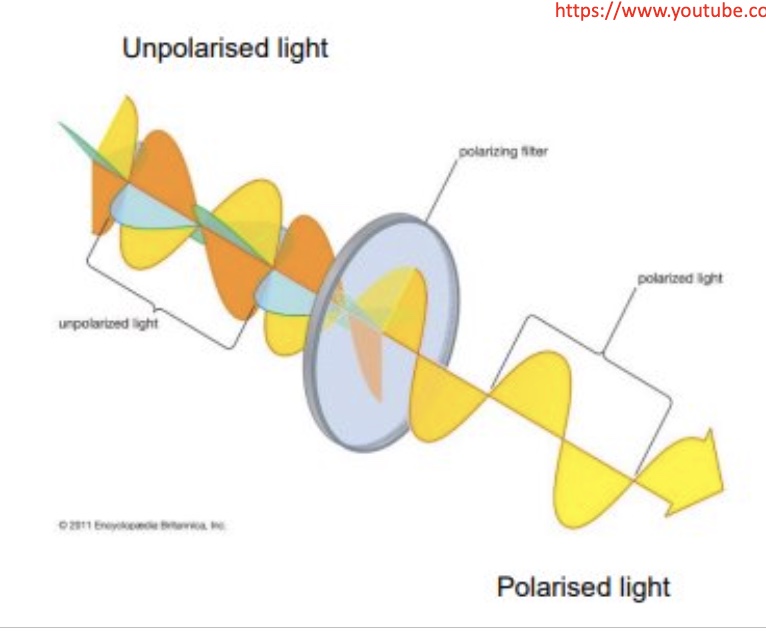
explain how polarised light can be used to distinguish between optical isomers
when the new plane of polarised light passes through a solution of an enantiomer, the plane gets rotated through a particular angle → essentially different enantiomers of the same substance will rotate the plane differently
one enantiomer will rotate the plane of light to the left (anti-clockwise) + the other will rotate the plane of light to the right (clockwise) by equal amounts
when would the sample be optically active?
when the sample contains only one form of the optical isomer, there will be a rotation of the light → the sample is said to be optically active
when would the sample be optically inactive?
when both optical isomers are present in equal quantities, no rotation of the plane of polarised light is observed so the sample is said to be optically inactive
what is a racemic mixture (racemate)?
a mixture that contains equal amounts of each enantiomer
why is a racemic mixture optically inactive?
because it contains equal amounts of the two enantiomers so each rotate the plane of polarised light in opposite directions equally
enantiomers also commonly have different simple characteristics, give 3 examples of this

some drugs are what?
optically active
explain why some drugs are optically active + what this can lead to
this is because the drug may have a specific enzyme based (or similar) receptor
looking at the lock and key model, we see that the receptor will only accept a very particular isomer shape
this can lead to effective + ineffective drug isomers → sometimes there can be racemic mixtures
how would you overcome the problem of having effective + ineffective drug isomers? give 3 solutions but give the problems with each one

give 2 examples of drugs that contains enantiomers
ibuprofen
thalidomide
ibuprofen is often sold as a what?
as the racemic mixture
what was thalidomide used to treat? outline the problems it had
