enatiomers
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
What are diastereomers?
Stereoisomers that differ at some but not all chiral centers (not mirror images).
diff from enantiomers bc enantiomers differ a every chiral carbon, while diasteremers differ only at some.
so for these some are the same and some are opposite.
Do diastereomers have identical chemical properties?
No, they have different chemical properties due to different spatial arrangements.
Diasteromer example
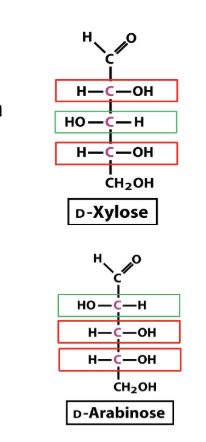
What does "D" mean in D-sugar?
"D" stands for dexter (Latin for right)—a sugar is called a D sugar if the chiral carbon atom furthest away from the he carbonyl group has an OH group on that group on the right side
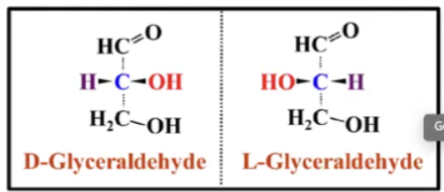
Leva - means left a OH group on the furthest chiral carbon from carbonyl group is called an L sugar
epimers
Epimers are sugars that differ in configuration at only one chiral carbon.
a type of diasteromer
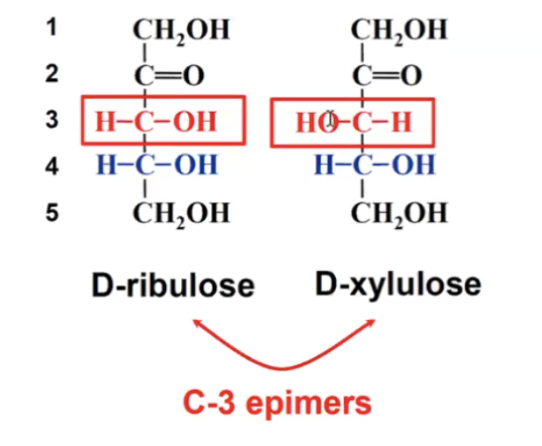
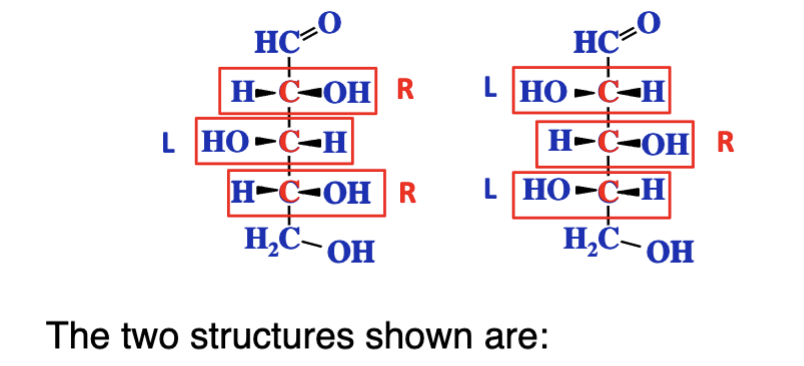
Enantiomers of each other
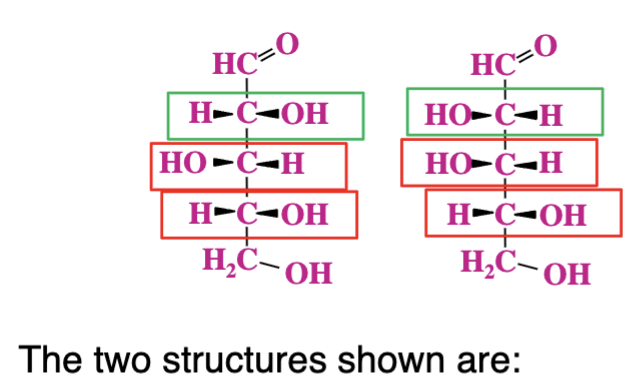
Both D sugars, diatereomers, epimers
How many stereoisomers does a sugar with n chiral centers have?
It has 2ⁿ stereoisomers.
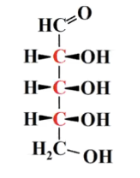
how many does glyceraldheyhde have?
2^1= 2 (1 chiral centre)
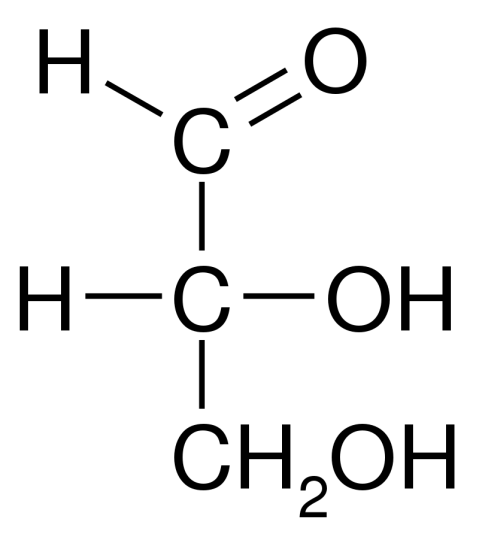
How many stereoisomers does an aldose with 5 carbon atoms have?
2³=8
8 steroisomers (half of these are D and other half are L)

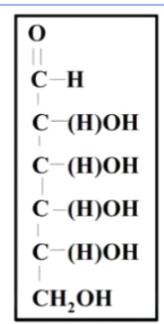
2^4= 16
stereoisomers=16=x
half of the steroisomers are D half are L so the amount that is D is 8 = Y
so 16 and 8

What happens to the number of isomers each time a carbon atom is added to a sugar?
The number of isomers doubles.
If we have 2 sugars then we'd have 2 conformation is 2 (21=2)
If we have 2 sugars with 2 chiral atoms we see 22=4
If we have 3 chiral atoms then 23=8
24=16
So every time we add carbon, we are doubling the isomers
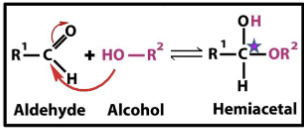
Aldehyde + Alcohol =
Hemiacetal
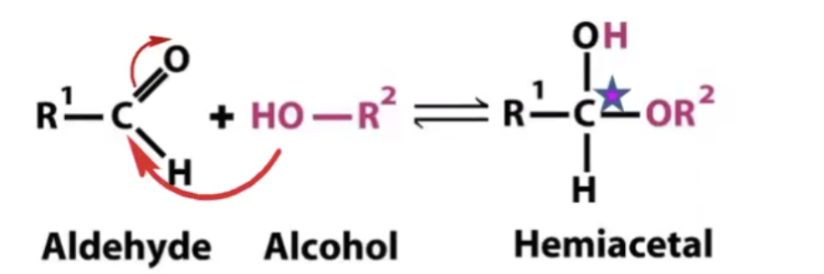
( note: the original carbonyl carbonyl carbon becomes carbon upon formation of hemicetals).
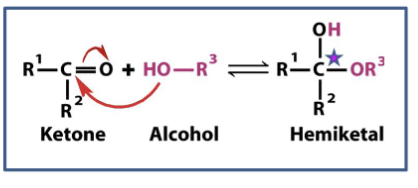
Ketone + Alcohol =
Hemikital
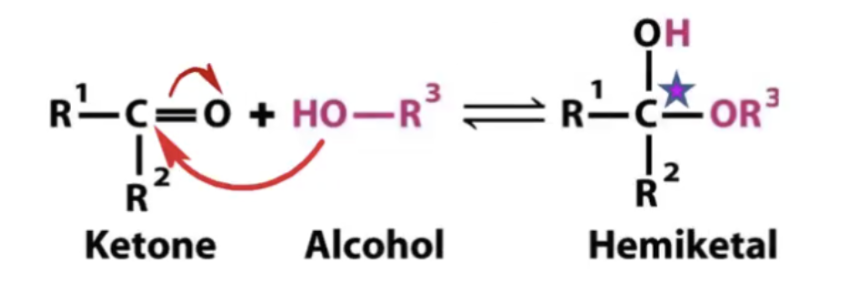
( note: the original carbonyl carbonyl carbon becomes carbon upon formation of hemiketal).
where do hemiketal and hemicetals form
can be between molecules or within the same sugar because it contains both alcohol and aldehyde and ketone groups in the same molecule.
What happens when hemiacetal or hemiketal form intramolecularly?
a ring structure forms.
the oxygen of the alcohol group nucleophilically attacks the carbonyl group carbon on the same molecule
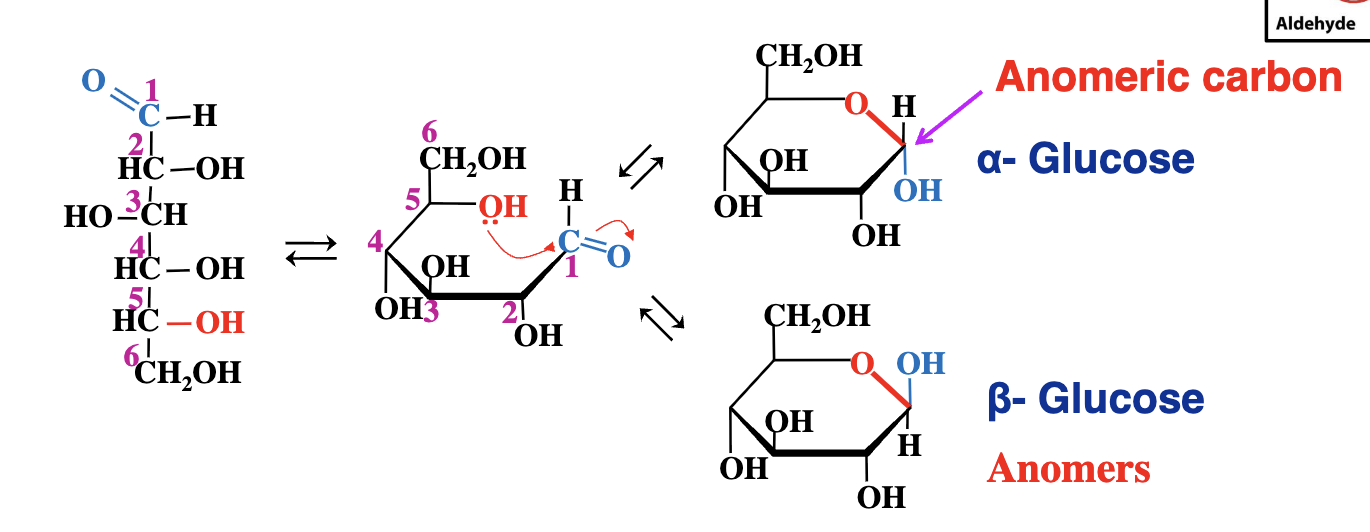
Cyclization of glucose process
The OH group at carbon 5 attacks the carbonyl carbon of the aldhyde group to form a six membered ring
Ring forms
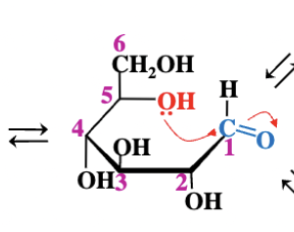
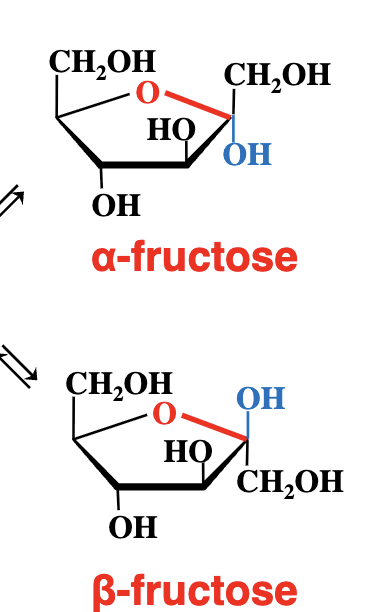
C1 is now assymetric - and two stereoisomers are born (alpha and beta)
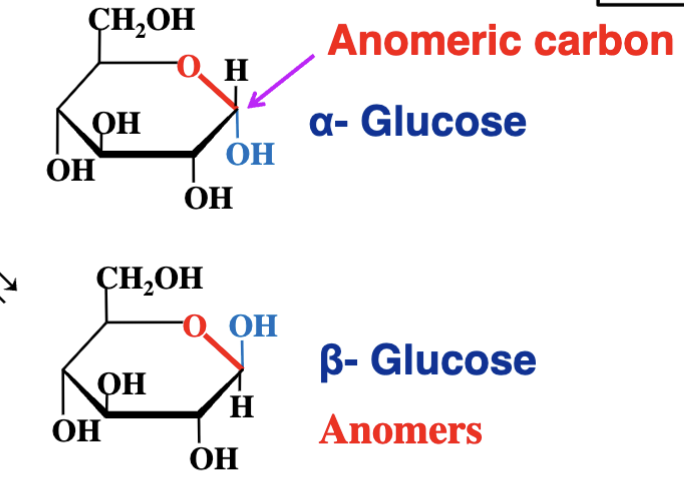
These isomeric forms that differ only in their configuration around the hemiacetal or hemiketal carbon are called anomers. (meaning they only vary in the carbon taht was attacked by the alcohol group). this carbon being the anomeric carbon.
What are anomers?
Isomers that differ only at the anomeric carbon's configuration (resulting from ring formation).
alpha vs beta anomers
above - beta
below - alpha
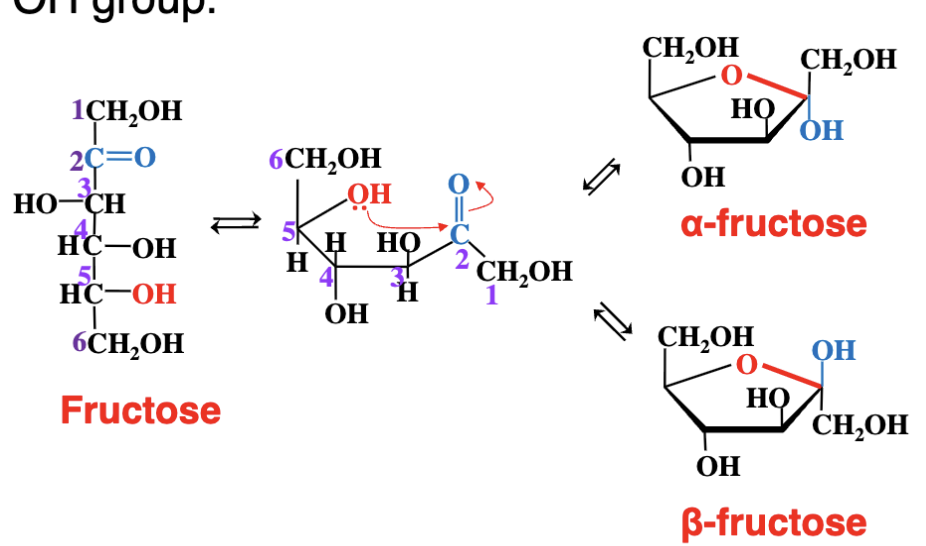
What happens during the cyclization of a ketose like fructose?
The same as for glucose (an aldose)
However in this case we get a 5 membered ring.
What initiates cyclization in a ketose sugar like fructose?
The nucleophilic oxygen of an OH group reacts with the electrophilic carbon of the carbonyl group. - just like we see with glucose (an aldose)
orders of cyclization
aldose —>
cyclziation of ketose (liek fructose)
Mutarotation
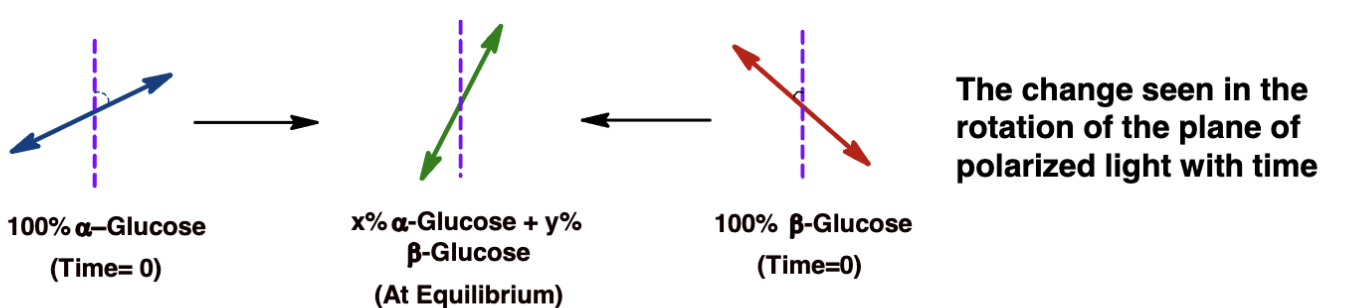
When glucose is dissolved in water, it switches back and forth between its ring forms (α and β) by briefly opening up into a linear-chain form.
This back-and-forth is called mutarotation, and it keeps happening until the amounts of α and β forms settle into equillibrium.
so. during this we see a mix of alpha, beta and linear forms
What physical change is observed during mutarotation?
A change in the rotation of plane-polarized light over time.
What forms of glucose are present at equilibrium in water?
Approximately 1/3 α-D-glucose, 2/3 β-D-glucose, and trace amounts of the linear form.
Why is β-D-glucose more prevalent at equilibrium than α-D-glucose?
Because in the α form, the OH groups are closer together, causing more steric strain. (atoms are too close together causing repulsion)
B-D-gluocse has OH groups spaced farther apart stabilizing it.
Does mutarotation occur only with glucose?Does mutarotation occur only with glucose?
No, mutarotation can occur with any sugar that can form a hemiacetal or hemiketal.
Mutatoration process example
Here's how it works step by step (for glucose, for example):
Start in α-D-glucose form (a ring).
The ring opens up into the linear form of glucose.
The linear form can then re-close into:
the α form again, or
the β form instead.
This process can repeat — ring ⇄ linear ⇄ other ring form — over and over.
This interconversion is called mutarotation.
The sugar cannot go directly from α to β without first opening into the linear form.
This is why mutarotation requires water — it allows the ring to open and close
What two types of rings can form when sugars cyclize?
Pyranose (6 membered ring)
Oh group further from carbonyl attacks carbonyl carbon
Furanose (5 membered ring)
OH group closer to the carbonyl attacks it.
What determines whether a sugar forms a pyranose or furanose ring?
The thermodynamic stability of the ring which is dependent on the particular geometry of the molecule.