Module 8 ~ Lipids
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
What are the structural characteristics of Lipids?
a defining feature of insolubility in water
lower in weight than other biomolecules
form aggregates rather than polymers
What are the functional characteristics of lipids?
energy storage
structural components of membranes
active roles: signaling (intracellular and extracellular, and enzyme co-factors & vitamins
What does a fatty acid look like?
hydrocarbon with a carboxylic head: the head is the first carbon
differ in length and degree
usually an even number of C’s (12-24)
What is the significance of double bonds within fatty acids?
saturated have NO double bonds
unsaturated = one double
poly = multiple
double = cis and separated by a methylene group
Which three points of potential variability does fatty acid nomenclature address?
length
presence/absence of double bonds
location of double bonds
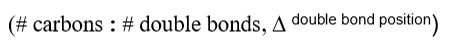
Which type of chains have a stronger association through hydrophobic and van der Waals interactions?
longer more than shorter
saturated more than unsaturated (4×4’s packed neatly)
What is the melting temperature a reflection of in fatty acids?
length and degree of saturation of the hydrocarbon tails
double bonds have a greater influence than the length of the tail on the fatty acid associations
inc length = inc melting temp
saturated + long = solid
unsaturated: the more double bonds the more liquid
What are fatty acids required for energy stored as?
triacylglycerols
What is the name of the intracellular space of energy storage in animal tissues?
adipocytes
What do shorter hydrocarbons mean in terms of double bonds?
more double bonds and more fluidity
What are ester linkages?
what joins the three fatty acids to a glycerol backbone
remove the polar carboxyl group to make a more hydrophobic molecule
How much more energy is in fats than carbs on a gram basis?
6 times
What is low oxidation state?
less oxygenated fuels burn more efficiently
more energy in bacon (fats) than in a bagel
What is hydration state?
lipids are hydrophobic with limited interaction with water providing a compact, dehydrated energy storage form
like the jelly powder vs making the jello: water makes it heavy
What is saponification?
treating fats with a base
making soap from fat
treatment of fat with a strong base breaks the ester linkages to release free fatty acids
What do micelles do?
work in detergents and soap to capture hydrophobic molecules because of its hydrophilic head and phobic tail
What is olestra?
taste and looks like fat, but can’t be processed by our digestive system
eight fatty acids linked to a sugar group
causes loose stools
What do waxes serve as?
esters of long-chain fatty acids and long chain alcohols
insoluble and high melting temp (60-100 C)
on leaves, fruits, animal skin and feathers
What do membrane bilayers do?
define cells and regulate the composition of the intracellular environment
spontaneous
How can membrane lipids be classified?
based on their backbone (glycerol or sphingosine)
by polar head group (phospho. vs glyco)
What is the most abundant lipids in membranes?
glycerophospholipids
What is the make-up of glycerophospholipids?
a glycerol backbone with a phosphate at the C3 position (Glycerol 3-phosphate)
Glycerol-3Phosphate + 2 fatty acids = phosphatide or diacylglycerol 3-phosphate
In glycerophospholipids, which is the point of attachment for a variety of polar head groups?
the phosphate group
How can the polar head groups in glycerophospholipids differ?
their charge: positive, negative, or neutral
have specialized functions above and beyond just polar head groups
What does phosphatidylserine do?
healthy cell, they are positioned inside and pointing inside the cell
cancerous, infected: they become pointed outside and triggers programed cell death (apoptosis)
What does phosphatidylinositol do?
involved in intracellular signal transduction and messenger
Why do some plants use sugar and sulfate polar head groups instead of phosphate?
sometimes its a limiting resource
conserve use of phosphate for more critical applications such as nucleic acids
these are called galactolipids or sulfolipids
What are spingolipids?
derived from sphingosine, a long chain amino alcohol
structurally similar to monoacly glycerol
How is ceramide formed?
single fatty acid linked by amide bond (amino and carboxyl) to form ceramide
structurally similar to diacylglycerol
What are the common sphingolipids?
sphingomyelins: phosphocholine
cerebrosides: sing sugar, glycosphingolipid
gangliosides: multiple sugar, glycosphingolipids
What determine blood types?
glycosphingolipids pointing out for the cell to recognize them
What do membrane lipids of extremophiles often contain?
ether linkages
branch points within the hydrocarbon tails
membrane spanning hydrocarbon tails composed from a single molecule
What are sterols?
structural membrane lipids
contain four fused ring steroid nucleus: 3 -six carbon rings and a 5 carbon D ring
rigid and nearly planar
precursors for many biologically active products
base in cholesterol
What is cholesterol?
serves for mediating membrane fluidity and serving as a precursor of steroid hormones and bile salts
is breaking the movement and breaking the crowd in smaller groups, dampening movement of fluidity
What are some active roles of lipids?
intracellular signaling molecules
hormones
enzyme factors
enzyme cofactors
pigments
vitamins
What are eicosanoids?
paracrine hormones: localized and specific
derivatives of fatty acids: arachidonic acid, is the precursor for other classes
What are the three types of eicosanoids and what are their roles?
Prostaglandins
constrictions of blood vessels, fever and inflammation
Thromboxanes
involved in blood clot formation
Leukotrienes
smooth-muscle contraction: produced in a different pathway
What can block fever and blood clot formation?
Aspirin, but doesn’t block Leukotrienes because it has a specific pathway
What do steroid hormones do?
hydrophobic oxidized sterol derivatives
carried through blood stream by carrier proteins
Pass through plasma membrane to bind receptors in the nucleus
alter patterns of gene expressions and metabolism
How do phosphatidylinositols act as intracellular signal?
have 4,5 biphosphate on the inner membrane face
takes info that’s outside and convert it to intracellular responses
receptor causes the reaction inside
What signaling molecule does phosphatidylinositols have
the phospholipase C
triggers cleavage of the polar head group to produce inositol to release calcium which in turn activates specific protein kinases, the body diffuses and does so as well (not redundant, a safety feature)
*has a domino effect
What are vitamins?
four lipids: ADEK
all contain rings and long aliphatic side chains
highly hydrophobic
differ widely in their functions
What does Vitamin D do?
regulates calcium uptake and deposition
obtain from diet or produced endogenously
endogenous production (body does it by itself) ONLY through exposure of UV light
What is associated with insufficient and excessive of Vit D?
insufficient = skeletal defects (Rickets)
excessive = calcification of soft tissues
Where is Vit A obtained from?
liver, egg yolks, and milk products
*some animals have sufficient Vit A in their livers to present a danger to humans if they were to consume them
What are the three forms of Vit A?
alcohol (retinol)
aldehyde (retinal): light sensitive compound with a role in vision, “red eye” in photos results from retinal
retinoic acid
What is vitamin E?
a reducing reagent that scavenges oxygen free radicals
may prevent damage to fatty acid in membranes
often used as an additive in cosmetics
What does the deficiency of Vitamin E cause?
scaly skin, muscular weakness and sterility
What is Vitamin K?
required for the synthesis of blood coagulation proteins
was used as rat poison where is caused rodents to suffer uncontrolled bleeding
now given to individuals who suffer excessive blood clotting