Quantum Mechanics Unit 12 Key Terms
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
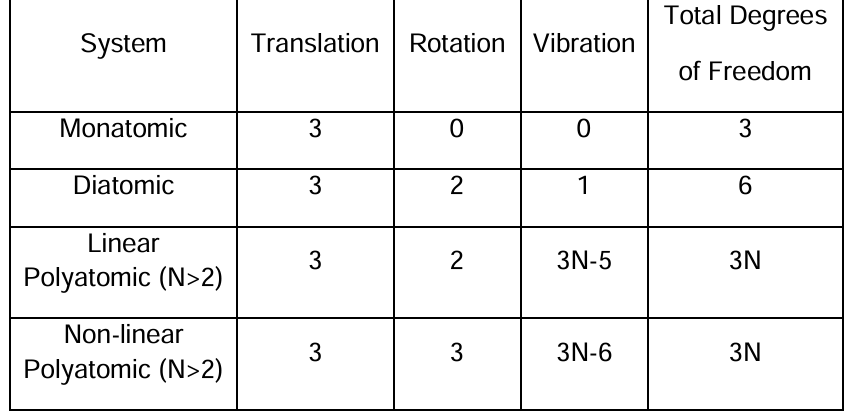
translational, rotational, vibrational degrees of freedom
monoatomic: 3 translational.
diatomic: 3 translational, 2 rotational, 1 vibrational. 6 total
polyatomic: linear - 3 translational, 2 rotational, 3N-5 vibrational, 3N total
nonlinear - 3 translational, 2 rotational, 3N-6 vibrational, 3N total
rotational term
Rotational energy of a molecule in wavenumbers F(J), predicted by the rigid rotator model and dependent on the rotational constant B. Add more
nonrigid rotator model
Actual energy levels are slightly different (lower) from rigid rotator model, as nonrigid rotator model factors in centrifugal distortion, or the way molecules move further apart as rotation gets faster, molecule distortion as it rotates Add more
centrifugal distortion constant
As molecules distort or move further apart as they rotate, the centrifugal distortion constant is subtracted from the rotational term to lower the overall energy level.
vibrational term
The vibrational energy of a molecule in wavenumbers, G(n)
overtones
A series of less intense lines (frequencies at lower intensities), almost integral multiples of the fundamental resulting from transitions from n = 0 to n = 2 (first overtone) and from n = 0 to n = 3 (second overtone).
anharmonic oscillator
Considers and accurately represents the potential energy far from the equilibrium distance, energy results lower than predicted by the harmonic-oscillator model. Energy levels are not evenly spaced, and the energy difference between levels gets smaller as quantum number n increases.
anharmonicity constant
quantifies how a molecule's actual vibrational energy levels deviate from those of an ideal harmonic oscillator. Much smaller than the harmonic term
R , P, and Q branches
Absorption in IR regions of spectra, R branch at high frequency (where lines get closer together as J increases), P branch at low frequencies (lines get farther apart as quantum number (J?) increases, and Q branch, where the lines are very close in the spectra (what does this mean) due to vibration-rotation interactions
vibration-rotation interaction
The dependence of the rotational constant on the vibrational state n, explains the unequal spacing between lines in vibration-rotation spectra
dissociation energies: De and D0
De - the difference between the minimum of the potential energy curve and the dissociated atoms
D0 - the difference between the ground vibrational level and the dissociated atoms
n’ progression
Series of lines with different vibrational states, shows transitions between energies of lower and upper electronic states. Can be used to get information about vibrational parameters of excited electronic states
Franck-Condon principle
Electronic transitions are vertical; because the motion of the electrons is almost instantaneous relative to the motion of nuclei, when the molecule makes a transition from one electronic state (electron transition), the nuclei do not move appreciably during the transition. The intensity of a vibronic transition is proportional to the overlap between the wave function in the upper and lower vibronic states.
Allows for the estimation of relative intensity of vibronic transitions
principal axes
3 axes i.e. X, Y, and Z, that are chosen so Ixy, Iyz, and Ixz vanish. In molecules with some degree of symmetry i.e. one principal axis is perpendicular per molecular plane in planar molecules, while the C3 axis is one principal axis in CH3Cl
principal moments of inertia
Ixx, Iyy, and Izz
linear, spherical, symmetric, or asymmetric rotor
oblate/prolate symmetric top
centrifugal distortion
normal modes or normal coordinates
stretching (symmetric/antisymmetric), bending, and torsion
parallel/perpendicular band
IR/Raman activity
transition dipole moment
deexcitation processes
radiative/nonradiative transitions
fluorescence
internal conversion
intersystem crossing
phosphorescence
vibrational relaxation
0–0 transition
LASER
stimulated emission
spontaneous emission
Einstein coefficient
fluorescence lifetime
population inversion
pump light
gain medium
pumping source
coherence