Carboxylic Acid: Reactions
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
Acid-metal reaction (redox)
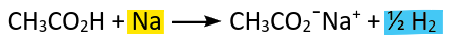
Acid-alkali reaction (redox)

Acid-carbonate reaction (redox)

Why can carboxylic acids undergo nucleophilic substitution while aldehydes and ketones cannot?
C=O bond is polar as O is more electronegative
C=O(carboxylate) carbon carries the partial positive charge and susceptible to nucleophlic attack
the leaving group -OH- is a relatively stable species
For ketones and aldehydes:
H- and R-(alkyl grp) are unstable species and may not be eliminated easily
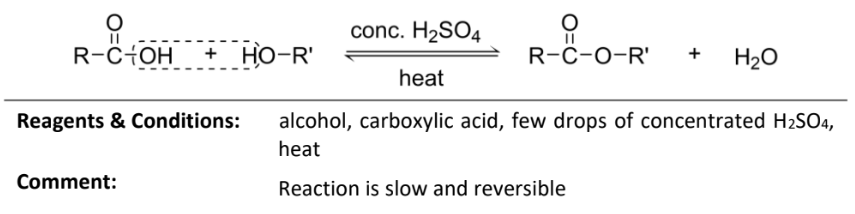
Condensation reaction with alcohol
RnCs: few drops of concentrated H2SO4, heat
Note: reaction is slow and reversible
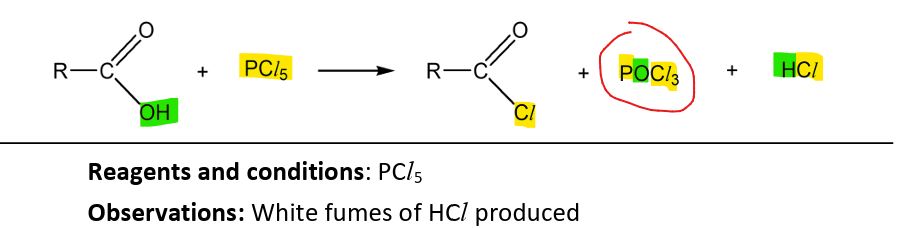
Formation of acyl chlorides
RnCs: PCl5
Observations: Dense white fumes of HCl
Note:
good distinguishing test for alcohols
reaction has same products as the one with alcohol except the acyl chloride
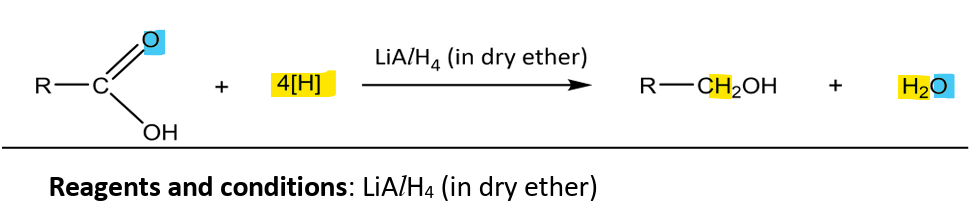
Reduction to primary alcohols
RnCs: LiAlH4 (Lithium aluminium hydride)
Note:
cannot reduce carboxylic acids to aldehydes
weaker reducing agents such as NaBH4 or H2 with catalyst cannot reduce (FYI: lithium is more electronegative than boron, more negative charge is borne by H atom, making LiAlH4 a stronger RA)
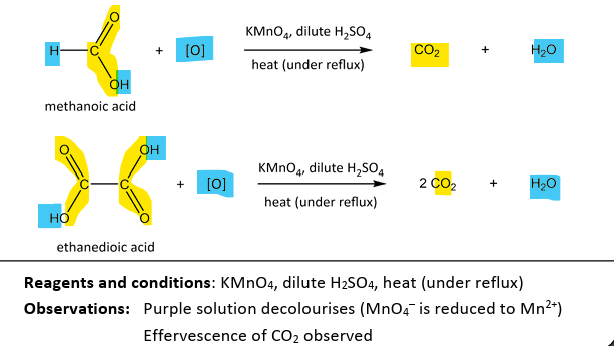
Oxidation of methanoic acid and ethanedioic acid
RnCs: KMnO4, diilute H2SO4, heat (under reflux)
Observation: Purple(MnO4- ) solution declourises(Mn2+)
Note:
other carboxylic acids have no easily oxidisable groups (it is alr very difficult to raise oxidation state +3 of C in -COOH to +4 in CO2)
methanoic acid has a C-H bond that can be oxidised
ethanedioic acid has an unusually weak C-C bond due to electron-wtihdrawing O atoms