Elements, Compounds and Mixtures VMA
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
A substance made of only one kind of matter and having definite properties
Pure substance
A pure substance that cannot be broken down into other substances
Element
The basic particle from which all elements are made
Atom
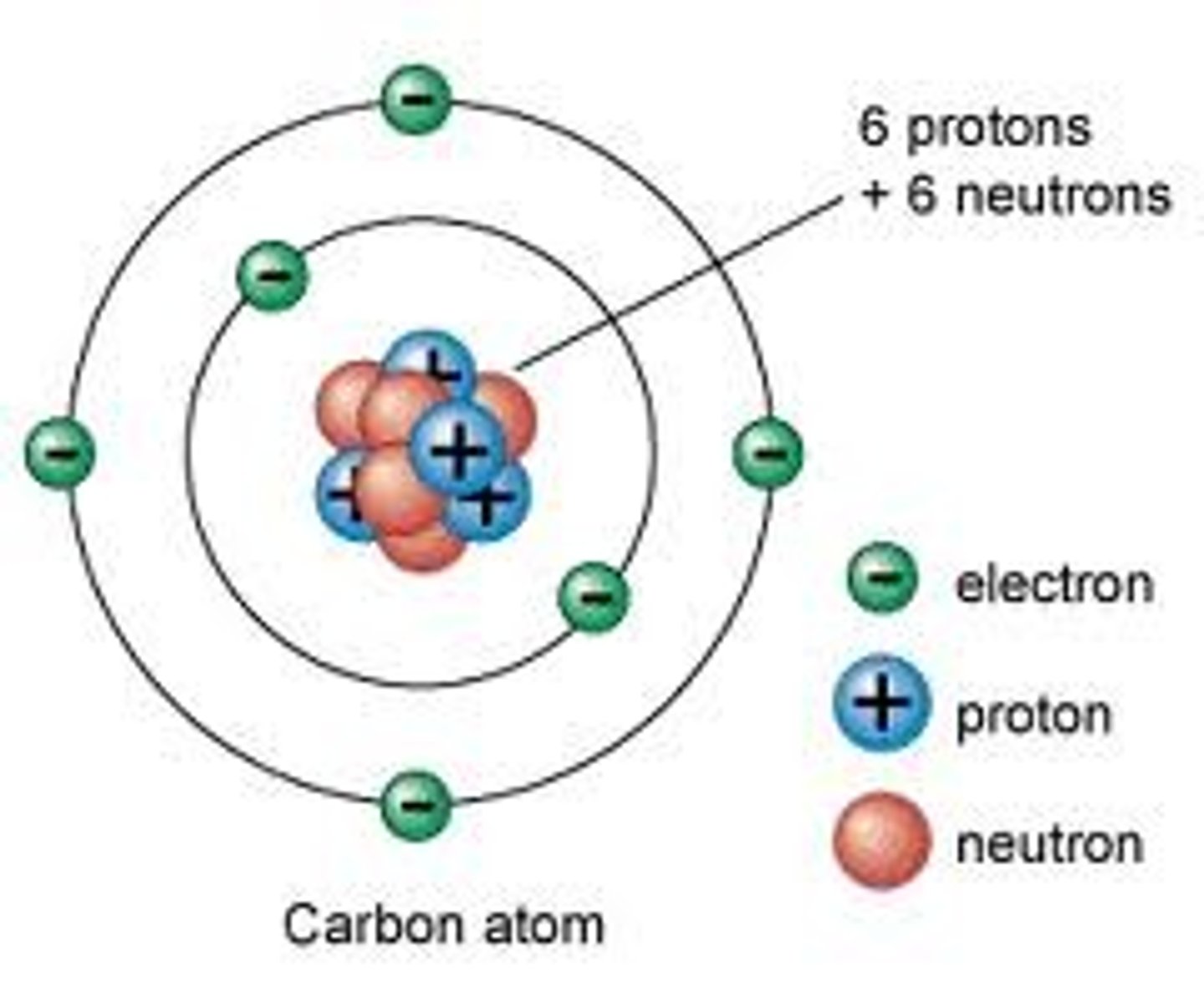
A substance made up of atoms of two or more different elements joined by chemical bonds
Compound
A mixture in which you can see the different parts. They are in different forms/phases
Heterogeneous mixture

A mixture in which you cannot see the different parts Ex milk and water
Homogeneous mixture

A mixture that forms when one substance dissolves another.
Solution
A mixture in which particles can be seen and easily separated by settling or filtration
Suspension
A mixture containing small, undissolved particles that do not settle out.
Colloid
A characteristic of a pure substance that can be observed without changing it into another substance
Physical property

A liquid's resistance to flowing
Viscosity

The temperature at which a solid becomes a liquid
Melting point
The temperature at which a liquid changes to a gas
Boiling point

A process that separates materials based on the size of their particles.
Filtration

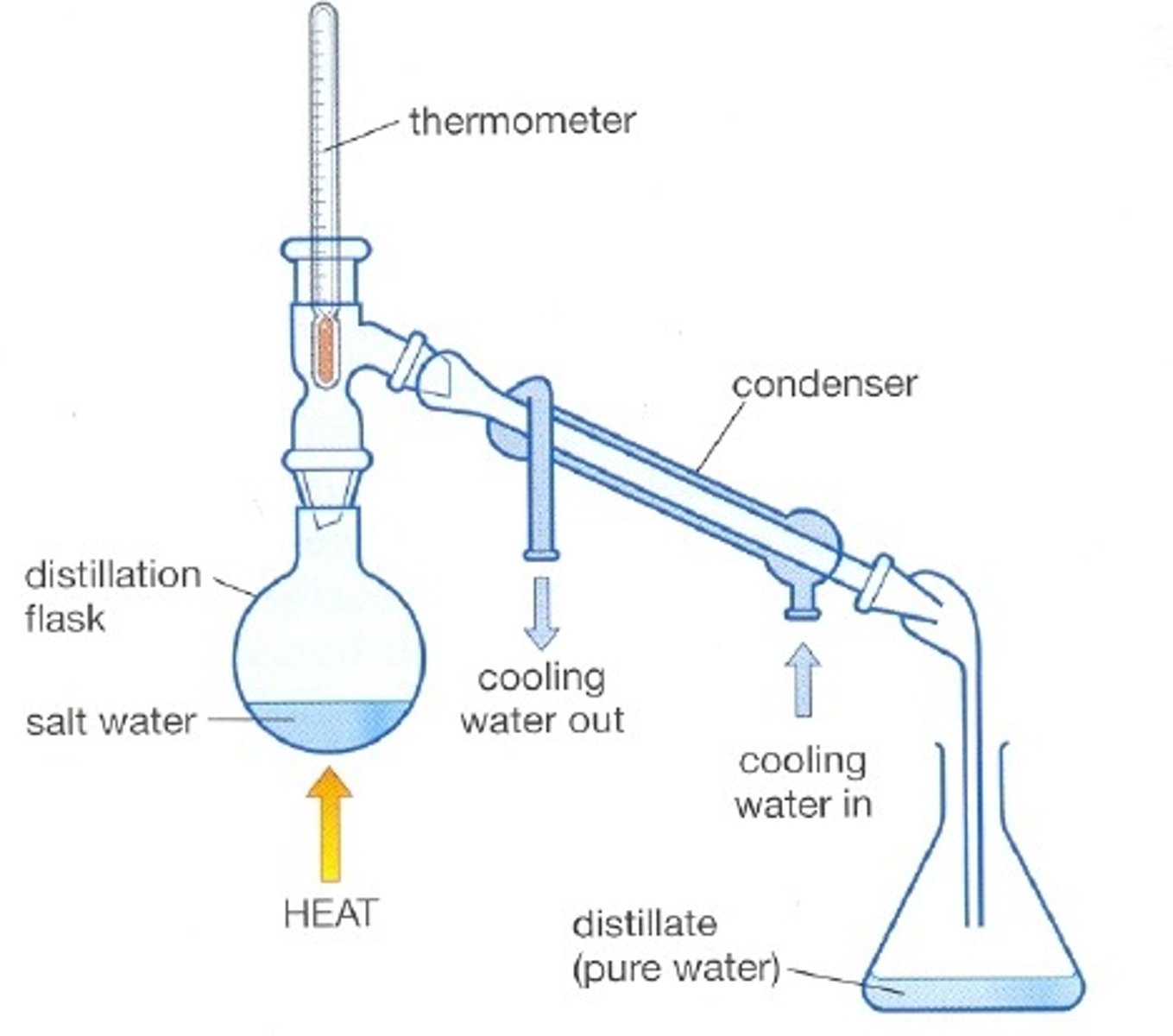
The process of purifying a liquid by boiling it and condensing its vapors
Distillation
A change of matter from one form to another without a change in chemical properties
Physical change

A characteristic of a pure substance that describes its ability to change into different substances
Chemical property
A change in matter that produces one or more new substances
Chemical change

The ability of a substance to burn
Flammability

How readily a substance combines chemically with other substances.
Reactivity

A solid that forms from a solution during a chemical reaction.
Precipitate

any mixture of two or more immiscible liquids in which one liquid is dispersed in the other
Emulsion