Chapter 19: Disorders Associated with the Immune System
1/94
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
95 Terms
What do allergies and asthma have to do with the hygiene hypothesis?
observation that tribal population and children growing up on farms have less prevalence of allergies than children in urban settings
Possibly due to wider range of microbial exposures in farm setting; also led to lower asthma rates
What does inflammatory bowel diseases have to do with the hygiene hypothesis?
possible link of lack of normal microbiota metabolic products leading to chronic inflammatory state
What is dysbiosis?
imbalance that causes adverse effects in humans
What is antibiotic therapy?
kills normal gut microbiota, causing Clostridium difficile to proliferate
What are possible causes of IBDs?
ulcerative colitis and Crohn’s disease
How do scientists treat Crohn’s disease with worms?
Whipworm eggs suppress T helper cell pathways
How do fecal transplants help with C. difficile infections?
taking gut microbiota from a healthy indivigual and transplanting it into the patient
What is hypersensitivity?
antigenic response beyond normal
occurs when sensitized by previous exposure to an antigen (allergen)
What are the four typer of hypersensitivity?
anaphylactic
cytotoxic
immune complex
delayed cell-mediated
What is Immunopathology?
The study of hypersensitivity
What does the hygiene hypothesis say?
limiting exposure to pathogens may lower immune tolerance and the ability to cope with harmless antigens
What is type I hypersensitivity?
(anaphylactic)
<30 minutes
IgE binds to mast cells or basophils; causes degranulation of mast cell or basophil and release of reactive substances such as histamine
Anaphylactic shock from drug injections and insect venom; common allergic reactions such as hay fever, asthma
What do histamines do in type I hypersensitivity?
increase the permeability of blood capillaries
What does Leukotrienes do in type I hypersensitivity?
cause prolonged contraction of blood capillaries
What does Prostaglandins do in type I hypersensitivity?
affect smooth muscle and increase mucus secretion
What is type II hypersensitivity?
(Cytotoxic)
5-12 hours
Activation of complement by the combination of IgG or IgM antibodies with an antigenic cell, causes cell lysis or damage by macrophages
Transfusion reactions, serum sickness
ABO blood group system
antibodies form against certain carbohydrate antigens on RBCs
A antigens, B antigens, or both
Type O RBCs have no antigens
What is the Rh blood group system in type II hypersensitivity?
Rh factor antigen (Rh+) found on RBCs of 85% of the population
Rh+ blood given to an Rh- recipient will stimulate anti-Rh antibodies in the recipient
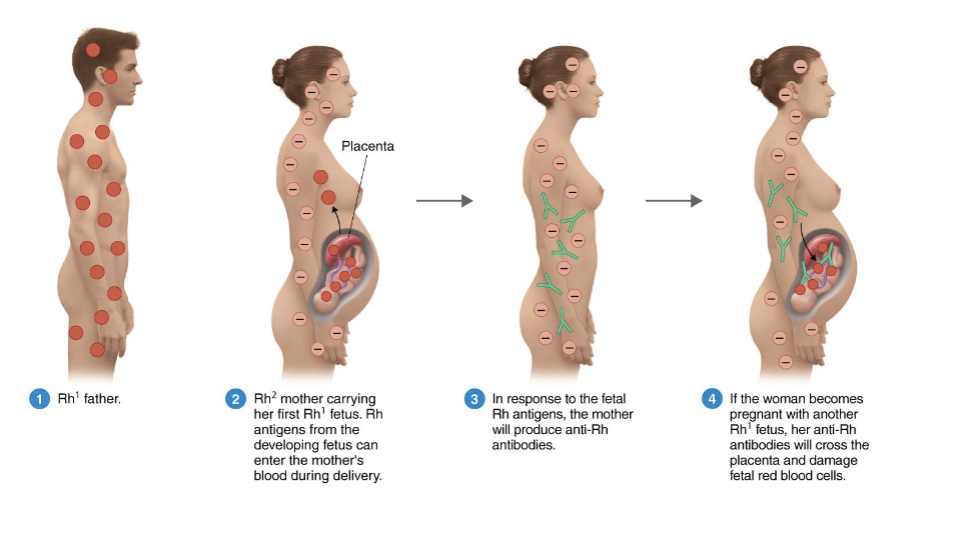
What is hemolytic disease of the newborn (HDNB)?
Rh- mother with an Rh+ fetus causes the mother to produce anti-Rh antibodies
Second Rh+ fetus will receive anti-Rh antibodies, damaging fetal RBCs
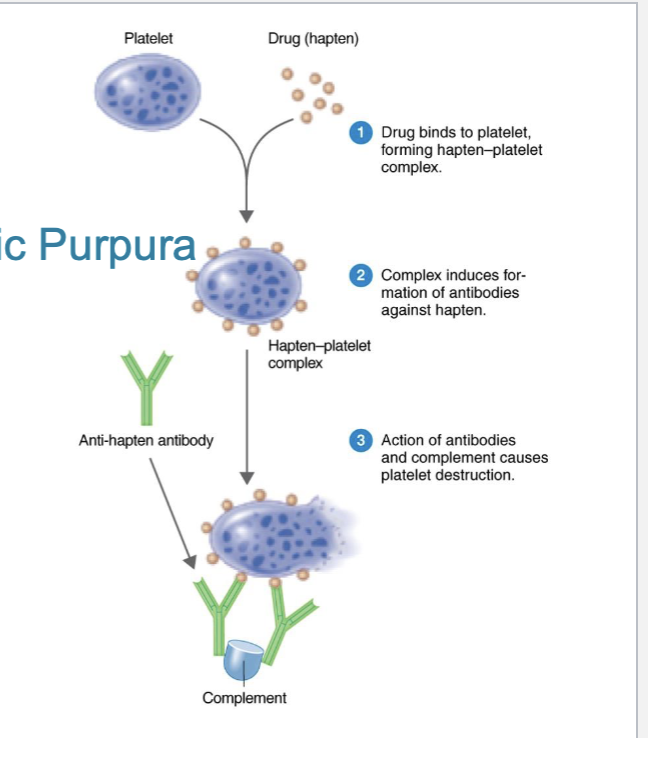
What are drug-induced cytotoxic reactions?
Thrombocytopenic purpura
platelets combine with drugs, forming a complex that is antigenic
antibody and complement destroy platelets
Agranulocytosis
drug-induced immune destruction of granulocyts
Hemolytic anemia
drug-induced immune destruction of RBCs
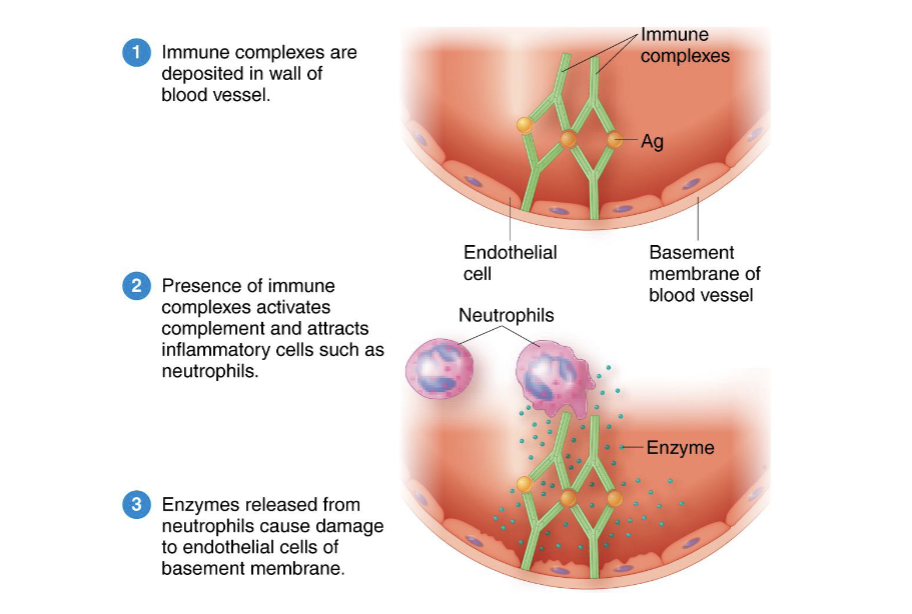
What is type III hypersensitivity?
(Immune complex)
3-8 hours
Antibodies form against soluble antigens in the serum
form immune complexes that lodge in the basement membranes beneath the cells
activate complement, causing inflammation
Arthus reactions, serum sickness
What is Arthus reactions?
Rare side-effect of toxoid-containing vaccines
Occurs in glomeruli and other vessel walls due to complement activation in a patient with already circulating IgG to an injected antigen
What is serum sickness?
occur with swelling and inflammation due to injection of foreign serum
What is type IV hypersensitivity?
(Delayed cell-mediated, or delayed hypersensitivity)
24-48 hours
Antigens are phagocytized and presented to receptors on T cells, causing sensitization
Re-exposure to antigen causes memory cells to release destructive cytokines
What is allergic contact dermatitis?
haptens combine with proteins in the skin, producing an immune response
allergic response to poison ivy, cosmetics, metals, and latex
What is Systemic anaphylaxis?
(anaphylactic shock)
results when an individual sensitized to an antigen is exposed to it again
may result in circulatory collapse and death
treated with epinephrine
What is localized anaphylaxis?
usually associated with ingested or inhaled antigens
symptoms depend on route of entry
hives, hay fever, and asthma
How do you prevent anaphylactic reactions?
Antigens are inoculated beneath the epidermis to test for a rapid inflammatory reaction (wheal)
What is desensitization?
increasing dosages of antigen injected beneath the skin
produces IgG, which act as blocking antibodies to intercept and neutralize antigens
What are autoimmune diseases?
immune system responds to self antigens, causing damage to the organs
Autoimmunity is loss of self-tolerance
ability to discriminate self from non-self
Cytotoxic, immune complex, or cell-mediated
What are cytotoxic autoimmune reactions?
Multiple sclerosis
autoantibodies, T cells, and macrophages attack myelin sheath of nerves
compromises nerve impulse transduction
symptoms range from fatigue and weakness to severe paralysis
Etiology unknown; may involve genetic susceptibility and/or infective agent(s)
What are immune complex autoimmune reactions?
antibodies react with cell-surface antigens
graves’ disease
abnormal antibodies in the thyroid produce excessive amounts of hormones
Myasthenia gravis
antibodies coat acetylcholine receptors; muscles fail to receive nerve signals
Immune complexes of antibodies and complement deposit in tissues
Systemic lupus erythematosus
immune complexes form in kidney glomeruli
Rheumatoid arthritis
immune complexes form in the joints
What are cell-mediated autoimmune reactions?
mediated by T cells that attack tissues
insulin-dependant diabetes mellitus
T cell destruction of insulin-secreting cells
Psoriasis and psoriatic arthritis
autoimmune disorders of the skin
What are Histocompatibility antigens?
self antigens on cell surfaces
What is the major histocompatibility complex (MHC)?
genes encoding histocompatibility antigens
What is the HLA complex?
MHC genes in humans
What are certain HLAs related to?
increased susceptibility to specific diseases
What are the selected diseases related to specific human Leukocyte antigens?
Multiple sclerosis
Rheumatic fever
Addison’s disease
Graves’ disease
Hodgkin’s lymphoma
What does HLA typing do?
identifies and compares HLAs
What has to happen in tissue surgery?
The donor and the recipient must be matched by tissue typing
Uses standardized antisera or monoclonal antibodies specific for HLAs
What is a risk for transplants?
They may be attacked by T cells, macrophages, and complement-fixing antibodies
What is true about transplants and privileged sites?
transplants to privileged site and privilege tissue do not cause immune response
cornea transplants. heart valve transplants
What are stem cells?
master cells capable of renewing themselves and differentiating into organ-specific specialized cells
What are embryonic stem cells (ESCs)?
harvested from blastocysts; used to regenerate tissues and organs
Pluripotent-can generate all types of cells
What are adult stem cells?
stem cells in adult tissues that have differentiated
can become induced pluripotent stem cells by introducing genes
What are hematopoietic stem cell transplants?
AKA bone marrow transplants
goal is to enable the recipient to produce healthy blood cells
graft-versus-host (GVH) disease can result from transplanted bone marrow that contain immunocompetent cells
What is an autograft?
use of one’s own tissue
What is an Isograft?
Use of identical twins tissue
What is an Allograft?
Use of tissue from another person
What is a Zenotransplantation product?
Use of nonhuman tissue
must overcome hyperacute rejection: response to nonhuman antigens
What is Ummunosuppresion to prevent transplant rejection?
Prevents a cell-mediated immune response to transplanted tissues
What do Cyclosporine and tacrolimus do?
suppress IL-2, disprupting cytotoxic T cells
What does Sirolimus do?
inhibits cellular and humoral immunity
What does Mycophenolate do?
inhibits the proliferation of T cells and B cells
What does Basiliximab do?
chimeric monoclonal antibody that blocks IL-2
How are cancer cells removed?
immune surveillance
What kind of antigens do Cancer cells have?
tumor-associated antigens that mark them as nonself
What do CTLs (activated Tc cell) and Macrophages do to cancer cells?
lyse them
What are the limitation to the immune system and cancer?
no antigenic epitope for the immune system to target
Tumor cells reproduce too rapidly
Tumor becomes vascularized and invisible to the immune system
What do endotoxins do to cancer?
(corey’s toxins) stimulate TNF that interferes with the blood supply of cancers
What are vaccines used for prophylaxis?
cervical, anal, and throat cancer (HPV), liver cancer (hepatitis B)
What are monoclonal antibodies?
Herceptin for breast cancer
Immunotoxin combines a Mab with a toxic agent
Targets and kills a tumor without damage to healthy cells
What are immunodeficiencies?
absense of a sufficient immune response
What are congenital immunodeficiencies?
due to defective or missing genes
What are acquired immunodeficiencies?
develop during an individuals life
due to drugs, cancers, or infections
What are acquired immunodeficiency syndrome?
virus causing the loss of immune function (HIV), selectively infects helper T cells
What is the structure of HIV?
Genus Lentivirus
Retrovirus
Two identical + stranded RNA genome molecules, reverse transcriptase enzyme, phospholipid envelope
gp120 glycoprotein spikes
How is HIV spread?
dendric cells and carried to the lymphoid organs; contacts activated T cells
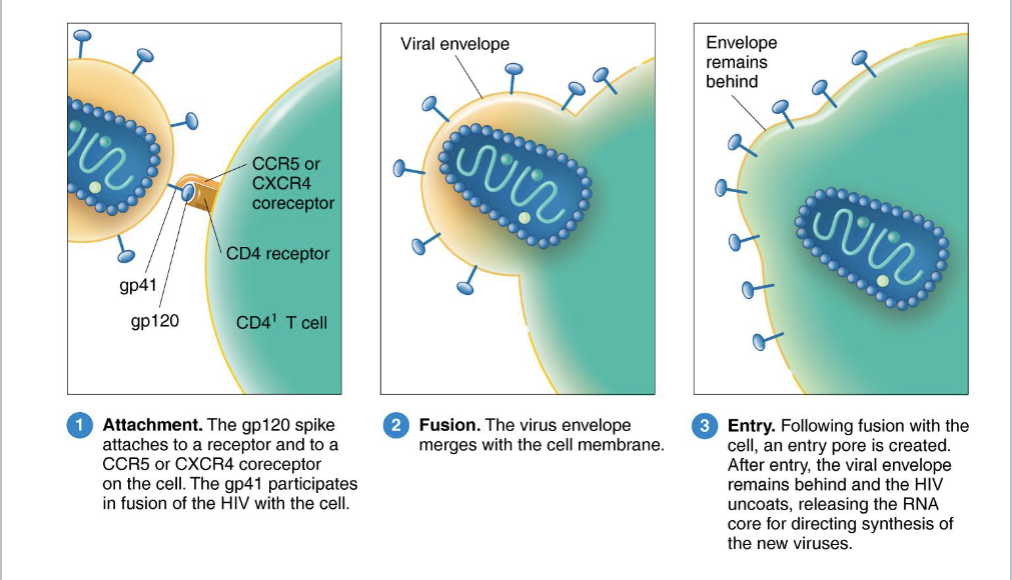
What does HIV do inside the cell?
viral RNA is transcribed into DNA using reverse transcriptase
What happens to DNA when HIV gets into the cell?
integrated into the host’s DNA
What is Active infection?
new viruses bud from the host cell
What is latent infection?
DNA is hidden in the chromosome as a provirus
some become memory T cells that serve as the reservoir for HIV
What does the virus undergo inside the cell?
rapid antigenic changes and a high rate of mutation
What is HIV-1?
related to viruses that infect chimpanzees and gorillas
99% of cases
Group M (majority) accounts for 90%
What is HIV-2?
not often encountered outside of west Africa
Less pathogenic than HIV-1
Longer asymptomatic period with lower viral load and morality rate than HIV-1
What is Phase 1 of HIV infeciton?
Asymptomatic or lymphadenopathy
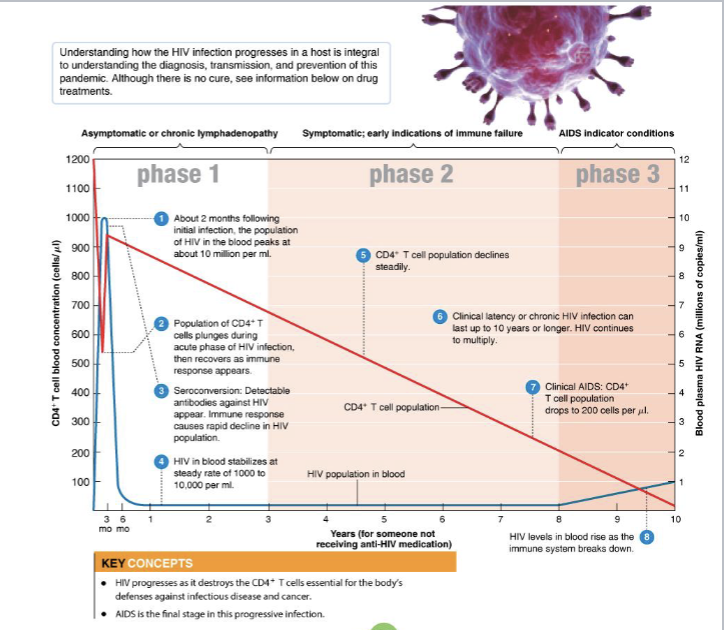
What is phase 2 of HIV infection?
CD4+ T cells decline steadily; only a few infected cells release the virus; few serious disease symptoms (persistent infections, fever, and oral leukoplakia).
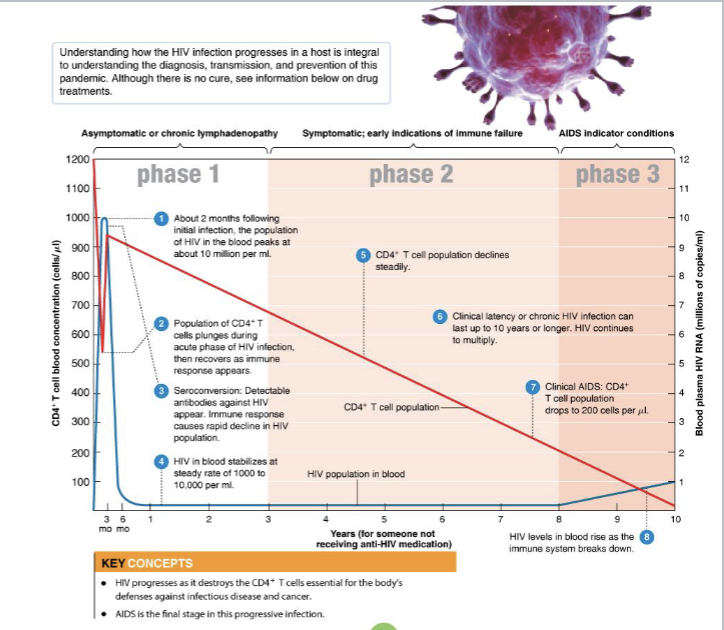
What is phase 3 of HIV infection?
AIDS develops; the CD4+ is count below 200 cells/μl; indicator conditions [healthy 500 to 1500 CD4+ T cells/μl]
![<p>AIDS develops; the CD4+ is count below 200 cells/μl; indicator conditions [healthy 500 to 1500 CD4+ T cells/μl]</p>](https://assets.knowt.com/user-attachments/ffb9e7fb-6b61-4d57-8aaf-e4e817066f69.png)
What happens once the HIV establishes the pool of latenly infected CD4+ T cells?
it is impossible to clear the infection
What is the impact of age of survival with HIV infection?
Older adults and young children do not a have fully developed immune system, making them more susceptible
What is the Seroconverion?
Period of time between infection and the appearance of antibodies
How are HIV antibodies detected?
ELISA
How are viruses detected
Western blotting or APTIMA (RNA testing)
How are plasma viral load (PVL) determined?
by PCR or nucleic acid hybridization
How long does HIV survive outside the cell
6 horus
How long does HIV survive inside the cell?
1.5 days
What are the routes of transmission of HIV?
sexual contact, breast milk, transplacental infection of a fetus, blood-contaminated needles, organ transplant, artificial insemination, and blood transfusion
anal intercourse is the worst
What are biomedical intervesiton on preventing HIV?
use of condoms, health services/HIV testing, needle programs
What are the behavioral intervention of preventing HIV?
sex education, safe infant feeding programs, counseling
What are structural interventions?
making changes in social, economic, political, and environmental factors to reduce vulnerability to HIV
What is highly active antiretroviral therapy (HAART)?
use of combinations of drugs to minimize survival of resistant strains.
What are fusion/entry inhibitors?
Targets the gp41 region of the viral envelope that prevents fusion of the virus with the cell
Enfuvirtide and maraviroc
What are reverse transcriptase inhibitors?
nucleoside reverse transcriptase inhibitors (NRTIs)
Tenofovir and emtricitabrine
Non-nucleoside reverse transcriptase inhibitors (NRTIs)
Efavirenz
What are Integrase inhibitors?
inhibit HIV integrase that integrates cDNA into the host chromosome
Raltegravir, dolutegravir, elvitegravir
What are protease inhibitors
inhibit proteases that cleave viral precursor proteins into structural and functional proteins
Atazanacvir, indinavir, and saquinavir
What are Tetherins?
Tether viruses to the cells, preventing their release and spread