proteins 2
1/73
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
74 Terms
What is a dipeptide?
Two amino acids joined by a peptide bond.
formed by a condensation reaction
What is a polypeptide?
Polymer chain of many amino acids linked by peptide bonds.
via condensation reactions
What is quaternary structure?
More than one polypeptide chain e.g. haemoglobin
Formed by interactions between polypeptides
(hydrogen bonds, ionic bonds, disulfide bridges)
What is the induced-fit model?
The active site is not initially complementary to the substrate
Active site moulds around substrate on binding.
What is the effect of pH
on enzyme-controlled
reaction?
Too high or too low a pH will interfere with the charges in the amino acids in the active site.
This breaks the ionic and hydrogen bonds holding the tertiary structure in place
therefore the active site changes shape and the enzyme denatures
Different enzymes have a different optimal pH
How does enzyme concentration affect rate of reaction?
At low enzyme concentrations, there will be fewer collisions between the enzyme and substrate.
At high enzyme concentrations, the rate plateaus because there are more enzymes than the substrate, so there are empty active sites.
How does substrate concentration affect rate of reaction?
At low substrate concentrations, there will be fewer collisions
between the enzyme and substrate.
At high substrate concentrations, the rate plateaus because all the enzyme active sites are saturated
What is a competitive inhibitor?
A molecule that is similar shape to substrate binds to the active site.
What is a non-competitive inhibitor?
A molecule that binds to an enzyme at the allosteric site
causing the active site to change shape
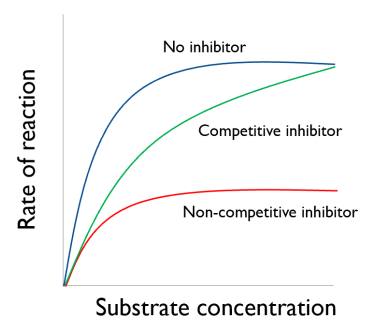
Inhibitor Graphs
With a high enough substrate concentration, the competitive inhibitors are knocked out of the active site and the rate of reaction will return to the same as with no
inhibitor.
The rate of reaction with a non-competitive inhibitor will be lower at all substrate concentrations.
Why do different enzymes have different optimals?
Their structures suit different cellular environments.