Ozone depletion (3.3.3.3)
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
What is ozone is why is it beneficial?
Ozone, O3, is useful as it absorbs UV radiation, which is harmful.
How do CFCs lead to ozone depletion?
CFCs absorb a lot of UV radiation, which causes homolytic fission of the C-Cl bond (not C-F as stronger), forming radicals that react with ozone and cause it to decompose.
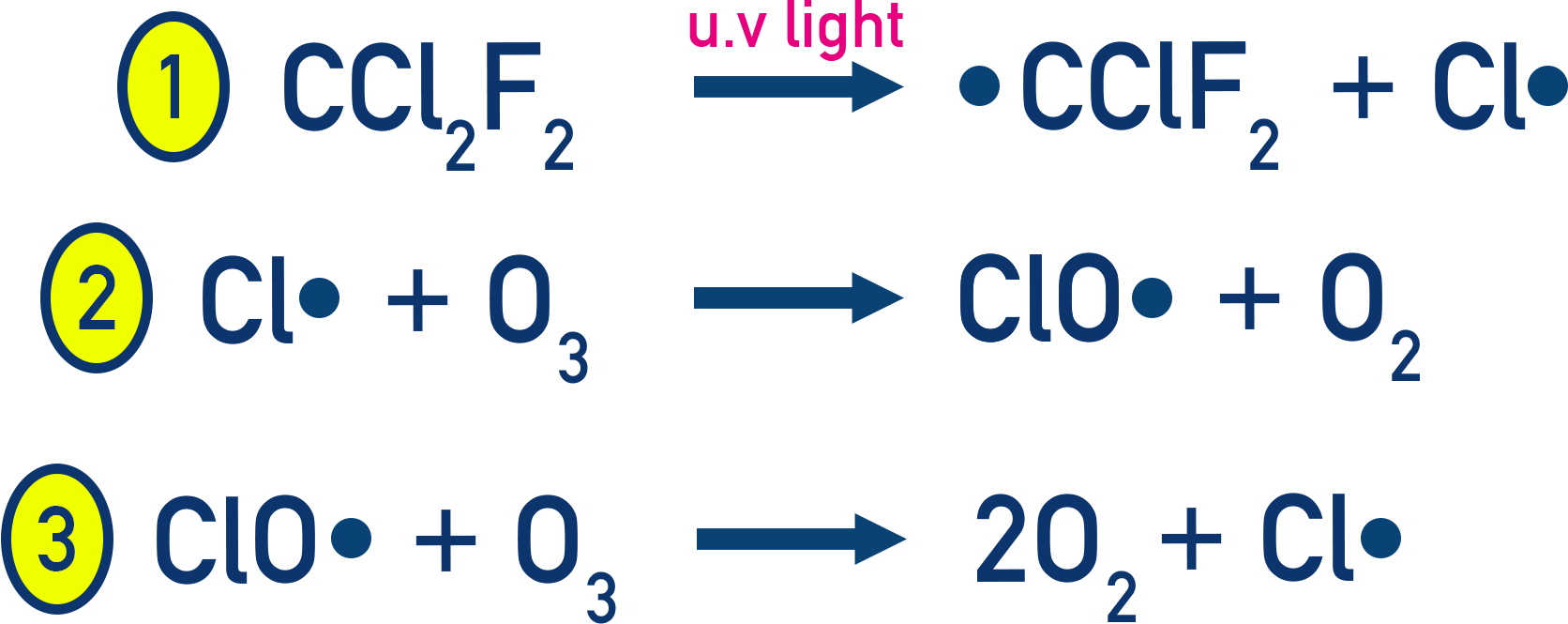
What is the mechanism and overall reaction of the depletion of ozone?
1) Initiation
CF2Cl2 → C. F2Cl + Cl.
2) Propagation
O3 + Cl. → O2 + .ClO
.ClO + O → O2 + Cl.
Overall equation
O3 + O → 2O2
Also:
What is another reaction that causes ozone depletion?
1) Initiation
N2 + O2 → 2NO.
2) Propagation
NO. + O3 → O2 + .NO2
.NO2 + O → NO. + O2
Overall equation
O3 + O → 2O2
What has been done to prevent the depletion of ozone?
CFCs were banned
CFC-free (HFCs) solvents are being produced to help minimise ozone depletion and global warming