Synaptic Transmission 1
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
synaptic transmission chemical process
electrical signal from AP in presynaptic terminal, influx of Ca2+, release of vesicles, binding of neurotransmitters to receptors in postsynaptic membrane, ion channels open resulting in a flow of ions, result in Action potential (if excitatory)
Model synapses
Neuromuscular Junction, Calyx of Held, Squid giant synapse
the neuromuscular junction
the interface between your nervous and skeletomuscular systems.

the motor end plate
presynaptic specializations formed by motor axons that branch from ventral horn in spinal cord to reach muscle fiber
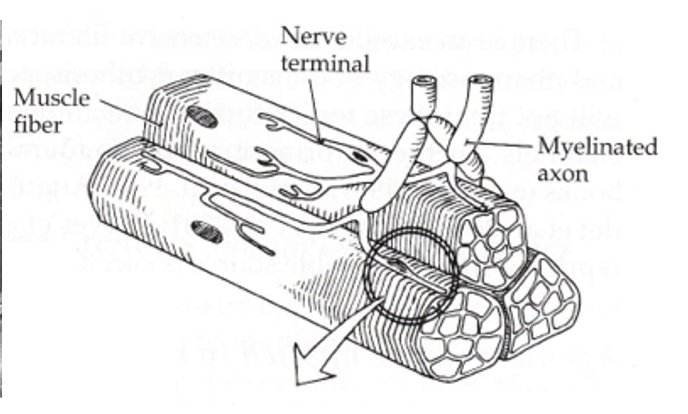
one way traffic
alpha motor neurons can have outputs to more than one fiber, while muscle fibers can only have one motor end plate each
junctional folds
the motor end plate makes many contacts with the muscle cell, which has several membrane invaginations called junctional folds just beneath presynaptic release sites
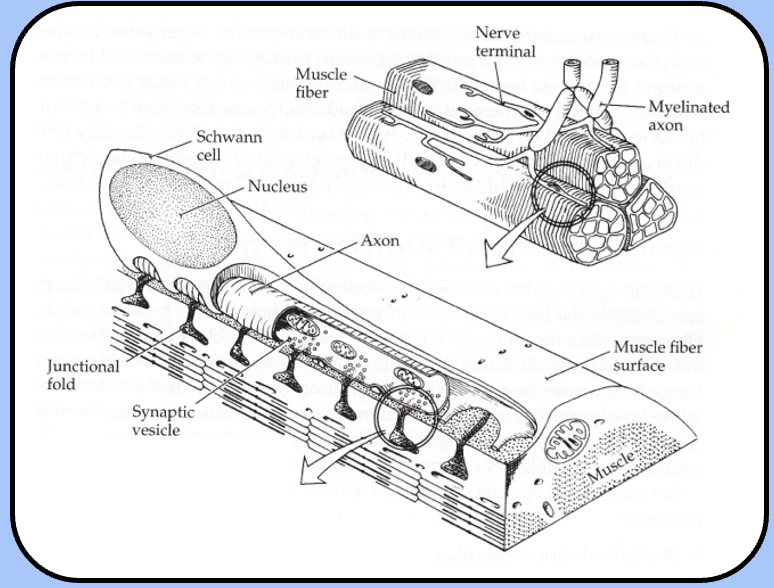
ultrastructure of NMJ
basal lamina is positioned in the synaptic cleft: fuzzy structure formed from the ectodermal and mesodermal germ layer interactions
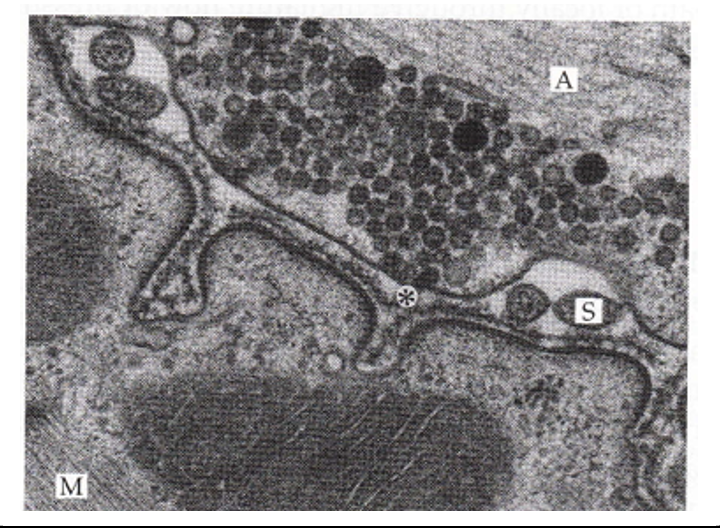
the post synaptic membrane of the NMJ
nicacetyl choline receptors (nAChRs) scaffold/support proteins and sodium channels
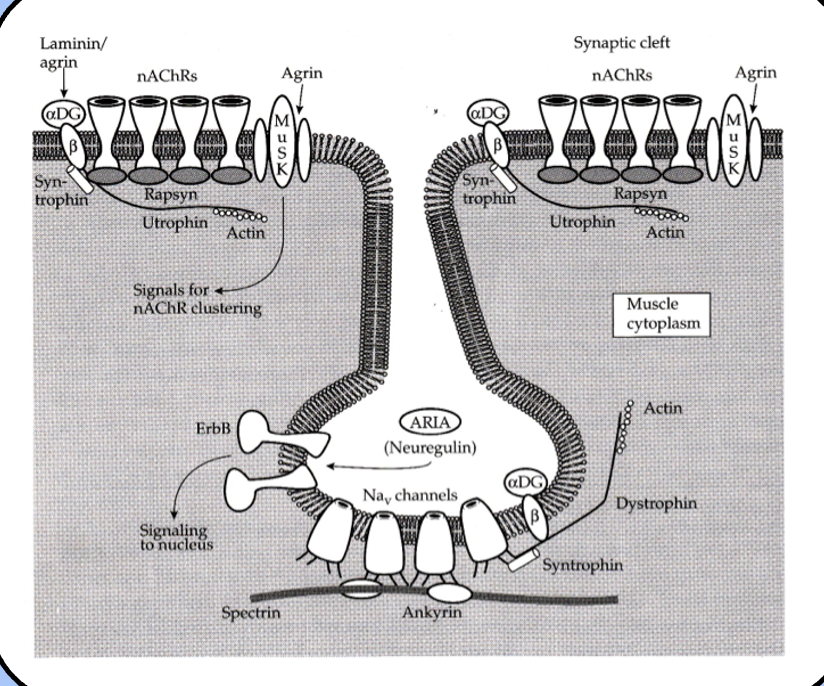
T-tubule system
although the junctional folds hold plenty of ion channels, they aren’t sufficient to spread the action potential to the entire muscle fiber.
a sophisticated network of membrane invaginations called the T-tubule system carries the action potential to every “nook and cranny,” of the fiber.
the T-tubules are essentially inside-out axons.
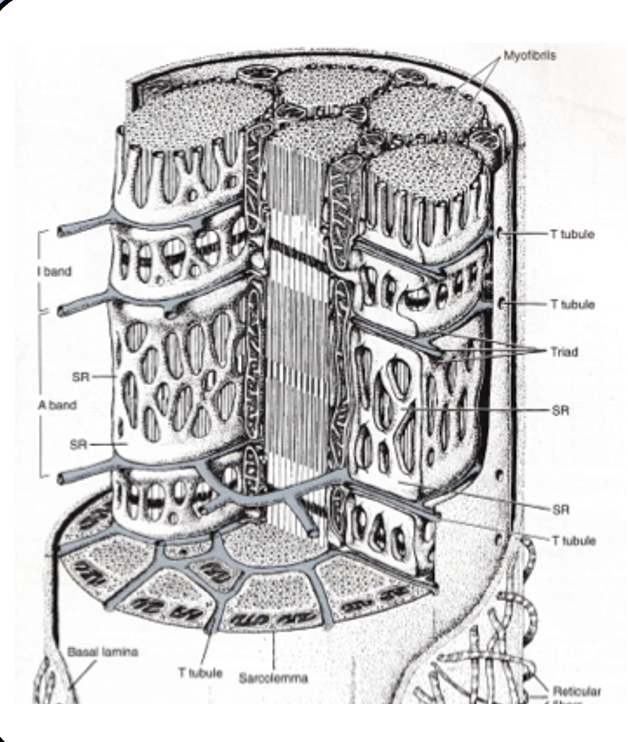
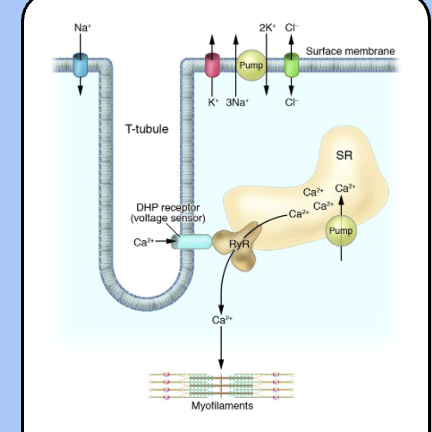
excitation-contraction coupling
the action potential travels throughout the T-tubule system to trigger the contraction of the muscle fiber.
depolarization activates an L-type voltage-gated calcium channel (often called the dihydropyridine receptor), which is physically coupled to a calcium channel located on the SER membrane called the ryanodine receptor (RyR).
once the ryanodine receptor opens calcium ions flood the cytoplasm and interact with the contractile apparatus of the sarcomere.
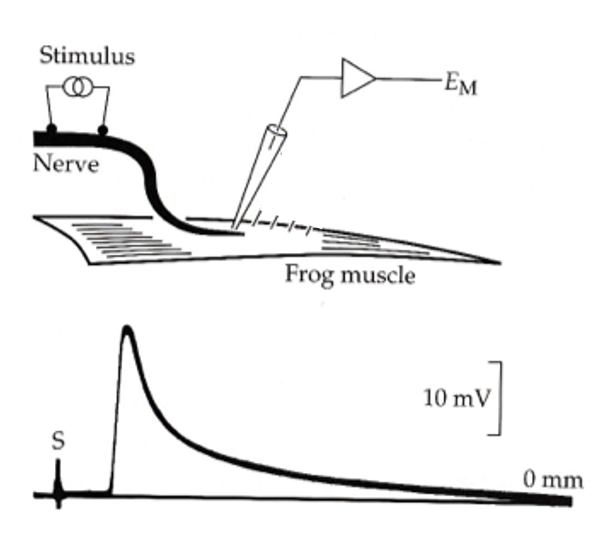
the intracellular glass microelectrode
glass capillaries could be pulled to an extremely sharp point, filled with KCl, and connected to external amplifiers through a silver wire.
how were action potentials prevented/held below threshold?
tubocurarine is commonly used to limit the size of the muscle cell’s response below threshold.

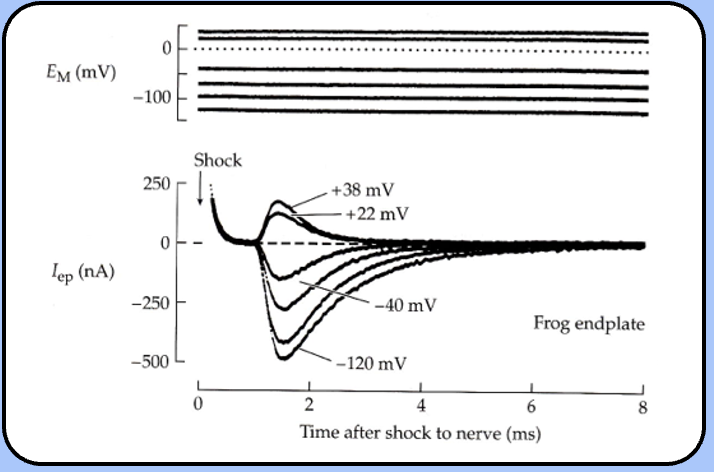
the end plate potential
- a shock to the nerve elicits a depolarization in the muscle cell called the end plate potential (EPP)—because it is largest near the motor end plate.
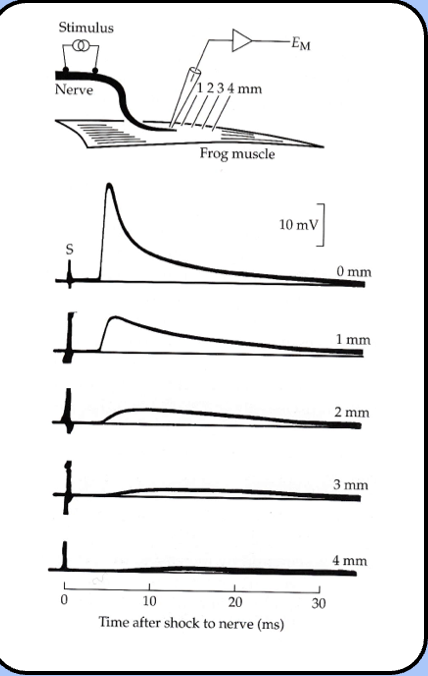
location dependence of the EPP
Katz proposed that the EPP resulted from an influx of current near the end plate
Katz’s observations of the EPP
- Katz noticed that the EPP decayed according to the membrane time constant of the muscle cell.
- he also noticed that the EPP decreases as the microelectrode is moved away from the motor end plate—it also becomes wider like any propagated signal experiencing electrical filtering.
the End Plate Current
- if the membrane potential is held constant by a voltage-clamp circuit, a shock to the nerve elicits a postsynaptic current called the end plate current (EPC).
calcium dependence of the EPP
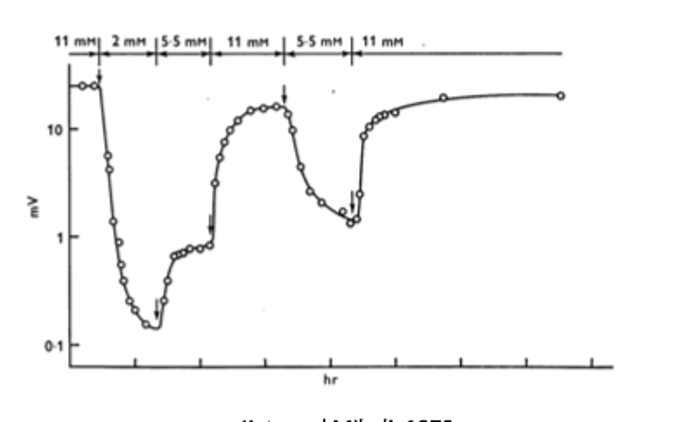
- one early observation of the EPP was that its magnitude was dependent on the concentration of extracellular calcium.
Ca2+ binding partner
calcium was hypothesized to influence the EPP by binding to an intracellular binding partner (X) to form the complex CaX—the mysterious CaX complex was thought to be related to the EPP through a proportionality constant (k).
if neurotransmitter release is proportional to [CaX], than the EPP should scale linearly—if release depends on multiple CaXs, then the EPP should scale according to [CaX]n.
![<ul><li><p><span style="font-family: "Helvetica Light";">calcium was hypothesized to influence the EPP by binding to an intracellular binding partner (X) to form the complex CaX—the mysterious CaX complex was thought to be related to the EPP through a proportionality constant (k).</span></p></li></ul><ul><li><p><span style="font-family: "Helvetica Light";">if neurotransmitter release is proportional to [CaX], than the EPP should scale linearly—if release depends on multiple CaXs, then the EPP should scale according to [CaX]<sup>n</sup>.</span></p></li></ul><p></p>](https://assets.knowt.com/user-attachments/8987cc80-0a9a-475b-84c6-7aad4474cc21.png)
relationship between EPP magnitude and Ca2+
non-linear meaning that multiple CaX’s were required for neurotransmitter release
EPP as a function of Ca2+
EPP = k[CaX]4
suggesting that there are four CaX complexes that work cooperatively to trigger neurotransmitter release
![<p><span style="font-family: "Helvetica Light";">EPP = k[CaX]<sup>4</sup></span></p><ul><li><p>suggesting that there are four CaX complexes that work cooperatively to trigger neurotransmitter release</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/14303820-083c-4cb1-9892-e0a325b02c84.png)
the squid giant synapse
using the squid giant synapse to observe how Ca2+ entered the presynaptic terminal
presynaptic calcium currents
when the presynaptic terminal is depolarized, voltage-gated calcium channels mediate an inward calcium current that increases as the channel opens, but decreases as the membrane potential approaches the reversal potential.
interestingly, the largest calcium currents are associated with the offset of the depolarizing stimulus
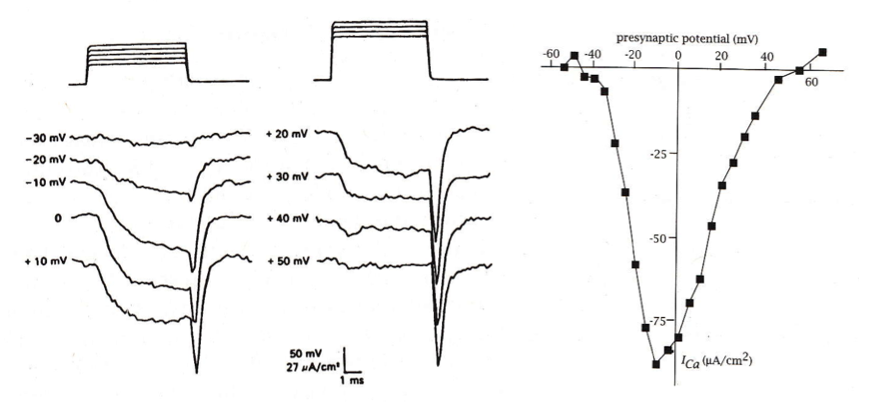
why are the calcium currents so large at the offset of the voltage stimulus?
what happens to calcium currents when the membrane potential is stepped to the reversal potential?
only the ‘off-response’ is observed- the massive influx in inward current
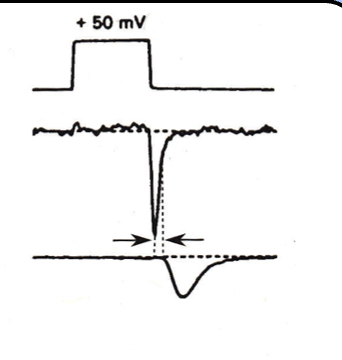
what happens in simultaneous pre- and postynaptic recordings of the calcium current in response to a step depolarization?
in simultaneous pre- and postsynaptic recordings, the calcium current flowing in response to a step depolarization increases slowly, and does not induce a large PSC—the PSC actually reaches its peak after the voltage step.
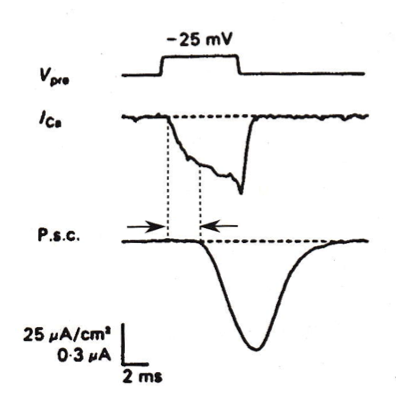
cooperativity between Ca2 and the PSC
the slope of the log-log plot between current and [Ca2+] is near 1 or slightly sub linear for the calcium current, and about 4 for the PSC—this is consistent with the notion that the presynaptic calcium current is not the source of cooperativity for neurotransmitter release.
![<p><span style="font-family: "Helvetica Light";">the slope of the log-log plot between current and [Ca<sup>2+</sup>] is near 1 or slightly sub linear for the calcium current, and about 4 for the PSC—this is consistent with the notion that the presynaptic calcium current is not the source of cooperativity for neurotransmitter release.</span></p>](https://assets.knowt.com/user-attachments/1179e5a9-617f-45be-8b01-7df4d4b465c2.png)
calcium influx during the presynaptic action potential
- the calcium conductance slowly rises, and reaches its peak during the afterhyperpolarization (note that gCa had to be multiplied by 50 in order to appreciate it alongside gNa and gK.
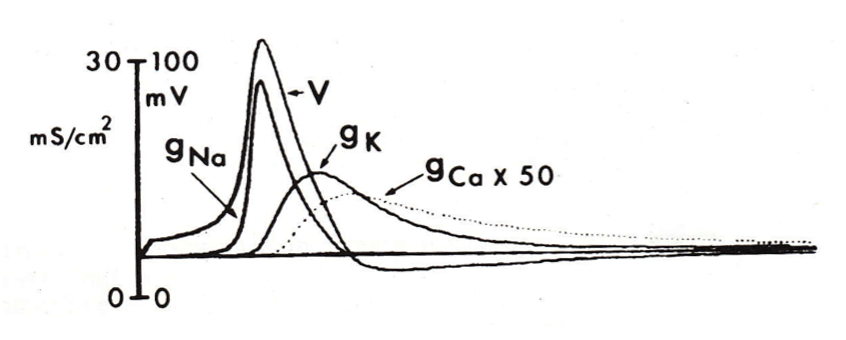
calcium and neurotransmitter release
- once the presynaptic action potential has triggered an influx of calcium, several calcium ions bind to the presumed calcium sensor and trigger the process of neurotransmitter release.
two main types of VGCCs
- the most basic system is the distinction between high voltage activated (HVA) and low voltage activated (LVA) channels.
HVA channel types
- HVA channels fall into two groups, the L-type channels and the P/Q-, N-, and R-type channels.
LVA channels categories
also called T-type channels
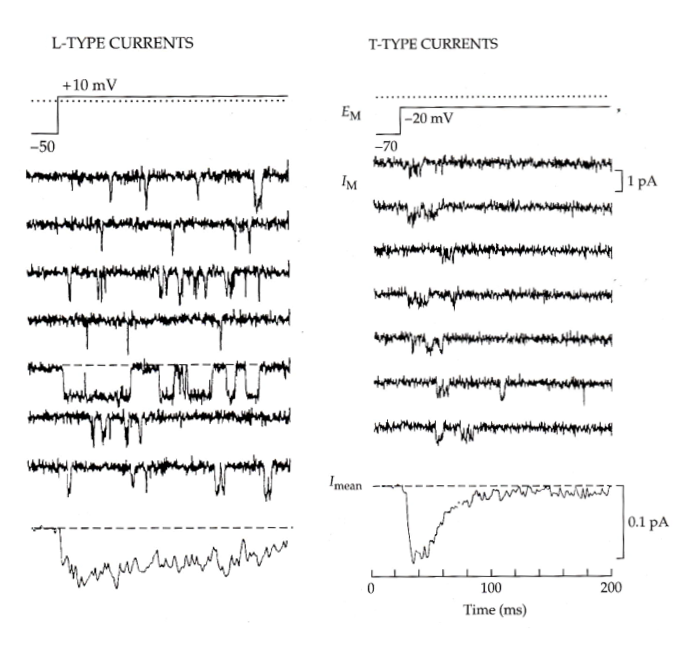
L-type vs T-type currents
Ensemble average of all these currents to get the macroscopic current (graph at the bottom)
-evidence of inactivation from unitary currents as opposed to the macroscopic current
- We see the current flatline in the second half, meaning a lower probability of opening- different from l-type which is dispersed throughout
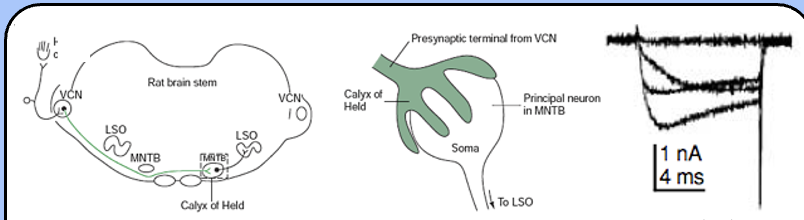
the Calyx of Held
an unusually large synapse in the auditory brainstem—it connects the ventral cochlear nucleus (VCN) with the medial nucleus of the trapezoid body (MNTB).
the presynaptic calcium currents in the calyx of Held activate fairly rapidly and inactivate (at least partially)
w-agatoxin IVA
IVA specifically binds to a subclass of HVA calcium channels called P/Q-type channels, or Cav2.1
how to determine which calcium channels are most present
only a small fraction of the presynaptic calcium current is sensitive to ω-CgTx, whereas the vast majority of this current is sensitive to ω-AgTx—P/Q-type calcium channels seem to be the dominant component of the presynaptic current.
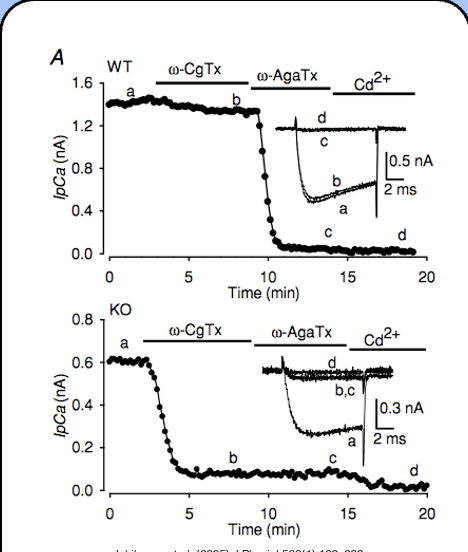
blocking N-type calcium channels
- ω-conotoxin GVIA (ω-CgTx)
blocking P/Q-type calcium currents
ω-agatoxin IVA (ω-AgTx) specifically blocks P/Q-type calcium channels, and Cd2+ blocks all calcium channels
topology of VGCCs
Psuedotetrameric like sodium channels, but has beta subunits that interact with the channels (auxillary subunits) and modulate the properties of the channel.
A2 delta:
intracellular beta subunit
Gamma subunit
tetraspannin
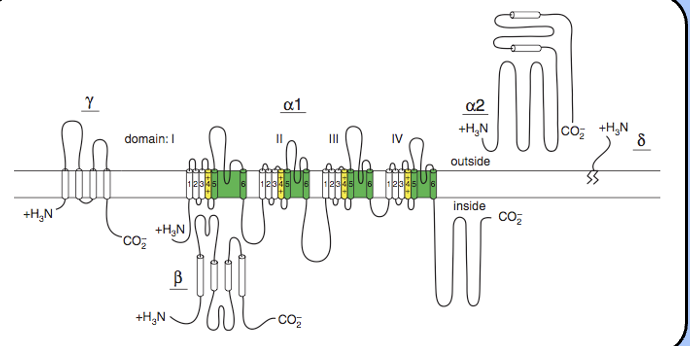
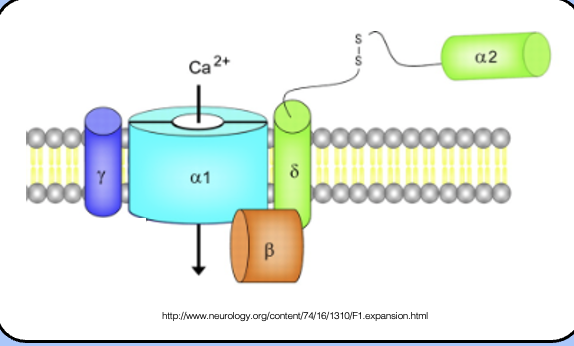
The VGCC and its subunits
- VGCCs don’t act alone—instead, they form large multimeric complexes with several β subunits.
- VGCC a subunits are accompanied by β, γ, and an α2-δ subunit—the details of these interactions are not fully understood.
how exactly does the Action potential contribute to the Ca2+ change in conductance?
The action potential induces channel openings, depolarization induces channel openings, AHP occurs but Ca2+ channels are still open, so the driving force becomes much larger, postsynaptic ‘off’ response is observed
why is the post synaptic ca2+ off response so fast
Bc the channels are closing with hyperpolarization- the response is only observed bc the change in driving force/hyperpolarization is so much fastr than closing of channels